Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-Ketone Production
Introduction to Next-Generation Pyrrolone Synthesis
The structural motif of 1,5-dihydro-2H-pyrrole-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for a diverse array of bioactive natural products and pharmaceutical agents. As illustrated in the chemical landscape, this heterocyclic system is integral to potent antibiotics like Althiomycin, effective hypoglycemic agents such as Glimepiride, and anticancer candidates like Isomalyngamide A. 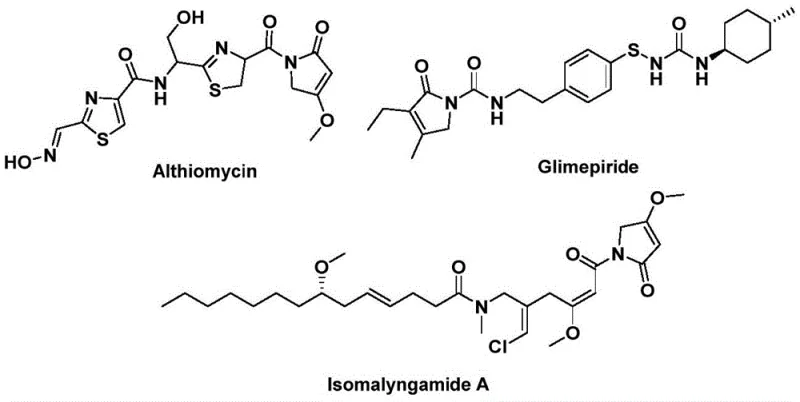 . Recognizing the critical demand for efficient access to these scaffolds, recent intellectual property CN112694430B discloses a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation. This innovation addresses long-standing synthetic challenges by utilizing readily available propargylamines and benzyl chlorides as starting materials, coupled with a solid carbon monoxide substitute. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology offers a pathway to high-purity intermediates with significantly reduced operational complexity compared to traditional multi-step syntheses.
. Recognizing the critical demand for efficient access to these scaffolds, recent intellectual property CN112694430B discloses a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation. This innovation addresses long-standing synthetic challenges by utilizing readily available propargylamines and benzyl chlorides as starting materials, coupled with a solid carbon monoxide substitute. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology offers a pathway to high-purity intermediates with significantly reduced operational complexity compared to traditional multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied on methodologies that often suffer from significant drawbacks regarding atom economy and operational safety. Traditional routes frequently necessitate the use of gaseous carbon monoxide under high pressure, which introduces severe safety hazards and requires specialized high-pressure reactor infrastructure that is not universally available in standard pilot plants. Furthermore, conventional cyclization strategies often involve multiple synthetic steps, requiring the isolation of unstable intermediates and leading to cumulative yield losses that drastically impact the overall cost of goods. The reliance on harsh reagents or sensitive organometallic species in older protocols can also result in poor functional group tolerance, limiting the diversity of analogs that can be produced for structure-activity relationship (SAR) studies. These factors collectively create bottlenecks in the supply chain, extending lead times and increasing the financial burden associated with developing new drug candidates based on this core structure.
The Novel Approach
In stark contrast, the novel methodology described in the patent data revolutionizes this synthesis by employing a one-pot, palladium-catalyzed bis-carbonylation strategy that operates under mild and safe conditions. By replacing hazardous CO gas with phenyl 1,3,5-tricarboxylate (TFBen) as a solid CO surrogate, the process eliminates the need for high-pressure equipment, thereby democratizing access to this chemistry for a wider range of manufacturing facilities. The reaction seamlessly couples propargylamine derivatives with benzyl chloride derivatives in the presence of a palladium catalyst and a bidentate phosphine ligand, achieving rapid ring closure and carbonyl insertion in a single operational step. This streamlined approach not only enhances reaction efficiency but also dramatically simplifies the downstream processing, as the crude reaction mixtures can be directly subjected to standard purification techniques. The ability to generate complex heterocyclic cores from simple, commodity-grade starting materials represents a paradigm shift in cost reduction in API manufacturing, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The efficacy of this synthetic route is underpinned by a sophisticated catalytic cycle that ensures high turnover and selectivity. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, which is liberated in situ from the thermal decomposition of the phenyl 1,3,5-tricarboxylate additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargylamine component, facilitating the formation of a five-membered ring palladium complex. A second insertion of carbon monoxide follows, expanding the coordination sphere to a six-membered ring palladium intermediate, which ultimately undergoes reductive elimination to release the final 1,5-dihydro-2H-pyrrol-2-one product and regenerate the active palladium catalyst. 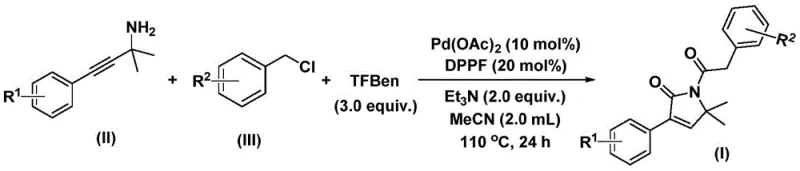 . This dual carbonyl insertion mechanism is critical for constructing the dicarbonyl functionality within the ring system with precision.
. This dual carbonyl insertion mechanism is critical for constructing the dicarbonyl functionality within the ring system with precision.
