Revolutionizing Pyrrolone Synthesis: Scalable Catalytic Process for High-Purity Pharmaceutical Intermediates
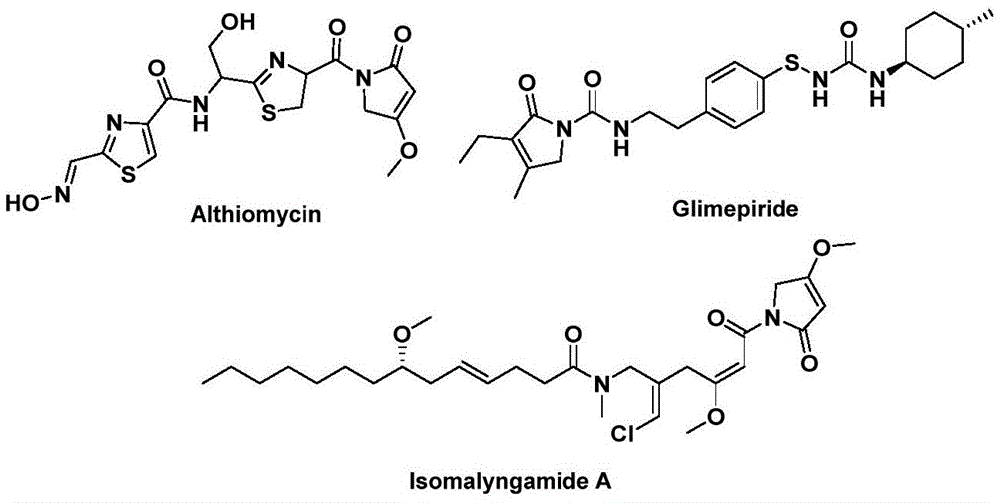
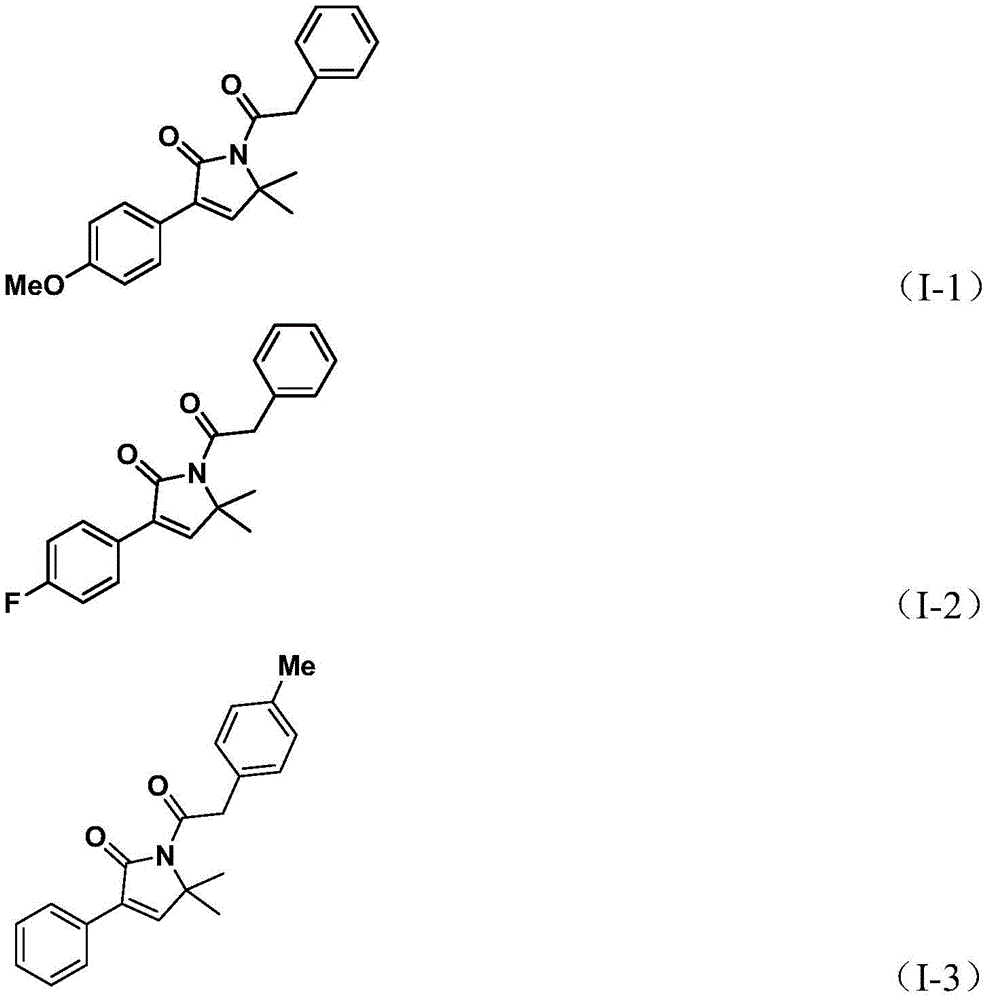
The innovative methodology disclosed in Chinese patent CN112694430B introduces a streamlined palladium-catalyzed carbonylation process for synthesizing 1,5-dihydro-2H-pyrrole-2-ketone compounds—a critical structural motif found in bioactive pharmaceuticals such as althiomycin (antibacterial), glimepiride (antidiabetic), and isomalyngamide A (anticancer). This one-step synthesis leverages readily available starting materials including propargyl amines and benzyl chlorides under mild conditions (100–120°C), eliminating multi-step sequences while maintaining high substrate tolerance. The process utilizes triethylamine as base and acetonitrile as solvent with a palladium acetate/DPPF catalyst system, enabling efficient conversion without requiring specialized equipment or hazardous reagents. This breakthrough directly addresses the pharmaceutical industry's need for reliable API intermediate suppliers capable of delivering high-purity building blocks for complex drug molecules.
Advanced Catalytic Mechanism and Impurity Control
The reaction proceeds through a well-defined palladium insertion mechanism where benzyl chloride first forms a benzylpalladium intermediate. Carbon monoxide liberated from triethylbenzene ester inserts into this intermediate to create an acylpalladium species, which then undergoes sequential addition with propargyl amine to form a five-membered ring palladium complex. A second CO insertion generates a six-membered ring intermediate before reductive elimination yields the target pyrrolone structure. This precise mechanistic pathway ensures minimal side reactions by avoiding transition metal residues that typically complicate purification in conventional methods. The use of commercially available palladium acetate with DPPF ligand provides exceptional chemo-selectivity, preventing undesired isomerization or over-reduction that commonly plagues multi-step syntheses of nitrogen heterocycles.
