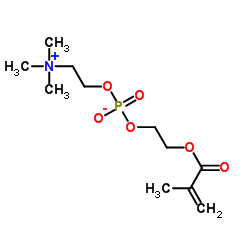
2-Methacryloyloxyethyl Phosphorylcholine
- CAS No.67881-98-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity MPC monomer designed for enhancing biocompatibility in medical devices and polymer coatings.
Request Bulk PricingProduct Technical Details
Product Overview
2-Methacryloyloxyethyl Phosphorylcholine, commonly known as MPC, represents a cutting-edge functional monomer utilized extensively in the development of advanced biomedical materials. With the CAS registry number 67881-98-5, this compound mimics the outer structure of cell membranes, offering exceptional biocompatibility properties. Our manufacturing process ensures high purity levels suitable for sensitive medical and pharmaceutical applications. The chemical structure incorporates a phosphorylcholine group which is pivotal for reducing protein adsorption and preventing thrombus formation on material surfaces.
Key Mechanisms and Benefits
The unique efficacy of this specialty chemical lies in its ability to form a hydration layer on polymer surfaces. This water-rich interface acts as a barrier against biological fouling, making it ideal for implants and diagnostic devices. By integrating this monomer into polymer chains, manufacturers can significantly improve blood compatibility and tissue compatibility without compromising the mechanical strength of the base material. The result is a surface that is recognized by the body as friendly, reducing immune responses and inflammation risks.
Industrial Applications
This product serves as a critical component in various high-performance sectors. Primary applications include the coating of cardiovascular devices such as stents and catheters to prevent clotting. It is also widely used in the production of contact lenses to enhance comfort and moisture retention. Furthermore, the chemical is essential in drug delivery systems where controlled release and minimal interaction with blood components are required. Researchers also utilize this monomer for creating biosensors that require stable performance in biological fluids.
Quality Specifications
We adhere to strict quality control protocols to ensure every batch meets international standards for biomedical-grade chemicals. The following table outlines the key analytical parameters verified during our production process.
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | White crystal | Conform |
| Assay (HPLC) | ≥97.0% | 97.22% |
| Heavy metals | <20 ppm | Pass |
| Arsenic | <2 ppm | Pass |
| Conclusion | Conforms to Factory Standard | |
Why Choose Our Supply
As a global manufacturer, we provide consistent supply chains and comprehensive technical support. Our facility is equipped to handle bulk orders while maintaining the integrity of this sensitive monomer. We offer full documentation including Certificates of Analysis (COA) and safety data sheets. Partnering with us ensures access to premium materials that meet the rigorous demands of modern medical device fabrication and specialty polymer synthesis.
- Enhanced surface biocompatibility for medical implants
- Superior resistance to protein adsorption and cell adhesion
- Validated high purity suitable for regulatory submissions
- Stable supply chain for large-scale production needs
- Technical support for polymer formulation optimization