Scalable FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Introduction to Advanced Quinazolinone Manufacturing
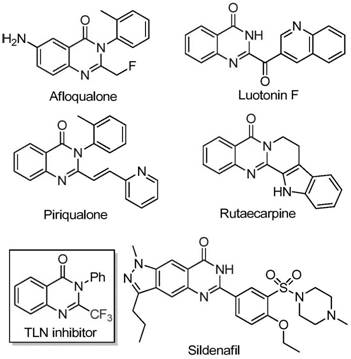
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current manufacturing paradigms. Quinazolinones are privileged scaffolds found in numerous drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory activities. The strategic introduction of a trifluoromethyl group further enhances these properties by improving metabolic stability, lipophilicity, and bioavailability. This technical insight analyzes the novel iron-catalyzed cyclization strategy that enables the efficient construction of these valuable cores.
Traditional synthetic approaches often rely on expensive fluorinating agents or harsh conditions that limit substrate scope and increase production costs. In contrast, the methodology described in CN111675662B utilizes a cost-effective iron catalyst system to drive the cyclization of readily available isatins and trifluoroethylimidoyl chlorides. This shift represents a significant advancement for reliable pharmaceutical intermediate suppliers aiming to optimize their supply chains. By leveraging abundant earth-abundant metals instead of precious catalysts, this process aligns with modern green chemistry principles while delivering high-purity products essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for synthesizing trifluoromethyl-substituted quinazolinones have been plagued by significant economic and operational inefficiencies. Conventional routes typically employ trifluoroacetic anhydride or ethyl trifluoroacetate as the trifluoromethyl source, reacting with anthranilamides or isatoic anhydrides. These processes frequently suffer from severe reaction conditions, requiring strict anhydrous environments or cryogenic temperatures that escalate energy consumption. Furthermore, the atom economy of these traditional pathways is often poor, generating substantial chemical waste that complicates disposal and increases environmental compliance costs.
Another critical drawback of legacy methods is the narrow substrate scope. Many existing protocols fail to tolerate diverse functional groups on the aromatic ring, limiting the chemical space available for medicinal chemists during lead optimization. Low yields and the necessity for expensive, specialized reagents further exacerbate the cost burden, making cost reduction in pharmaceutical intermediate manufacturing difficult to achieve. The reliance on precious metal catalysts in some alternative methods introduces additional purification challenges to meet stringent residual metal specifications required by regulatory bodies.
The Novel Approach
The innovative strategy outlined in the patent overcomes these hurdles by employing a direct cyclization between trifluoroethylimidoyl chloride and isatin derivatives. This approach utilizes ferric chloride (FeCl3) as a Lewis acid catalyst, which is not only inexpensive but also highly effective in promoting the necessary bond formations. The reaction proceeds smoothly in polar aprotic solvents like DMF under an air atmosphere, eliminating the need for costly inert gas protection systems. This simplicity translates directly into operational ease and reduced capital expenditure for manufacturing facilities.
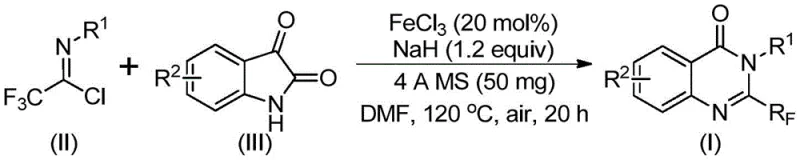
Moreover, this novel route demonstrates exceptional functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and methoxy groups on both the isatin and the imidoyl chloride components. The ability to synthesize a wide array of derivatives from common starting materials significantly accelerates the drug discovery timeline. By avoiding the use of hazardous fluorinating gases or unstable intermediates, the process enhances workplace safety and simplifies regulatory approval for commercial production. The high conversion rates observed across multiple examples validate the robustness of this method for industrial application.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The success of this synthesis relies on the unique reactivity of the iron catalyst in facilitating a cascade of bond-forming events. Mechanistically, the reaction likely initiates with the activation of the carbonyl group of the isatin by the Lewis acidic FeCl3 species. Simultaneously, the base (NaH) deprotonates the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial step forms a key carbon-nitrogen bond, creating a trifluoroacetamidine intermediate that serves as the precursor for cyclization.
Following the initial coupling, the iron catalyst promotes a decarbonylation event, which is crucial for establishing the quinazolinone core structure. This step involves the extrusion of carbon monoxide and the subsequent intramolecular cyclization to close the six-membered ring. The presence of the trifluoromethyl group at the 2-position is retained throughout this transformation, ensuring the final product possesses the desired electronic properties. The use of 4A molecular sieves in the reaction mixture plays a vital role in scavenging trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material or deactivate the catalyst.
