Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access privileged scaffolds like quinazolinones, which serve as the structural backbone for numerous bioactive agents ranging from antifungals to anticancer drugs. As highlighted in recent intellectual property developments, specifically Patent CN112480015B, a significant breakthrough has been achieved in the efficient construction of 2-trifluoromethyl substituted quinazolinones. This patent discloses a novel multi-component one-pot method that leverages transition metal catalysis to streamline the synthesis of these valuable heterocycles. The introduction of the trifluoromethyl group is particularly strategic, as it profoundly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these intermediates highly desirable for modern drug discovery programs. By utilizing a palladium-catalyzed carbonylation cascade, this technology addresses critical bottlenecks in traditional synthetic routes, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production.
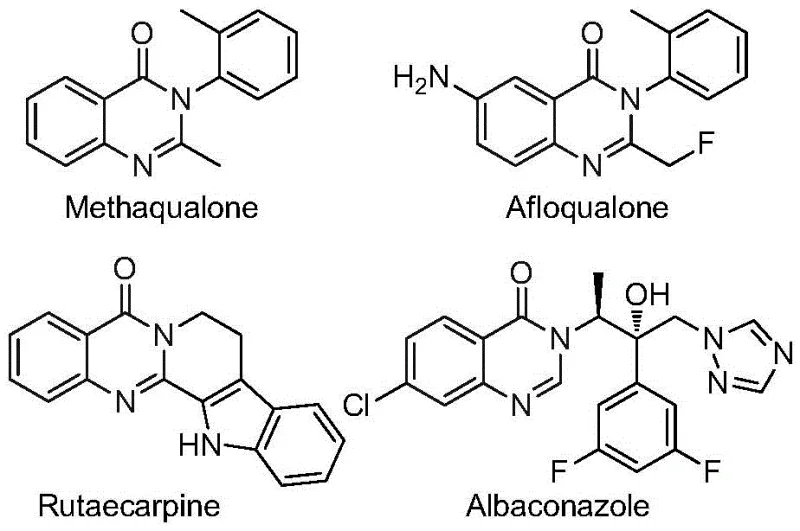
The significance of this technological advancement cannot be overstated for R&D directors and process chemists who are tasked with optimizing supply chains for complex active pharmaceutical ingredients (APIs). The ability to synthesize these fluorinated heterocycles from simple, commodity-grade starting materials represents a paradigm shift in how we approach the manufacturing of high-value pharmaceutical intermediates. Unlike legacy methods that often rely on hazardous gases or multi-step sequences with poor atom economy, this one-pot approach consolidates multiple bond-forming events into a single operational unit. This consolidation not only reduces the physical footprint required for manufacturing but also minimizes the potential for material loss between steps, thereby enhancing the overall process mass intensity (PMI). For a reliable pharmaceutical intermediate supplier, adopting such innovative protocols is essential to maintaining competitiveness in a market that demands both speed and sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has been plagued by significant operational and economic challenges that hinder their widespread adoption in industrial settings. Traditional pathways often necessitate the use of high-pressure carbon monoxide gas, which introduces severe safety hazards and requires specialized, capital-intensive reactor infrastructure that many contract manufacturing organizations (CMOs) lack. Furthermore, conventional strategies frequently depend on pre-activated substrates, such as 2-bromoformylanilines or acid anhydrides, which are not only expensive to procure but also generate substantial amounts of stoichiometric waste during the activation process. Another critical limitation is the narrow substrate scope associated with older catalytic systems; many established methods fail to tolerate sensitive functional groups or require harsh reaction conditions that lead to decomposition of the desired product. These factors collectively contribute to elevated production costs, extended lead times, and a higher environmental burden, making the cost reduction in API manufacturing difficult to achieve using legacy technologies.
The Novel Approach
In stark contrast, the methodology described in Patent CN112480015B offers a transformative solution by employing a multi-component coupling strategy that bypasses the need for gaseous CO and pre-functionalized precursors. This novel approach utilizes nitro compounds, which are ubiquitous, inexpensive, and commercially available in vast structural diversity, as the primary nitrogen source. By integrating a solid carbon monoxide surrogate, molybdenum hexacarbonyl (Mo(CO)6), the reaction proceeds under standard atmospheric pressure conditions, drastically simplifying the engineering requirements and enhancing operational safety. The one-pot nature of this synthesis means that the reduction of the nitro group, the formation of the amidine intermediate, and the subsequent carbonylative cyclization all occur sequentially in the same vessel without the need for intermediate isolation. This telescoping of reactions results in a drastic simplification of the workflow, significantly reducing solvent consumption and labor costs while improving the overall yield profile. For procurement managers, this translates to a more resilient supply chain where raw material availability is guaranteed, and production timelines are compressed.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
To fully appreciate the sophistication of this synthetic route, one must delve into the intricate mechanistic details that govern the transformation. The reaction is initiated by the in situ reduction of the nitro compound to the corresponding amine, likely facilitated by the molybdenum species or the palladium catalyst under the thermal conditions employed. Once the amine is generated, it undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. This step is crucial as it installs the necessary nitrogen framework for the subsequent ring closure. Following this, the palladium catalyst, coordinated by the bidentate phosphine ligand dppp, undergoes oxidative addition into the carbon-iodine bond of the imidoyl chloride moiety (or a related aryl iodide species depending on the specific substrate design), generating a reactive organopalladium(II) species. Simultaneously, the thermal decomposition of Mo(CO)6 releases carbon monoxide in a controlled manner, which then inserts into the carbon-palladium bond to form an acyl-palladium complex.
