Advanced Catalytic Routes for High-Purity O-Alkynyl Phenylcyclobutanone and Naphthalenone Production
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways for complex molecular scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN112920033A introduces a significant advancement in this domain by detailing a highly efficient preparation method for o-alkynyl phenylcyclobutanone and its subsequent conversion into naphthalenone derivatives. This technology addresses long-standing challenges in constructing fused ring systems containing cyclobutanone motifs, which are increasingly valued in medicinal chemistry for their unique strain energy and reactivity profiles. By leveraging a optimized palladium-catalyzed cross-coupling followed by a Lewis acid-mediated cyclization, this process offers a streamlined route to high-value intermediates that were previously difficult to access with high purity and yield.
For research and development teams focusing on novel drug candidates, the ability to rapidly synthesize these structures with minimal impurity profiles is paramount. The disclosed method not only simplifies the operational complexity but also enhances the overall atom economy of the synthesis. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented methodologies allows us to offer superior custom synthesis services that align with the rigorous quality standards demanded by global regulatory bodies. The following analysis delves into the technical specifics and commercial implications of this innovative synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-substituted cyclobutanones bearing ortho-alkynyl groups has been fraught with difficulties using traditional transition metal catalysis. Standard Sonogashira coupling conditions, which typically employ palladium-copper co-catalysis, often fail to deliver satisfactory results when applied to sterically hindered or electronically deactivated o-bromophenyl cyclobutanone substrates. Literature precedents indicate that attempting to introduce an alkynyl group at the bromine position on the carbon ring frequently leads to low conversion rates, extensive formation of homocoupling byproducts, and difficult-to-remove palladium black residues. Furthermore, conventional methods often struggle with the subsequent ring-opening and cyclization steps required to generate the naphthalenone core, necessitating harsh reaction conditions that can degrade sensitive functional groups. These inefficiencies result in prolonged development timelines and increased costs associated with extensive purification protocols, making scale-up for commercial production economically unviable for many organizations.
The Novel Approach
The methodology described in the patent overcomes these hurdles through a meticulously optimized catalytic system that decouples the coupling and cyclization events for maximum control. Instead of relying on generic conditions, the inventors identified that using specific palladium sources like Pd(TFA)2 in conjunction with bulky phosphine ligands such as DPEphos dramatically improves the coupling efficiency. This novel approach enables the reaction to proceed smoothly at moderate temperatures around 90 °C in 1,4-dioxane, avoiding the decomposition issues seen in earlier attempts. 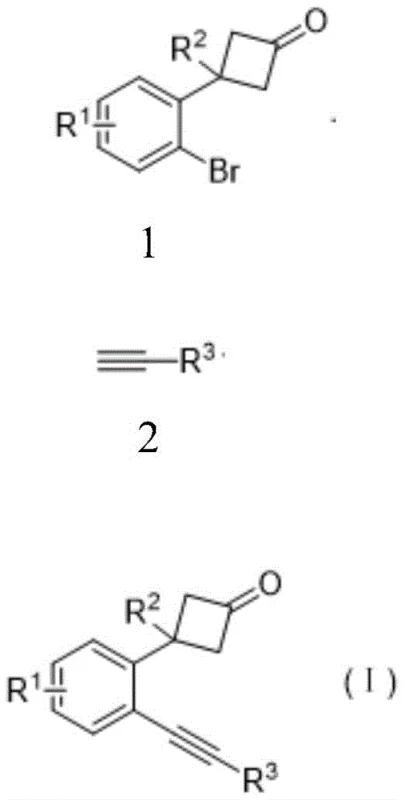 Furthermore, the subsequent transformation into naphthalenone utilizes a distinct copper-based Lewis acid system that facilitates a conjugated ketene intermediate formation, leading to a clean intramolecular cycloaddition. This two-stage strategy ensures that each step is optimized independently, resulting in significantly higher overall yields and a much cleaner crude product profile that simplifies downstream processing.
Furthermore, the subsequent transformation into naphthalenone utilizes a distinct copper-based Lewis acid system that facilitates a conjugated ketene intermediate formation, leading to a clean intramolecular cycloaddition. This two-stage strategy ensures that each step is optimized independently, resulting in significantly higher overall yields and a much cleaner crude product profile that simplifies downstream processing.
Mechanistic Insights into Pd-Catalyzed Coupling and Cu-Mediated Cyclization
The success of this synthetic route lies in the precise mechanistic orchestration of the catalytic cycles involved. In the first stage, the palladium catalyst undergoes oxidative addition with the o-bromophenyl cyclobutanone, forming a reactive organopalladium species. The presence of the DPEphos ligand is crucial here, as its wide bite angle stabilizes the palladium center and facilitates the transmetallation step with the terminal alkyne. Unlike traditional copper-free or standard copper-co-catalyzed systems which might lead to Glaser homocoupling of the alkyne, this specific ligand environment directs the reaction exclusively towards the desired cross-coupled product. The base, preferably potassium carbonate, serves to deprotonate the terminal alkyne without promoting unwanted side reactions on the strained cyclobutanone ring. This careful balance of reactivity ensures that the o-alkynyl phenylcyclobutanone intermediate is formed with high fidelity, preserving the integrity of the four-membered ring for the subsequent transformation.
