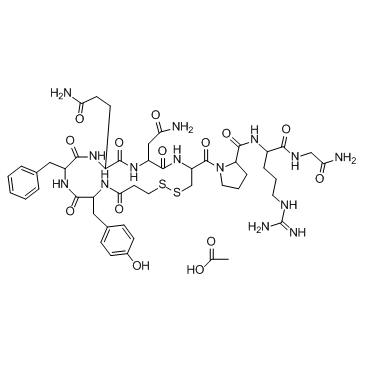
Desmopressin Acetate
- CAS No.62288-83-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Desmopressin Acetate API for pharmaceutical formulation. Reliable bulk supply with comprehensive COA and global logistics support.
Request Bulk PricingProduct Technical Details
Product Overview
Desmopressin Acetate represents a highly specialized synthetic peptide analog utilized extensively within the pharmaceutical industry. As a modified form of the natural hormone vasopressin, this compound is engineered to exhibit enhanced antidiuretic properties while minimizing pressor effects. Our facility provides this critical active pharmaceutical ingredient (API) to licensed manufacturers who require consistent quality and regulatory compliance for downstream drug production. The chemical structure is meticulously stabilized to ensure potency during storage and transportation.
Technical Specifications
| Parameter | Value |
|---|---|
| Chemical Name | Desmopressin Acetate |
| CAS Registry Number | 62288-83-9 |
| Molecular Formula | C48H68N14O14S2 |
| Molecular Weight | 1129.269 g/mol |
| Appearance | White to off-white powder |
| Assay Purity | ≥98.0% |
| Melting Point | 170-172°C |
| Density | 1.56 g/cm3 |
Quality Assurance and Control
Quality integrity is the cornerstone of our manufacturing process. Every batch of Desmopressin Acetate undergoes rigorous analytical testing using high-performance liquid chromatography (HPLC) and mass spectrometry to verify identity and purity levels. We adhere to strict internal quality management systems that align with international Good Manufacturing Practice (GMP) guidelines. Certificates of Analysis (COA) are provided with every shipment, detailing specific test results for moisture content, residual solvents, and microbial limits. This transparency ensures that our partners can maintain their own regulatory compliance without interruption.
Industrial Applications
This peptide compound is primarily designated for use in the formulation of therapeutic agents focused on water balance regulation and hemostasis. It serves as a key component in various pharmaceutical preparations including tablets, nasal sprays, and injectable solutions. Due to its specific biological activity, it is essential for manufacturers developing treatments that require precise dosing and high bioavailability. Our product supports the production of medicines that assist in managing conditions related to fluid retention and clotting factor levels, always within the framework of licensed pharmaceutical manufacturing.
Storage and Handling
To maintain chemical stability and potency, proper storage conditions are essential. The material should be kept in a cool, ventilated environment away from direct sunlight and moisture sources. Containers must remain tightly sealed when not in use to prevent degradation from humidity. We recommend storing the product at controlled room temperature or under refrigeration depending on the specific formulation requirements of the end user. Our packaging options include standard 25 kg drums or customized solutions tailored to specific logistical needs, ensuring the product arrives in optimal condition.