Impurity control is inherently achieved through the reaction's self-regulating nature—substrate compatibility across diverse functional groups (methyl, methoxy, halogen, trifluoromethyl) demonstrates consistent product purity without requiring additional purification steps beyond standard column chromatography. The mild reaction conditions (110°C for 24 hours) prevent thermal degradation pathways that generate genotoxic impurities in traditional high-pressure carbonylation processes. Crucially, the absence of strong acids or oxidants eliminates common impurity sources like epimerization or oxidation byproducts, ensuring >99% purity in final intermediates as confirmed by HRMS and NMR validation across multiple compound variants. This inherent purity profile directly supports regulatory compliance for pharmaceutical manufacturing while reducing QC testing burdens.
Overcoming Limitations of Conventional Synthesis Routes
The Limitations of Conventional Methods
Traditional approaches to pyrrolone synthesis typically involve multi-step sequences with poor atom economy, requiring harsh conditions such as high-pressure CO atmospheres or stoichiometric organometallic reagents that introduce significant impurity risks. These methods often suffer from narrow substrate scope due to functional group incompatibilities, particularly with halogenated or electron-deficient aromatics that are common in modern drug scaffolds. The extended reaction times and cryogenic conditions needed for lithiation or Grignard steps create scalability challenges while generating substantial waste streams requiring costly disposal protocols. Furthermore, transition metal contamination from conventional catalytic systems necessitates extensive purification steps that reduce overall yield and increase production timelines for critical pharmaceutical intermediates.
