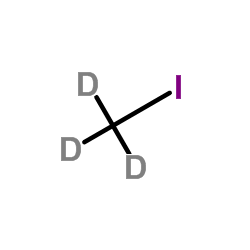
Iodomethane-d3
- CAS No.865-50-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity deuterated methylating agent for research and analytical applications.
Request a Quote & Full COAProduct Technical Details
Product Overview
Iodomethane-d3 represents a critical stable isotope-labeled reagent designed for advanced pharmaceutical research and analytical chemistry. As a deuterated analog of methyl iodide, this compound serves as an essential tool for introducing isotopic labels into complex organic molecules. Our manufacturing process ensures exceptional isotopic purity, making it ideal for mass spectrometry internal standards and nuclear magnetic resonance spectroscopy applications.
The production of this deuterated reagent adheres to strict quality control protocols. Each batch undergoes rigorous testing to verify deuteration levels and chemical purity. This commitment to quality ensures reliable performance in sensitive analytical procedures and synthetic pathways where isotopic integrity is paramount.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 865-50-9 |
| Molecular Formula | CD3I |
| Molecular Weight | 144.958 |
| Appearance | Colorless clear liquid |
| Purity | ≥99.5% |
| Deuteration | ≥99.8 atom%D |
| Density | 2.2±0.1 g/cm3 |
| Boiling Point | 40.3±3.0 °C at 760 mmHg |
| Melting Point | -66.5ºC |
Industrial Applications
This specialized chemical finds extensive use in drug metabolism and pharmacokinetics studies. Researchers utilize Iodomethane-d3 to trace metabolic pathways without altering the chemical behavior of the parent compound. The minimal kinetic isotope effect ensures that biological systems process the labeled molecule similarly to its non-labeled counterpart.
In addition to biological research, this reagent is vital for quantitative analysis using liquid chromatography-mass spectrometry. It serves as an internal standard to correct for variations in sample preparation and instrument response. This application enhances the accuracy and precision of quantitative measurements in regulatory compliance testing.
- Pharmaceutical process development and optimization
- Environmental pollutant tracking and analysis
- Mechanistic studies in organic synthesis
- Calibration standards for analytical instrumentation
Quality And Safety
We maintain comprehensive documentation for every shipment, including certificates of analysis and safety data sheets. Our facility implements advanced containment systems to handle volatile compounds safely. Customers receive products in secure packaging designed to prevent leakage and maintain stability during transit.
Handling this material requires appropriate safety precautions due to its volatility and toxicity. Users should operate within certified fume hoods and wear suitable personal protective equipment. Proper storage conditions include keeping the container tightly closed in a cool, well-ventilated area away from incompatible substances.