Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Applications
Introduction to Next-Generation Imidazole Synthesis
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds has become a cornerstone of modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As highlighted in recent literature, imidazole compounds serve as critical nitrogen-containing five-membered heterocycles found in numerous bioactive molecules, ranging from antihistamines to complex coordination ligands. 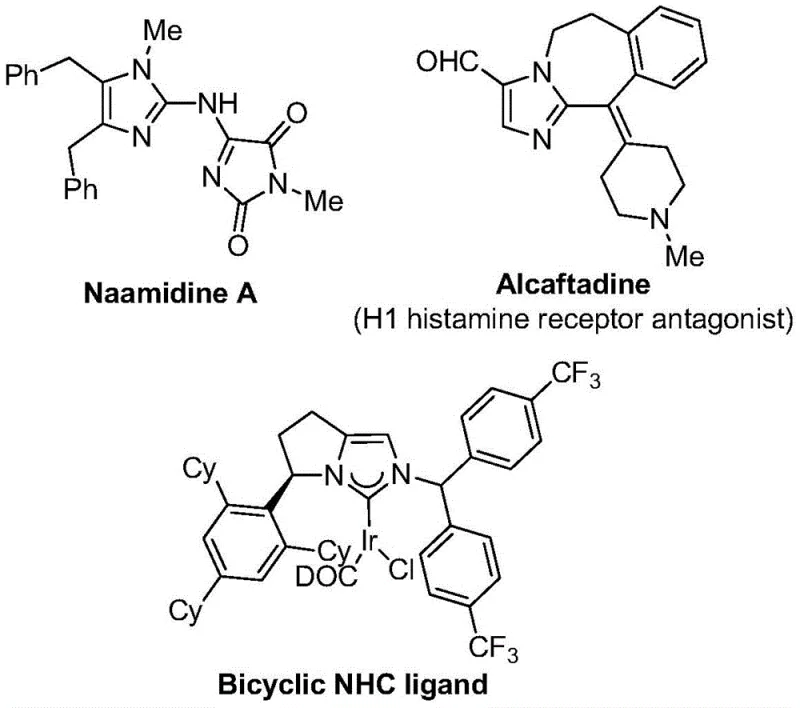 Patent CN111423381B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses long-standing challenges in synthetic efficiency and substrate compatibility. This novel approach leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained trifluoroethyliminato chloride, propargylamine, and diaryl iodide as starting materials. By shifting away from harsh traditional conditions, this technology offers a robust pathway for generating high-purity pharmaceutical intermediates, positioning it as a vital tool for R&D teams seeking to optimize their synthetic routes for complex API backbones.
Patent CN111423381B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses long-standing challenges in synthetic efficiency and substrate compatibility. This novel approach leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained trifluoroethyliminato chloride, propargylamine, and diaryl iodide as starting materials. By shifting away from harsh traditional conditions, this technology offers a robust pathway for generating high-purity pharmaceutical intermediates, positioning it as a vital tool for R&D teams seeking to optimize their synthetic routes for complex API backbones.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on direct reactions between synthons containing the trifluoromethyl group and suitable substrates. While effective in specific contexts, these conventional methods often suffer from significant drawbacks that hinder their utility in large-scale manufacturing. Common issues include the requirement for extremely harsh reaction conditions, such as high temperatures and pressures, which can lead to thermal decomposition of sensitive functional groups. Furthermore, many traditional protocols utilize hazardous reagents like trifluorodiazoethane, which poses serious safety risks due to its explosive nature and toxicity. The limited substrate scope of older methodologies often restricts the diversity of substituents that can be introduced at the 1 and 5 positions of the imidazole ring, thereby limiting the chemical space available for drug discovery optimization. Additionally, the reliance on stoichiometric amounts of expensive or difficult-to-handle reagents often results in poor atom economy and generates substantial waste, creating environmental and cost burdens for process chemistry teams.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in patent CN111423381B presents a highly efficient and versatile alternative centered on palladium catalysis. 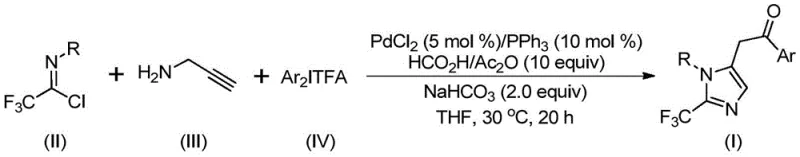 This innovative route employs a multicomponent reaction strategy where trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts converge in the presence of a palladium catalyst and a carbon monoxide surrogate. The reaction proceeds under remarkably mild conditions, typically at 30°C for 16 to 24 hours, which drastically reduces energy consumption and minimizes the risk of side reactions associated with thermal stress. A key feature of this approach is the use of formic acid and acetic anhydride as a safe, in-situ source of carbon monoxide, eliminating the need for handling toxic CO gas cylinders. The protocol demonstrates exceptional functional group tolerance, accommodating various substituents such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, and nitro groups on the aryl rings without compromising yield. This broad compatibility allows chemists to rapidly synthesize a diverse library of 2-trifluoromethyl imidazole derivatives, accelerating the lead optimization phase in drug development projects.