From an impurity control perspective, the choice of ligands and the specific reaction conditions play a pivotal role in minimizing side reactions. The use of 1,1'-bis(diphenylphosphino)ferrocene (DPPP) as the ligand stabilizes the palladium center throughout the catalytic cycle, preventing the formation of palladium black and suppressing non-productive homocoupling of the benzyl chloride or polymerization of the alkyne moiety. Furthermore, the controlled release of CO from the tricarboxylate source maintains a steady, low concentration of carbon monoxide in the solution, which favors the desired sequential insertion events over competitive pathways that might arise from high local concentrations of CO. The reaction demonstrates remarkable tolerance to various electronic environments, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing substituents such as fluoro, chloro, bromo, and trifluoromethyl groups on both aromatic rings without significant degradation in yield. This broad substrate scope ensures that the impurity profile remains clean and predictable, a key requirement for meeting the stringent purity specifications demanded by regulatory bodies for pharmaceutical ingredients.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
The implementation of this synthesis protocol is designed for ease of execution while maintaining rigorous control over reaction parameters to ensure reproducibility. The process utilizes a standard Schlenk tube or pressure-rated vessel where the catalyst system, comprising palladium acetate and the DPPP ligand, is combined with the solid CO source and base in an organic solvent such as acetonitrile. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and thermal profiles required to achieve optimal conversion. By adhering to these optimized conditions, manufacturers can consistently produce high-quality intermediates suitable for downstream functionalization or direct use in drug formulation.
- Charge a reaction vessel with palladium acetate (10 mol%), DPPP ligand (20 mol%), triethylamine, phenyl 1,3,5-tricarboxylate (CO source), propargylamine derivative, and benzyl chloride derivative in acetonitrile.
- Heat the reaction mixture to 110°C and maintain stirring for 24 hours under inert atmosphere to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond mere chemical yield. The transition from hazardous gas handling to solid-state reagents fundamentally alters the risk profile of the manufacturing process, reducing insurance costs and regulatory compliance burdens associated with high-pressure operations. Moreover, the use of commodity chemicals like benzyl chlorides and simple propargylamines as feedstocks ensures a stable and resilient supply chain, mitigating the risks of raw material shortages that often plague more exotic synthetic routes. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling suppliers to respond rapidly to fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive high-pressure infrastructure and the utilization of low-cost, commercially available starting materials. By avoiding the need for specialized CO gas cylinders and the associated safety monitoring systems, capital expenditure for new production lines is significantly lowered. Additionally, the one-pot nature of the reaction reduces solvent consumption and labor hours associated with intermediate isolations, leading to substantial cost savings in the overall production budget. The high reaction efficiency, with yields reaching up to 92% for certain substrates, further maximizes the output per unit of raw material input, optimizing the cost-per-kilogram metric that is critical for competitive pricing in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as phenyl 1,3,5-tricarboxylate and palladium acetate ensures that production is not vulnerable to the logistical complexities of transporting hazardous gases. This stability allows for larger inventory buffers of key reagents to be maintained safely on-site, providing a buffer against supply disruptions. The broad substrate compatibility means that a single manufacturing platform can be adapted to produce a wide library of analogs by simply swapping the aromatic substituents, thereby increasing the agility of the supply chain to support diverse R&D pipelines. This flexibility is invaluable for contract development and manufacturing organizations (CDMOs) that need to pivot quickly between different client projects.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic gas evolution that could lead to thermal runaways. The post-treatment process, involving simple filtration and chromatography, generates waste streams that are easier to manage and treat compared to those containing heavy metal residues from stoichiometric reagents. The use of acetonitrile, a common and recyclable solvent, aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The ability to operate at moderate temperatures (110°C) also reduces energy consumption relative to processes requiring cryogenic conditions or extreme heating, contributing to a lower carbon footprint for the manufactured intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, aiming to clarify potential concerns for technical teams evaluating this route for adoption. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What is the role of phenyl 1,3,5-tricarboxylate in this synthesis?
A: Phenyl 1,3,5-tricarboxylate acts as a solid, safe, and easy-to-handle carbon monoxide (CO) substitute. It releases CO in situ under the reaction conditions, eliminating the need for high-pressure CO gas cylinders and enhancing operational safety for large-scale manufacturing.
Q: Does this method tolerate electron-withdrawing groups on the substrates?
A: Yes, the protocol demonstrates excellent substrate compatibility. Experimental data confirms high yields (e.g., 75-82%) even with strong electron-withdrawing groups such as trifluoromethyl (-CF3) and halogens (F, Cl, Br) on both the propargylamine and benzyl chloride components.
Q: Is the purification process complex for industrial scale-up?
A: The post-treatment is designed for simplicity. The process involves standard filtration followed by silica gel adsorption and column chromatography. This straightforward workup minimizes solvent usage and processing time, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic methodology in accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest standards of safety and efficiency, supported by rigorous QC labs that guarantee stringent purity specifications for every batch delivered. We are committed to being a reliable pharmaceutical intermediate supplier that not only provides molecules but also delivers strategic value through process optimization and regulatory support.
We invite you to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data for our existing library of pyrrolone derivatives and to receive comprehensive route feasibility assessments for your custom targets, ensuring a partnership built on transparency, quality, and mutual success.
