Nesiritide Acetate
- CAS No.114471-18-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
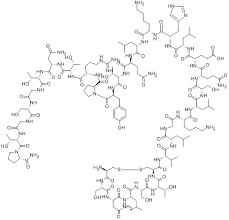
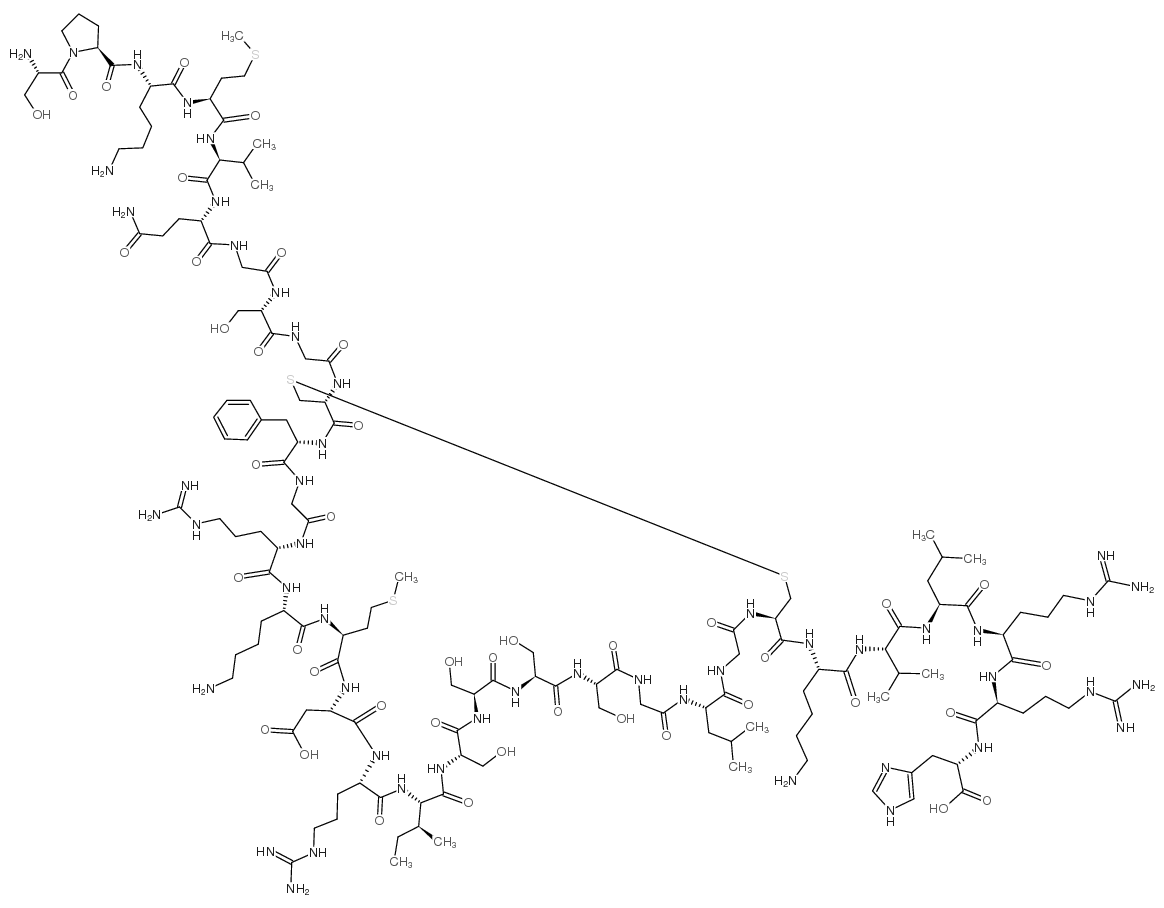
High-purity Nesiritide Acetate (≥98.0%), a recombinant 32-amino acid human B-type natriuretic peptide for advanced cardiovascular research and therapeutic development.
Request a Quote & Full COAProduct Technical Details
Product Overview
Nesiritide Acetate is a high-purity recombinant form of the endogenous 32-amino acid human B-type natriuretic peptide (BNP), originally secreted by ventricular cardiomyocytes in response to volume expansion and pressure overload. This biologically active peptide plays a critical role in modulating cardiovascular fluid homeostasis through targeted molecular pathways.
Mechanism and Biological Relevance
Nesiritide exerts its physiological effects primarily by binding to natriuretic peptide receptor-A (NPR-A), which activates guanylyl cyclase and elevates intracellular cyclic guanosine monophosphate (cGMP) levels. This cascade results in smooth muscle cell relaxation, vasodilation, natriuresis, and suppression of the renin-angiotensin-aldosterone system (RAAS). These mechanisms make it a valuable tool in cardiovascular pharmacology research and the development of novel hemodynamic therapeutics.
Specifications
| Molecular Formula | C₁₄₃H₂₄₄N₅₀O₄₂S₄ |
|---|---|
| Molecular Weight | 3464.04 g/mol |
| Density | 1.529 g/cm³ |
| Index of Refraction | 1.679 |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥98.0% |
| Storage | Store in a cool, ventilated place |
| Packaging | 25 kg/drum; custom packaging available upon request |
Industrial Applications
As a well-characterized bioactive peptide, Nesiritide Acetate is primarily utilized in:
- Preclinical and clinical research on heart failure and acute decompensated cardiac conditions
- Development of cardiovascular drug candidates targeting cGMP-mediated pathways
- Reference standard in analytical method validation and bioassays
- Formulation studies requiring high-integrity, sequence-defined human peptides
Our product is manufactured under stringent quality control protocols compliant with international regulatory standards, ensuring batch-to-batch consistency and suitability for sensitive biomedical applications.