Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharma Applications
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharma Applications
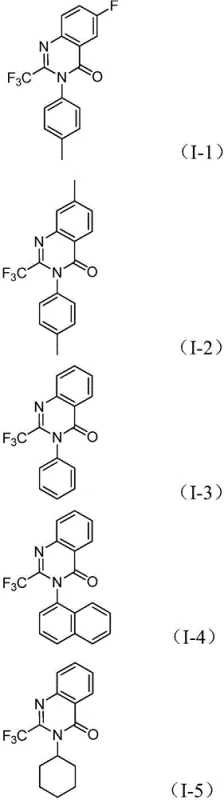
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly quinazolinones, which serve as critical scaffolds in numerous bioactive molecules ranging from antifungals to anticancer agents. Patent CN112480015B introduces a transformative multi-component one-pot strategy for synthesizing 2-trifluoromethyl substituted quinazolinones, addressing long-standing challenges in efficiency and substrate availability. This innovative approach leverages a palladium-catalyzed carbonylation cascade that merges trifluoroethylimidoyl chloride with nitro compounds, bypassing the need for hazardous high-pressure carbon monoxide gas. For R&D directors and procurement specialists, this technology represents a significant leap forward in process intensification, offering a pathway to high-purity pharmaceutical intermediates with reduced operational complexity and enhanced safety profiles suitable for commercial manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the quinazolinone core often rely on harsh reaction conditions that pose significant safety risks and economic burdens in an industrial setting. Conventional methods frequently necessitate the use of high-pressure carbon monoxide cylinders, which require specialized autoclaves and rigorous safety protocols to prevent leakage and exposure, thereby inflating capital expenditure and operational overheads. Furthermore, many established protocols depend on pre-functionalized starting materials such as 2-bromoformylanilines or 2-iodoanilines, which are not only expensive to procure but also generate substantial stoichiometric amounts of halogenated waste during the cyclization process. These legacy methods often suffer from narrow substrate scope, failing to tolerate sensitive functional groups required for modern drug discovery, and typically involve multi-step sequences with intermediate isolation that drastically reduce overall yield and throughput efficiency.
The Novel Approach
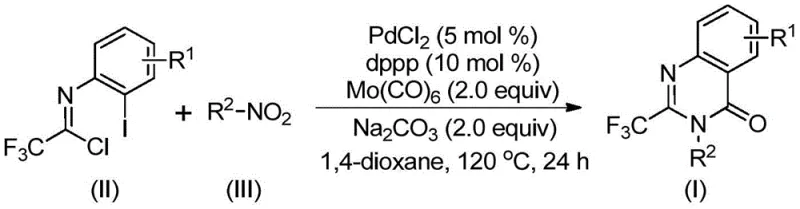
In stark contrast, the novel methodology disclosed in the patent utilizes a sophisticated palladium-catalyzed system that operates under atmospheric pressure conditions using solid Mo(CO)6 as a safe and controllable carbon monoxide surrogate. This strategic substitution eliminates the engineering complexities associated with high-pressure gas handling, allowing the reaction to proceed in standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. The use of readily available nitro compounds as nitrogen sources further democratizes the synthesis, as these feedstocks are commoditized globally and cost a fraction of their halogenated counterparts. By consolidating reduction, coupling, and cyclization into a single operational pot, this method minimizes solvent consumption and waste generation while delivering excellent yields across a broad range of electronic and steric environments.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of reduction, insertion, and elimination steps orchestrated by the palladium center. The cycle initiates with the in situ reduction of the nitro compound to the corresponding amine by Mo(CO)6, which simultaneously serves as the carbonyl source for subsequent steps. Following this reduction, a base-promoted condensation occurs between the generated amine and trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate, setting the stage for the metal-catalyzed cyclization. The palladium catalyst then undergoes oxidative addition into the carbon-iodine bond of the imidoyl chloride moiety, forming a reactive divalent palladium species that is primed for carbonyl insertion.
