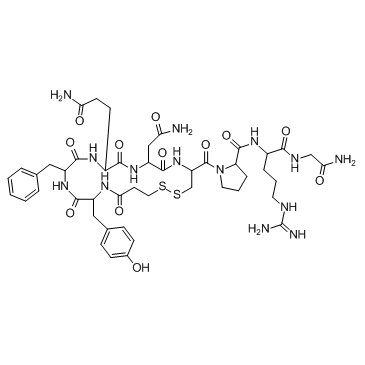
Desmopressin Acetate
- CAS No.16679-58-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Desmopressin Acetate API for pharmaceutical formulation. Meets strict pharmacopoeia standards for global distribution and licensed manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
Desmopressin Acetate is a sophisticated synthetic peptide analog designed for high-level pharmaceutical applications. As a modified form of the natural hormone vasopressin, this compound exhibits enhanced antidiuretic activity with significantly reduced pressor effects. Our manufacturing process ensures that every batch meets rigorous international standards for purity and stability, making it an ideal choice for licensed pharmaceutical producers seeking reliable active pharmaceutical ingredients.
We specialize in the large-scale production of complex peptide structures, utilizing advanced solid-phase synthesis techniques to guarantee consistency. This product is intended strictly for B2B transactions with qualified entities possessing the necessary regulatory approvals for handling prescription-grade materials. Our commitment to quality ensures that downstream formulations maintain efficacy and safety profiles required by global health authorities.
Technical Specifications
The following table outlines the critical physical and chemical parameters verified through our internal quality control laboratories. Each certificate of analysis provides detailed chromatographic data to confirm identity and potency.
| Parameter | Specification |
|---|---|
| CAS Number | 16679-58-6 |
| Molecular Formula | C46H64N14O12S2 |
| Molecular Weight | 1069.217 g/mol |
| Appearance | White to off-white powder |
| Assay (HPLC) | ≥98.0% |
| Density | 1.6±0.1 g/cm3 |
| Refractive Index | 1.713 |
Quality Assurance and Compliance
Quality is the cornerstone of our operation. We operate under strict Good Manufacturing Practice guidelines to ensure that all peptide building blocks and active ingredients are produced in controlled environments. Our quality assurance team conducts comprehensive testing at multiple stages of production, including raw material verification, in-process controls, and final product release testing.
- Full traceability from raw amino acids to finished peptide.
- Comprehensive documentation including COA, MSDS, and method validation reports.
- Regular audits to maintain compliance with international regulatory frameworks.
- Stability testing to guarantee shelf-life integrity under recommended storage conditions.
Industrial Applications
This high-purity peptide is primarily utilized in the manufacture of pharmaceutical preparations requiring precise hormonal regulation. It serves as a critical component for companies developing treatments related to water balance and coagulation factors. Due to its specific biological activity, it is essential that partners utilize this material within licensed facilities equipped to handle potent pharmaceutical compounds.
Our technical support team provides formulation guidance to help partners integrate this active ingredient into various dosage forms, including tablets and lyophilized powders. We emphasize the importance of adhering to prescribed handling procedures to maintain the structural integrity of the peptide chain during processing.
Packaging and Storage
To preserve the stability of Desmopressin Acetate, we utilize specialized packaging designed to protect against moisture and light degradation. Standard commercial quantities are available in sealed aluminum drums with inner polyethylene liners. Custom packaging solutions can be arranged to meet specific logistical requirements of our global partners.
Storage recommendations include maintaining a cool, ventilated environment away from direct sunlight and heat sources. Proper inventory management ensures that the material remains within specification throughout its designated shelf life. We offer global shipping services with temperature-controlled options where necessary to ensure product integrity upon arrival.