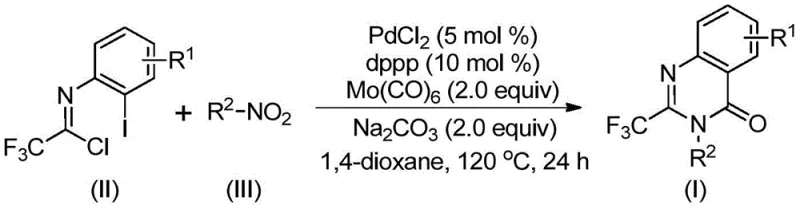
The final stages of the catalytic cycle involve an intramolecular nucleophilic attack by the nitrogen atom of the amidine onto the acyl-palladium center, leading to the formation of a seven-membered palladacycle intermediate. This cyclization step is promoted by the presence of the base, sodium carbonate, which helps to neutralize the acidic byproducts and drive the equilibrium forward. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium(0) catalyst for the next turnover. Understanding this mechanism is vital for impurity control, as it highlights the importance of maintaining precise stoichiometric ratios and temperature profiles to prevent side reactions such as homocoupling or incomplete carbonylation. The robustness of this catalytic system allows for excellent functional group tolerance, ensuring that sensitive moieties like halogens and ethers remain intact throughout the rigorous reaction conditions, thereby delivering high-purity intermediates suitable for downstream pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this advanced synthesis protocol requires careful attention to reaction parameters to maximize efficiency and reproducibility. The process begins with the precise weighing of the palladium catalyst, ligand, and additives, followed by their suspension in a high-boiling aprotic solvent such as 1,4-dioxane, which has been identified as the optimal medium for solubilizing all components and facilitating the reaction kinetics. The detailed standardized synthesis steps are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale execution.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic viability and logistical stability of chemical production, the adoption of this patented methodology offers compelling strategic benefits. The shift away from hazardous gaseous reagents and expensive activated substrates fundamentally alters the cost structure of manufacturing these complex heterocycles. By leveraging commodity chemicals like nitro compounds, manufacturers can decouple their production costs from the volatility of specialized reagent markets, ensuring more predictable budgeting and pricing stability for their clients. Furthermore, the operational simplicity of the one-pot process reduces the demand for highly skilled labor and complex equipment maintenance, contributing to a leaner and more agile manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure represents a massive capital expenditure saving, as facilities no longer require specialized autoclaves or extensive safety monitoring systems for toxic gases. Additionally, the use of Mo(CO)6 as a solid CO source allows for precise dosing and minimizes waste, leading to substantial cost savings in raw material consumption. The high atom economy of the multi-component reaction ensures that a greater proportion of the input mass is converted into the final product, reducing the cost per kilogram of the active intermediate. This efficiency is further amplified by the reduced need for intermediate purification steps, which typically consume large volumes of solvents and silica gel, thereby lowering both material and disposal costs significantly.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on nitro compounds and simple aryl halides, which are produced globally in massive quantities and are less susceptible to supply disruptions compared to custom-synthesized building blocks. The robustness of the reaction conditions means that production schedules are less likely to be impacted by batch failures or quality deviations, ensuring consistent on-time delivery to downstream API manufacturers. This reliability is critical for maintaining continuous production lines in the pharmaceutical sector, where delays can have cascading effects on drug launch timelines. By diversifying the supplier base for these generic starting materials, procurement teams can mitigate risks associated with single-source dependencies and geopolitical instability.
- Scalability and Environmental Compliance: The transition from batch processes involving hazardous gases to a safer, liquid-phase one-pot reaction facilitates easier scale-up from gram to tonne quantities without proportional increases in risk. The reduced generation of hazardous waste streams aligns with increasingly stringent environmental regulations, minimizing the regulatory burden and permitting costs associated with chemical manufacturing. The ability to run the reaction in common solvents like dioxane, which can be recovered and recycled, further enhances the sustainability profile of the process. This environmental stewardship not only protects the corporate reputation but also future-proofs the supply chain against tightening global emissions standards and waste disposal restrictions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method, derived directly from the experimental data and scope defined in the patent literature. These insights are intended to assist process development teams in evaluating the feasibility of this route for their specific project needs.
Q: What are the key advantages of this one-pot synthesis method over traditional routes?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap and readily available nitro compounds and trifluoroethylimidoyl chloride, offering broader substrate compatibility and higher operational safety.
Q: What catalyst system is required for this transformation?
A: The reaction employs a palladium catalyst system consisting of PdCl2 (5 mol %) and the ligand 1,3-bis(diphenylphosphino)propane (dppp, 10 mol %), with Mo(CO)6 serving as the solid carbon monoxide source.
Q: Can this process tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating various substituents such as halogens (F, Cl, Br), alkyl groups, and trifluoromethyl groups on both the aromatic ring and the nitrogen substituent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the global demand for fluorinated pharmaceutical intermediates continues to surge, partnering with a technically proficient CDMO is essential for translating innovative academic research into commercial reality. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs that ensure stringent purity specifications are met for every batch. We understand that the successful commercialization of complex molecules like 2-trifluoromethyl quinazolinones requires more than just a recipe; it demands a partner who can navigate the complexities of process safety, regulatory compliance, and supply chain logistics with equal expertise.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthesis technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our optimized manufacturing processes can accelerate your drug development timeline while maximizing your return on investment. Let us be your trusted ally in bringing the next generation of life-saving medicines to market.