This innovative route employs a multicomponent reaction strategy where trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts converge in the presence of a palladium catalyst and a carbon monoxide surrogate. The reaction proceeds under remarkably mild conditions, typically at 30°C for 16 to 24 hours, which drastically reduces energy consumption and minimizes the risk of side reactions associated with thermal stress. A key feature of this approach is the use of formic acid and acetic anhydride as a safe, in-situ source of carbon monoxide, eliminating the need for handling toxic CO gas cylinders. The protocol demonstrates exceptional functional group tolerance, accommodating various substituents such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, and nitro groups on the aryl rings without compromising yield. This broad compatibility allows chemists to rapidly synthesize a diverse library of 2-trifluoromethyl imidazole derivatives, accelerating the lead optimization phase in drug development projects.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to replicate or modify the process for specific applications. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the base, sodium bicarbonate, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align for the catalytic cycle. The palladium catalyst, generated from palladium chloride and triphenylphosphine, facilitates the activation of the alkyne moiety in the propargylamine derivative through a palladation event, forming a vinyl-palladium intermediate. This intermediate then isomerizes to a more stable alkyl-palladium species, setting the stage for the critical carbonylation step. Under the influence of carbon monoxide released from the formic acid/acetic anhydride mixture, an acyl-palladium intermediate is formed via insertion of CO into the palladium-carbon bond. The cycle culminates with the oxidative addition of the diaryl iodonium salt to the palladium center, generating a high-valent tetravalent palladium intermediate. Finally, reductive elimination occurs, releasing the desired 2-trifluoromethyl-substituted imidazole product and regenerating the active palladium catalyst for the next turnover. This intricate dance of organometallic steps ensures high selectivity and efficiency, minimizing the formation of regioisomers or polymeric byproducts that often plague alkyne functionalization reactions.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in maintaining product purity. High-temperature processes often promote degradation pathways or non-selective radical reactions that generate complex impurity profiles difficult to remove during downstream processing. By operating at near-ambient temperatures, this method suppresses thermal degradation of the sensitive trifluoromethyl group and the imidazole ring. Furthermore, the use of well-defined starting materials like trifluoroethylimidoyl chloride and specific diaryl iodonium salts ensures that the reaction trajectory is highly predictable. The post-treatment procedure described involves simple filtration followed by silica gel mixing and column chromatography, indicating that the crude reaction mixture is relatively clean. For industrial applications, this implies that extensive recrystallization or preparative HPLC steps might be avoided, streamlining the purification workflow and reducing solvent usage. The ability to tune the electronic properties of the aryl groups on both the imidoyl chloride and the iodonium salt allows for fine-tuning of the reaction kinetics, providing an additional lever for process engineers to optimize conversion rates and minimize residual starting materials.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to ensure reproducibility. The protocol is designed to be user-friendly, utilizing standard Schlenk techniques for air-sensitive catalysis but avoiding extreme conditions. The molar ratios are optimized to balance cost and conversion, typically employing a slight excess of the trifluoroethylimidoyl chloride and diaryl iodonium salt relative to the propargylamine to drive the reaction to completion. The choice of solvent is also critical; while several aprotic solvents were screened, tetrahydrofuran (THF) was identified as the optimal medium, providing superior solubility for all components and promoting the highest conversion rates. Detailed standardized synthesis steps, including precise addition orders and workup procedures, are outlined below to assist technical teams in adopting this methodology.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The shift towards a palladium-catalyzed process using commodity chemicals represents a strategic move towards cost-effective and sustainable manufacturing. By analyzing the input materials and process conditions, we can identify several key areas where operational expenditures can be optimized without sacrificing product quality.