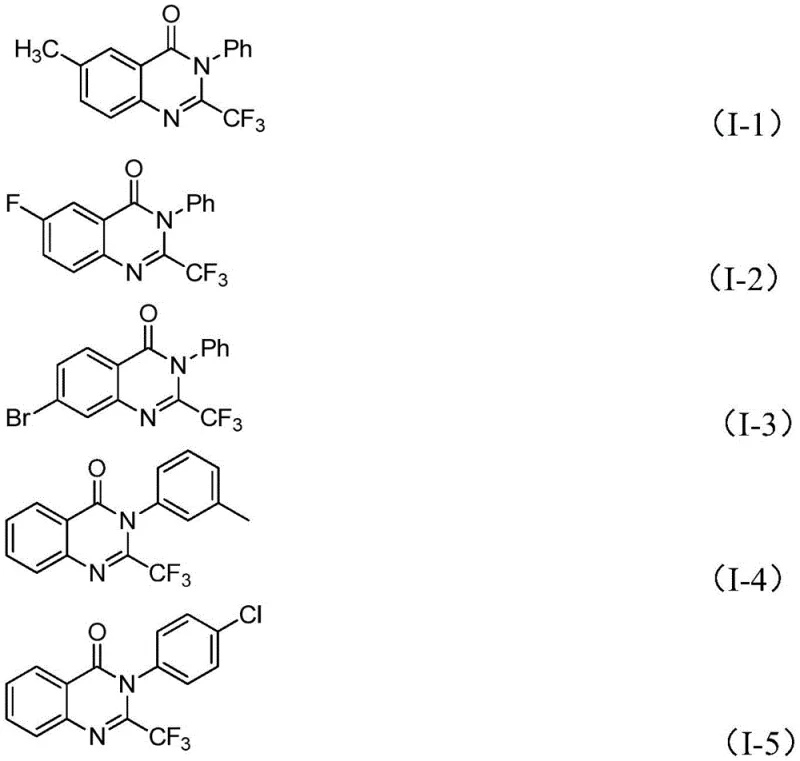
Impurity control is inherently managed by the high selectivity of the iron-catalyzed pathway. Unlike radical-based trifluoromethylation methods that often produce complex mixtures of regioisomers, this ionic mechanism directs the formation of the specific 2-trifluoromethyl isomer with high fidelity. The reaction conditions are optimized to minimize side reactions such as polymerization or over-fluorination. Post-reaction workup involving simple filtration and silica gel treatment effectively removes iron residues and inorganic salts, yielding a crude product that is easily purified to high standards via column chromatography. This streamlined purification profile is essential for maintaining high throughput in a commercial setting.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The protocol involves mixing the catalyst, base, and molecular sieves with the substrates in a solvent like DMF, followed by a staged heating process. Initially, the reaction is maintained at a moderate temperature to facilitate the coupling step, after which the temperature is elevated to drive the cyclization and decarbonylation to completion. Detailed standardized operating procedures regarding stoichiometry, addition rates, and quenching protocols are critical for reproducibility.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then increase temperature to 120°C and react for an additional 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final 2-trifluoromethyl quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this iron-catalyzed methodology offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By utilizing isatin and trifluoroethylimidoyl chloride, which are commodity chemicals available from multiple global vendors, companies can mitigate supply risk and avoid dependency on single-source specialty reagents. This diversification of the supply base enhances resilience against market fluctuations and logistical disruptions.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with ferric chloride results in a substantial decrease in raw material costs. Iron salts are orders of magnitude cheaper than palladium or rhodium complexes, and they do not require expensive phosphine ligands. Furthermore, the elimination of rigorous inert atmosphere requirements reduces utility costs associated with nitrogen or argon consumption. The simplified workup procedure minimizes solvent usage and waste disposal fees, contributing to a leaner overall cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted production schedules. The tolerance for air and moisture simplifies the engineering controls needed in the manufacturing plant, allowing for faster turnaround times between batches. Since the starting materials are stable and widely produced, lead times for raw material procurement are significantly shortened, enabling a more responsive just-in-time inventory strategy for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The use of DMF, while requiring careful handling, is a standard industrial solvent with well-established recovery and recycling protocols. The absence of toxic fluorinating gases like HF or Selectfluor improves the safety profile of the plant, reducing insurance premiums and regulatory scrutiny. This alignment with green chemistry metrics supports corporate sustainability goals and facilitates smoother regulatory filings for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the fit of this method within their existing manufacturing frameworks. The answers are derived directly from the experimental data and technical disclosures found in the patent literature.
Q: What are the key advantages of using FeCl3 for quinazolinone synthesis?
A: The use of Ferric Chloride (FeCl3) offers significant cost advantages over precious metal catalysts while maintaining high catalytic efficiency. It enables a robust cyclization process with excellent functional group tolerance, allowing for the synthesis of diverse derivatives without requiring expensive ligands or inert atmospheres.
Q: What starting materials are required for this novel preparation method?
A: The process utilizes readily available and inexpensive starting materials: isatin derivatives and trifluoroethylimidoyl chloride. These substrates are commercially accessible and eliminate the need for complex, multi-step pre-functionalization often required in traditional trifluoromethylation strategies.
Q: Does this method support large-scale production of quinazolinone intermediates?
A: Yes, the method is designed for scalability. It operates under relatively mild conditions (air atmosphere, standard solvents like DMF) and uses cheap catalysts, making it highly suitable for industrial scale-up from gram-level laboratory synthesis to multi-kilogram commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the demand for fluorinated heterocycles continues to surge in oncology and CNS drug development, securing a capable manufacturing partner is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the FeCl3-mediated cyclization to deliver superior intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for clinical and commercial use.
We invite you to collaborate with us to optimize your supply chain for quinazolinone-based therapeutics. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, high-quality chemical solutions.
