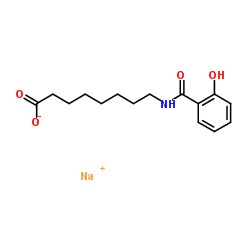
Salcaprozate Sodium
- CAS No.203787-91-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Salcaprozate sodium serving as a leading oral absorption enhancer for peptide and protein-based pharmaceutical formulations.
Request Bulk PricingProduct Technical Details
Product Overview
Salcaprozate sodium, widely recognized by its acronym SNAC, represents a breakthrough class of oral absorption enhancers designed to facilitate the delivery of macromolecular therapeutics. As a specialized chemical entity, it addresses the critical challenge of low bioavailability associated with peptide and protein-based drugs when administered via the oral route. Our manufacturing process ensures exceptional purity levels, making it an ideal candidate for advanced pharmaceutical formulations requiring precise delivery mechanisms.
This compound functions by temporarily modifying the local gastric environment, thereby protecting sensitive active pharmaceutical ingredients from enzymatic degradation and promoting transcellular absorption. The result is a significant improvement in therapeutic efficacy without compromising safety profiles. We supply this material under strict quality control standards suitable for global regulatory submissions.
Mechanism of Action
The primary function of Salcaprozate sodium lies in its ability to elevate local pH levels within the stomach, creating a protective microenvironment for acid-labile drugs. Furthermore, it interacts with cell membranes to increase permeability, allowing large molecules such as insulin and heparin to pass through the gastrointestinal barrier efficiently. This dual mechanism ensures that the active drug reaches systemic circulation in its intact form.
- Protects peptides from gastric acid degradation
- Enhances transcellular permeability across the intestinal wall
- Minimizes enzymatic breakdown during digestion
- Supports consistent pharmacokinetic profiles
Technical Specifications
Our production facility adheres to rigorous analytical standards to guarantee consistency across every batch. The following table outlines the key quality parameters verified through advanced chromatographic and spectroscopic methods.
| Parameter | Specification |
|---|---|
| Appearance | White solid |
| Identification | IR Conformity |
| Purity | ≥99.0% |
| Moisture Content | ≤2.5% |
| Single Impurity | ≤0.1% |
| Total Impurities | ≤1.0% |
| Assay | 98-102% |
Industrial Applications
While initially developed for oral insulin delivery, the utility of Salcaprozate sodium extends to various other therapeutic areas requiring enhanced oral bioavailability. It is particularly valuable in the development of nutraceuticals and specialty pharmaceuticals where patient compliance is improved by avoiding injectable routes.
Researchers utilize this material as a benchmark for formulation studies involving heparin, calcitonin, and other peptide hormones. Its solubility profile allows for versatile integration into solid dosage forms such as tablets and capsules. We recommend consulting our technical team for specific formulation guidance tailored to your project requirements.
Quality Assurance and Storage
Regulatory compliance is a cornerstone of our supply chain. We understand the stringent requirements for excipients used in clinical trials and commercial production. Our documentation supports Investigational New Drug (IND) applications, providing transparency on synthesis routes and impurity profiles. This level of detail accelerates the development timeline for our partners.
Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) verifying compliance with factory standards. Residual solvent testing confirms safety limits for ethanol, acetone, and acetonitrile. To maintain stability, the product should be stored in a cool, dry place away from direct sunlight and moisture. Proper handling ensures the longevity of its physicochemical properties during extended storage periods.
Packaging options are available to suit both laboratory-scale research and large-scale commercial manufacturing. We utilize moisture-barrier containers to prevent hygroscopic uptake during transit. Global logistics partners ensure timely delivery while maintaining cold chain or ambient conditions as specified. Our commitment to reliability makes us a preferred vendor for leading pharmaceutical companies.