In the second stage, the mechanism shifts to a Lewis acid-catalyzed pathway driven by Cu(OTf)2. The copper center coordinates with the carbonyl oxygen of the cyclobutanone, activating the ring towards nucleophilic attack or rearrangement. The patent proposes a mechanism involving the generation of a conjugated ketene intermediate through a ring-opening event, which then undergoes an intramolecular [2+2] cycloaddition with the proximal alkynyl group. 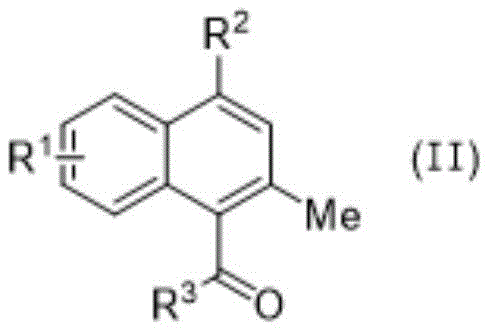 This is followed by a retro-[2+2] cycloaddition process that restores aromaticity and forms the stable naphthalenone skeleton. The inclusion of water as a co-reagent is critical, likely acting as a proton source to quench intermediates and regenerate the active copper catalyst. This mechanistic understanding allows for precise tuning of reaction parameters, ensuring that impurities arising from incomplete cyclization or polymerization are minimized, thereby delivering a product suitable for direct use in sensitive biological assays or further synthetic elaboration.
This is followed by a retro-[2+2] cycloaddition process that restores aromaticity and forms the stable naphthalenone skeleton. The inclusion of water as a co-reagent is critical, likely acting as a proton source to quench intermediates and regenerate the active copper catalyst. This mechanistic understanding allows for precise tuning of reaction parameters, ensuring that impurities arising from incomplete cyclization or polymerization are minimized, thereby delivering a product suitable for direct use in sensitive biological assays or further synthetic elaboration.
How to Synthesize O-Alkynyl Phenylcyclobutanone Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, strict adherence to the optimized parameters is essential. The process begins with the preparation of the reaction vessel under an inert atmosphere to prevent catalyst oxidation. The specific stoichiometry of 1.5 equivalents of terminal alkyne relative to the bromo-substrate ensures complete consumption of the valuable starting material while minimizing excess reagent waste. Maintaining the temperature at 90 °C for a duration of 12 hours provides the necessary activation energy without compromising thermal stability. Following the coupling reaction, the workup involves a neutralization step with dilute hydrochloric acid to remove residual base, followed by extraction and purification via silica gel chromatography. For the subsequent cyclization, the use of anhydrous chloroform and precise water addition (3 equivalents) is vital to drive the equilibrium towards the naphthalenone product. Detailed standardized operating procedures for these steps are outlined below to ensure reproducibility and safety.
- React o-bromophenyl cyclobutanone with terminal alkyne using Pd(TFA)2 and DPEphos ligand in 1,4-dioxane at 90 °C for 12 hours.
- Purify the resulting o-alkynyl phenylcyclobutanone via column chromatography using petroleum ether and ethyl acetate.
- Subject the purified intermediate to Cu(OTf)2 catalysis in chloroform with water at 65 °C for 18 hours to form naphthalenone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic advantages over legacy synthetic routes. The primary benefit lies in the significant reduction of manufacturing costs driven by the use of readily available and inexpensive starting materials. O-bromophenyl cyclobutanone and various terminal alkynes are commercially accessible commodities, eliminating the need for complex, multi-step precursor synthesis that often bottlenecks supply chains. Additionally, the high selectivity of the catalyst system reduces the burden on purification resources; fewer chromatography columns and less solvent consumption are required to achieve pharmaceutical-grade purity. This efficiency translates directly into lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives that many multinational corporations now mandate for their suppliers.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic copper co-catalysts in the initial coupling step, replaced by a more efficient palladium-ligand system, drastically lowers the cost of goods sold. By achieving yields exceeding 85% in many substrate variations, the process minimizes raw material waste and maximizes throughput per batch. The simplified purification protocol further reduces the consumption of high-purity solvents and silica gel, contributing to substantial cost savings in the overall production budget without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions means that production is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining continuous supply lines for downstream API manufacturing, reducing the risk of stockouts or delays caused by failed batches. Furthermore, the use of common solvents like 1,4-dioxane and chloroform ensures that solvent supply chains remain stable and unaffected by niche chemical shortages, providing a secure foundation for long-term procurement planning.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and atmospheric pressure, making it inherently safer and easier to scale from gram-scale laboratory synthesis to multi-kilogram commercial production. The absence of harsh reagents and the generation of minimal hazardous waste simplify the disposal process and reduce compliance costs associated with environmental regulations. This scalability ensures that as demand for the final drug product grows, the supply of this key intermediate can be ramped up quickly and efficiently to meet market needs without requiring massive capital investment in new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on catalyst selection, substrate compatibility, and process parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific project requirements.
Q: What catalyst system provides the highest yield for the Sonogashira coupling step?
A: The patent data indicates that using Pd(TFA)2 as the catalyst combined with the DPEphos ligand in 1,4-dioxane solvent yields the best results, achieving up to 90% conversion compared to other palladium sources.
Q: Can this method accommodate diverse substituents on the alkyne and aromatic ring?
A: Yes, the methodology demonstrates broad substrate tolerance, successfully processing various R groups including methyl, methoxy, phenyl, and substituted phenyl groups without significant loss in efficiency.
Q: What are the specific conditions required for the ring-expansion cyclization?
A: The cyclization requires Cu(OTf)2 (20 mol%) as a Lewis acid catalyst in chloroform solvent with 3 equivalents of water, heated to 65 °C for approximately 18 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Alkynyl Phenylcyclobutanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our team of expert chemists has thoroughly analyzed the patented processes for o-alkynyl phenylcyclobutanone and naphthalenone synthesis, confirming their potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume demands of even the largest pharmaceutical programs. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch we deliver meets the exacting standards required for clinical and commercial use.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary molecules. Let us help you accelerate your drug development timeline with reliable, cost-effective, and high-purity chemical solutions.