Upon heating, the Mo(CO)6 releases carbon monoxide which inserts into the carbon-palladium bond to generate an acyl-palladium intermediate, a crucial step that builds the carbonyl functionality of the quinazolinone ring. Subsequent intramolecular coordination of the nitrogen atom to the palladium center facilitates the formation of a seven-membered palladacycle, which ultimately undergoes reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product and regenerate the active catalyst. This detailed understanding of the catalytic cycle allows process chemists to fine-tune reaction parameters such as ligand selection and base strength to maximize turnover numbers and minimize the formation of side products, ensuring consistent quality for regulatory submissions.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction monitoring to ensure optimal conversion and purity profiles suitable for pharmaceutical applications. The protocol dictates the combination of palladium chloride, a bidentate phosphine ligand such as dppp, and sodium carbonate in an aprotic solvent like 1,4-dioxane to create the active catalytic environment. Detailed standardized synthesis steps see the guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, and Mo(CO)6 with trifluoroethylimidoyl chloride and nitro compound in an organic solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to ensure complete conversion and cyclization.
- Perform post-treatment via filtration and silica gel mixing, followed by column chromatography to isolate the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling value propositions that directly address the key pain points of cost volatility and supply chain fragility in the fine chemical sector. The shift from expensive, pre-activated halogenated substrates to commodity nitro compounds fundamentally alters the cost structure of the manufacturing process, enabling significant margin improvements without compromising on quality or yield. Additionally, the elimination of high-pressure equipment requirements reduces the barrier to entry for contract manufacturing organizations, allowing for more flexible production scheduling and faster response times to market demand fluctuations. The robustness of the catalytic system ensures consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply agreements with multinational pharmaceutical partners.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of high-cost starting materials with inexpensive nitro compounds that are abundantly available in the global chemical market. By removing the need for stoichiometric amounts of expensive halogenated precursors and avoiding the capital intensity of high-pressure reactor infrastructure, the overall cost of goods sold is drastically simplified and optimized. Furthermore, the one-pot nature of the reaction reduces solvent usage and energy consumption associated with multiple isolation and purification steps, contributing to substantial cost savings in utility and waste disposal expenditures.
- Enhanced Supply Chain Reliability: Utilizing nitro compounds as key building blocks enhances supply chain resilience because these chemicals are produced by a wide network of manufacturers worldwide, mitigating the risk of single-source dependency. The stability of the solid Mo(CO)6 reagent compared to gaseous carbon monoxide simplifies logistics and storage requirements, ensuring that production can continue uninterrupted even during periods of logistical constraint. This reliability is paramount for procurement managers who need to guarantee continuous material flow for downstream drug substance manufacturing without the risk of unexpected shutdowns due to reagent shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been demonstrated to work efficiently from gram scale to potential tonnage production without significant loss in performance. The use of a solid CO surrogate significantly lowers the environmental footprint by eliminating the risks associated with high-pressure gas leaks and reducing the energy load required for compression. Moreover, the high atom economy of the multi-component reaction aligns with green chemistry principles, facilitating easier regulatory approval and compliance with increasingly stringent environmental protection standards in major manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in an industrial context. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide clarity on feasibility and performance. Understanding these details is essential for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using nitro compounds in this synthesis?
A: Nitro compounds serve as inexpensive and readily available starting materials that eliminate the need for costly pre-activated halogenated substrates, significantly reducing raw material costs and simplifying the supply chain logistics for large-scale production.
Q: How does the one-pot method impact process safety and scalability?
A: By utilizing Mo(CO)6 as a solid carbon monoxide surrogate instead of high-pressure CO gas, the method drastically improves operational safety and allows for easier scale-up in standard reactor vessels without specialized high-pressure equipment.
Q: Is this method compatible with diverse functional groups for drug discovery?
A: Yes, the catalytic system exhibits excellent substrate tolerance, accommodating various substituents such as halogens, alkyl groups, and trifluoromethyl moieties on both the aromatic ring and the nitrogen substituent, facilitating rapid analog synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to translate complex laboratory innovations like this palladium-catalyzed protocol into reliable commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
