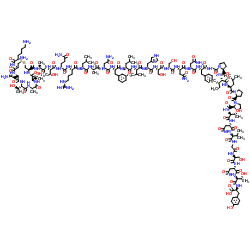
Pramlintide
- CAS No.151126-32-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Pramlintide peptide available for pharmaceutical research and development. Certified manufacturer supplying global markets with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
Pramlintide represents a significant advancement in synthetic peptide technology, serving as a stable analogue of the human hormone amylin. Manufactured under stringent quality control protocols, this product is designed to meet the rigorous demands of pharmaceutical research and development sectors. Our facility utilizes advanced solid-phase peptide synthesis techniques to ensure high structural integrity and biological activity consistency across all production batches.
As a leading global supplier, we understand the critical nature of peptide-based therapeutics in modern medicine. Pramlintide is engineered to mimic the physiological effects of native amylin, which is co-secreted with insulin by pancreatic beta cells following meal intake. This makes it an invaluable tool for extensive studies related to glucose regulation, metabolic disorders, and endocrine function. Our commitment to excellence ensures that every unit supplied adheres to international standards for purity, safety, and chemical identity.
Technical Specifications
| Parameter | Value |
|---|---|
| Product Name | Pramlintide |
| CAS Number | 151126-32-8 |
| Molecular Formula | C171H265N50O54S2 |
| Molecular Weight | 3949.43 |
| Appearance | White to off-white powder |
| Assay | ≥98.0% |
Quality Assurance and Manufacturing
Quality is the cornerstone of our manufacturing process. Each batch of Pramlintide undergoes comprehensive analytical testing using high-performance liquid chromatography (HPLC) and mass spectrometry to verify composition. We provide a detailed Certificate of Analysis (COA) with every shipment, verifying identity, purity, and potency without exception. Our production environment maintains strict temperature and humidity controls to prevent any degradation of the delicate peptide structure during synthesis and packaging.
- Strict compliance with Good Manufacturing Practice (GMP) guidelines.
- Advanced purification processes to remove truncation sequences and impurities.
- Stability testing under various storage conditions to ensure shelf life.
- Full traceability from raw amino acids to finished peptide product.
- Dedicated quality control team for batch release approval.
Industrial Applications
This peptide is primarily utilized in pharmaceutical research focused on diabetes management and metabolic health initiatives. It serves as a critical reference standard and active ingredient for formulation development in controlled laboratory settings. Researchers rely on our high-purity material to validate experimental models and develop new therapeutic strategies for metabolic syndromes. The stability of our Pramlintide ensures reliable and reproducible results in long-term scientific studies.
Packaging Storage and Logistics
To maintain optimal stability, Pramlintide is packaged in sealed containers protected from light and moisture ingress. Standard packing options include 25 kg drums, with customization available to suit specific logistical requirements for international shipping. We recommend storing the product in a cool, ventilated area away from direct sunlight and heat sources. Proper handling procedures should be followed to ensure safety and maintain product integrity during transport. Our logistics team ensures timely delivery with cold chain options available upon request for sensitive shipments.



