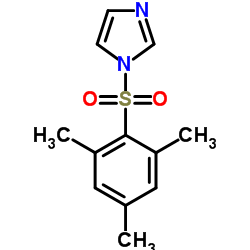
1-(2,4,6-trimethylphenyl)sulfonylimidazole
- CAS No.50257-39-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity condensation reagent for advanced organic synthesis. Reliable supply with comprehensive quality documentation.
Request a Quote & Full COAProduct Technical Details
Product Overview
1-(2,4,6-trimethylphenyl)sulfonylimidazole is a specialized organic compound widely recognized for its efficacy as a condensation reagent in complex chemical synthesis. This sulfonyl imidazole derivative features a unique structural configuration that enhances its reactivity, making it an essential tool for researchers and industrial chemists focused on forming amide or ester bonds. Our manufacturing process ensures exceptional batch-to-batch consistency, adhering to strict international quality standards.
As a key component in modern organic chemistry, this reagent facilitates efficient coupling reactions while minimizing side products. The steric hindrance provided by the trimethylphenyl group contributes to its selective reactivity profile, allowing for precise control during synthesis protocols. We supply this material to pharmaceutical, agrochemical, and fine chemical sectors globally.
Technical Specifications
Quality assurance is paramount in our production facility. Each batch undergoes rigorous analytical testing to confirm identity and purity levels. The following table outlines the critical physical and chemical parameters associated with this product.
| Parameter | Specification |
|---|---|
| CAS Number | 50257-39-1 |
| Molecular Formula | C12H14N2O2S |
| Molecular Weight | 250.32 g/mol |
| Appearance | Off-white to white crystalline powder |
| Purity (HPLC) | ≥98.0% |
| Melting Point | 95-99°C |
| Density | 1.23 g/cm3 |
| Refractive Index | 1.598 |
Applications and Benefits
This sulfonylimidazole derivative is primarily utilized as a condensation agent in the synthesis of heterocyclic compounds and peptide mimetics. Its high reactivity allows for milder reaction conditions compared to traditional coupling agents, reducing energy consumption and improving overall yield. The compound is particularly valuable in scenarios requiring high purity intermediates for downstream processing.
- Enhanced reaction efficiency due to optimized steric properties.
- Compatible with a wide range of organic solvents.
- Minimizes racemization during sensitive coupling steps.
- Supported by comprehensive technical data and safety documentation.
Storage and Handling
To maintain stability and performance, 1-(2,4,6-trimethylphenyl)sulfonylimidazole should be stored in a cool, ventilated environment away from direct sunlight and moisture. The product is classified under Hazard Class 8 with UN Number 1759, requiring appropriate handling procedures during transport. We recommend using personal protective equipment when handling this material in laboratory or industrial settings. Packaging options include 25 kg drums, with custom configurations available upon request to suit specific logistical needs.