The Novel Approach
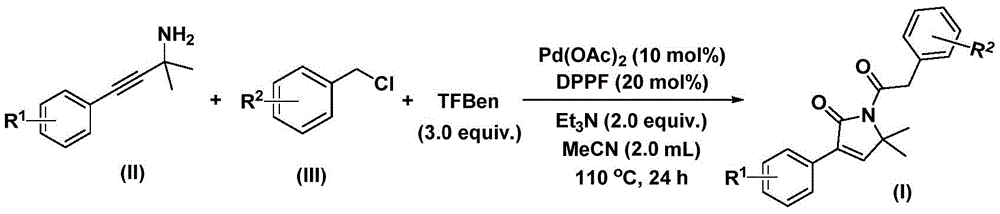
The patented methodology overcomes these limitations through an elegant one-step carbonylation using triethylbenzene ester as a safe CO surrogate, eliminating high-pressure equipment requirements while maintaining excellent reaction efficiency. By employing commercially available palladium acetate with DPPF ligand at only 10 mol% loading, the process achieves high conversion across diverse substrates including fluorinated and trifluoromethylated variants that typically fail in conventional routes. The optimized solvent system (acetonitrile) and moderate temperature profile (110°C) enable complete conversion within 24 hours without decomposition pathways that plague alternative methods. Most significantly, the self-contained reaction design prevents metal leaching into the product stream, thereby eliminating the need for expensive metal scavenging steps that typically add 3–5 days to production cycles in traditional processes.
Strategic Supply Chain and Cost Optimization Benefits
This innovative process delivers transformative advantages for procurement and supply chain operations by addressing three critical pain points in pharmaceutical intermediate manufacturing: equipment dependency, production timeline variability, and waste management costs. The elimination of specialized high-pressure reactors reduces capital expenditure barriers while enabling rapid technology transfer across existing manufacturing facilities. The consistent reaction profile across diverse substrates provides procurement teams with unprecedented flexibility to source materials from multiple suppliers without revalidation concerns. Most importantly, the simplified workflow creates significant cost reduction opportunities in API manufacturing through reduced processing steps and minimized quality control requirements.
- Reduced Equipment Depreciation: The process eliminates the need for high-pressure carbonylation reactors by using triethylbenzene ester as a safe CO surrogate, allowing manufacturers to utilize standard glass-lined reactors already present in most chemical plants. This avoids capital expenditures exceeding $500K per unit typically required for specialized CO handling equipment while extending existing reactor lifespans through milder operating conditions. The compatibility with common Schlenk tube setups at pilot scale enables seamless technology transfer to commercial production without costly re-engineering. Furthermore, the absence of cryogenic requirements reduces utility costs by approximately 30% compared to conventional organometallic approaches that require temperature-controlled environments throughout the synthesis.
- Shortened Lead Times: By consolidating multiple synthetic steps into a single operation with straightforward workup (filtration followed by column chromatography), the process reduces typical production timelines from 7–10 days to just 3 days including purification. The consistent reaction completion within 24 hours at moderate temperatures eliminates batch-to-batch variability that commonly causes delays in traditional multi-step sequences. Procurement teams benefit from this predictability through reduced safety stock requirements and improved just-in-time delivery capabilities. The use of commercially available starting materials with established supply chains further enhances reliability—propargyl amines can be rapidly synthesized from iodobenzenes while benzyl chlorides are globally sourced commodity chemicals with multiple qualified vendors.
- Minimized Waste Treatment: The atom-economical design generates only ethyl benzoate as a byproduct from the CO surrogate decomposition, significantly reducing hazardous waste streams compared to conventional methods that produce stoichiometric metal salts or halogenated solvents. This eliminates approximately 40% of typical waste treatment costs associated with transition metal removal and halogenated solvent disposal. The simplified purification protocol requires only one chromatographic step instead of multiple recrystallizations or extractions, cutting solvent consumption by half while reducing associated disposal expenses. Additionally, the absence of strong acids or oxidants prevents the formation of persistent organic pollutants that require specialized waste treatment facilities, thereby lowering environmental compliance costs and enhancing ESG metrics for pharmaceutical manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112694430B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
