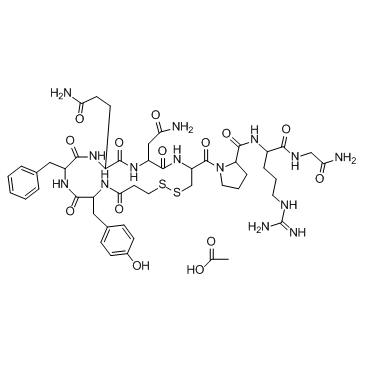
Lixisenatide
- CAS No.827033-10-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Lixisenatide peptide API for pharmaceutical formulation. Ideal for type II diabetes therapeutic development and research.
Request a Quote & Full COAProduct Technical Details
Product Overview
Lixisenatide represents a advanced class of synthetic peptide pharmaceuticals designed for specialized therapeutic applications. As a glucagon-like peptide-1 (GLP-1) receptor agonist, this compound is engineered to meet stringent pharmaceutical standards for efficacy and stability. Our supply chain ensures that every batch undergoes rigorous quality control to maintain consistency for downstream medicinal product formulation.
This peptide ingredient is manufactured using state-of-the-art solid-phase peptide synthesis techniques, ensuring high structural integrity and minimal impurities. It serves as a critical active pharmaceutical ingredient (API) for licensed pharmaceutical companies developing treatments for metabolic disorders. We prioritize compliance with international regulatory frameworks to support our partners in bringing safe and effective therapies to market.
Technical Specifications
Quality assurance is paramount in the production of peptide-based APIs. Our facility employs advanced analytical methods including HPLC and mass spectrometry to verify identity and purity. The physical characteristics are optimized for ease of handling during the drug manufacturing process.
| Parameter | Specification |
|---|---|
| Chemical Name | Lixisenatide |
| CAS Registry Number | 827033-10-3 |
| Appearance | White to off-white powder |
| Purity (Assay) | ≥98.0% |
| Storage Condition | Cool, ventilated place |
| Packing Size | 25 kg/drum or customized |
Industrial Applications
The primary application of this material is within the pharmaceutical sector for the development of antidiabetic medications. It is utilized as a key component in formulations intended to assist in the management of type II diabetes when combined with diet and exercise regimes. Research institutions and pharmaceutical manufacturers rely on this high-grade peptide for clinical trial material production and commercial drug manufacturing.
- Active ingredient for injectable diabetes therapies
- Reference standard for analytical method validation
- Component in combination therapy research
- Raw material for licensed pharmaceutical formulation
Quality Assurance and Storage
To maintain the stability and potency of the peptide, proper storage protocols must be followed. The material should be kept in a cool, well-ventilated environment away from direct sunlight and moisture. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. We adhere to strict packaging guidelines to prevent degradation during transit, ensuring the product arrives in optimal condition for immediate use in production lines.
Packaging and Logistics
Standard packaging consists of 25 kg drums, which are designed to protect the contents from environmental factors. However, we understand that different manufacturing scales require different solutions. Therefore, we offer customized packaging options according to specific customer requirements. Our global logistics network ensures timely delivery while maintaining the cold chain or specific storage conditions required for sensitive peptide materials. Partner with us for reliable supply and technical support.