Teriparatide Acetate
- CAS No.99294-94-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Teriparatide Acetate (CAS 99294-94-7) supplied for pharmaceutical research and manufacturing. Meets strict quality standards for peptide-based therapeutics.
Request Bulk PricingProduct Technical Details
Product Overview
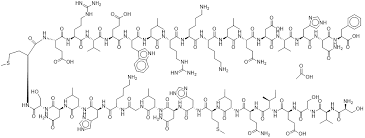
Teriparatide Acetate is a sophisticated synthetic polypeptide corresponding to the 1-34 N-terminal fragment of human parathyroid hormone (PTH). As a critical pharmaceutical ingredient, this compound plays a pivotal role in the development of advanced therapeutics focused on bone metabolism and skeletal health. Our facility specializes in the production of high-grade peptide sequences, ensuring that every batch of Teriparatide Acetate meets rigorous international standards for purity and biological activity.
This acetate salt form enhances the stability and solubility of the peptide, making it an ideal candidate for various pharmaceutical formulations. We understand the complexities involved in peptide synthesis and purification, which is why our manufacturing processes are designed to minimize impurities and maximize yield without compromising on quality.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Teriparatide Acetate |
| CAS Number | 99294-94-7 |
| Molecular Formula | C181H291N55O51S2 |
| Molecular Weight | 4117.70 g/mol |
| Appearance | White to off-white powder |
| Purity (Assay) | ≥98.0% |
| Storage Conditions | Cool, ventilated place, protected from light |
Industrial Applications
Primarily utilized within the pharmaceutical sector, Teriparatide Acetate serves as a key active pharmaceutical ingredient (API) for licensed manufacturers developing treatments for osteoporosis and related bone density disorders. The 1-34 amino acid sequence retains the biological activity of the full-length hormone, specifically targeting bone formation pathways when administered intermittently.
Beyond direct therapeutic formulation, this peptide is extensively used in preclinical research and development. Laboratories require high-purity standards to ensure accurate data during toxicology studies and efficacy trials. Our product supports these critical stages of drug development, providing researchers with reliable material that behaves consistently across experiments.
Quality Assurance and Manufacturing
Quality is the cornerstone of our peptide production. Each batch of Teriparatide Acetate undergoes comprehensive analytical testing, including High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS), to verify identity and purity. We maintain strict control over the synthesis route to prevent the formation of deletion sequences or modified byproducts.
- Comprehensive Certificate of Analysis (COA) provided with every shipment.
- Manufactured in compliance with relevant quality management systems.
- Scalable production capabilities to meet bulk demand.
- Technical support available for formulation guidance.
Storage and Handling
To maintain the integrity of the peptide structure, proper storage is essential. Teriparatide Acetate should be stored in a cool, ventilated environment away from direct sunlight and moisture. Containers must remain tightly sealed when not in use to prevent hydration or degradation. For long-term storage, refrigeration is recommended to preserve biological activity. Handling should be conducted in a controlled environment to minimize exposure to contaminants.
Global Supply Partnership
We are committed to being a reliable partner for global pharmaceutical companies and research institutions. Our logistics network ensures timely delivery while maintaining the cold chain requirements necessary for sensitive peptide products. Whether you require sample quantities for initial testing or large-scale bulk supplies for commercial production, our team is equipped to support your project timelines with competitive pricing and transparent communication.