- Cost Reduction in Manufacturing: One of the most significant economic drivers of this technology is the utilization of inexpensive and widely available starting materials. Trifluoroethylimidoyl chloride can be rapidly synthesized from corresponding aromatic amines, which are bulk commodities in the fine chemical industry. Similarly, propargylamine is a low-cost building block produced on a massive scale globally. The catalyst system relies on palladium chloride and triphenylphosphine, which, while precious metal-based, are used in low loading (5 mol% PdCl2 and 10 mol% PPh3), minimizing the impact on the overall bill of materials. Furthermore, the replacement of toxic gaseous carbon monoxide with a liquid mixture of formic acid and acetic anhydride eliminates the need for specialized high-pressure equipment and safety infrastructure associated with CO handling, leading to substantial capital expenditure savings. The mild reaction temperature of 30°C also translates to significantly lower energy costs for heating and cooling compared to traditional reflux conditions, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad availability of the requisite reagents. Unlike specialized fluorinating agents that may have limited suppliers and long lead times, the aryl amines and iodonium salts used here are accessible from multiple global vendors. The robustness of the reaction against functional group variations means that supply disruptions for specific substituted precursors can often be mitigated by switching to alternative analogs without redesigning the entire process. The simplicity of the post-treatment, involving standard filtration and chromatography, reduces dependency on complex purification resins or proprietary scavengers that might be subject to supply constraints. This flexibility ensures a more stable and continuous supply of the final 2-trifluoromethyl imidazole intermediates, critical for maintaining uninterrupted API production schedules.
- Scalability and Environmental Compliance: The protocol's design inherently supports scalability, having been demonstrated effectively at the gram level with clear pathways for expansion to kilogram and ton scales. The use of THF as a solvent is advantageous as it is a standard industrial solvent with well-established recovery and recycling protocols, aligning with green chemistry principles. The avoidance of explosive diazo compounds and toxic CO gas significantly lowers the environmental, health, and safety (EHS) risk profile of the manufacturing process. This reduction in hazard potential simplifies regulatory compliance and permitting processes for new production lines. Additionally, the high reaction efficiency and selectivity reduce the generation of hazardous waste streams, lowering disposal costs and supporting corporate sustainability goals. The ability to produce diverse derivatives through simple substrate changes allows for a flexible manufacturing platform capable of responding quickly to market demands for different API intermediates.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled answers to common questions regarding the synthesis and application of these compounds. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the key advantages of this palladium-catalyzed method over traditional imidazole synthesis?
A: This method operates under mild conditions (30°C) compared to traditional high-temperature cyclizations, utilizes cheap and readily available starting materials like propargylamine, and offers excellent functional group tolerance for diverse substrate design.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial large-scale production due to its simple operation, high reaction efficiency, and use of inexpensive catalysts like palladium chloride.
Q: What represents the carbon monoxide source in this carbonylation reaction?
A: Instead of using toxic gaseous carbon monoxide directly, this protocol employs a safe in-situ generation system using a mixture of formic acid and acetic anhydride as the carbonyl surrogate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
The emergence of efficient synthetic routes like the one described in CN111423381B underscores the dynamic nature of pharmaceutical intermediate manufacturing. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such advanced chemistries to the market. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs ensuring that every batch of 2-trifluoromethyl imidazole meets the exacting standards required for drug substance synthesis. We understand that consistency and reliability are paramount for our partners, and our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed processes, including efficient metal scavenging and solvent recovery.
We invite you to explore how our capabilities can support your next project. Whether you require custom synthesis of novel imidazole derivatives or scale-up of existing routes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and cost-efficiency in the competitive landscape of modern drug development.
