Advanced Palladium-Catalyzed Synthesis of High-Purity 2-Trifluoromethyl Imidazole Compounds Enabling Commercial Scale-Up
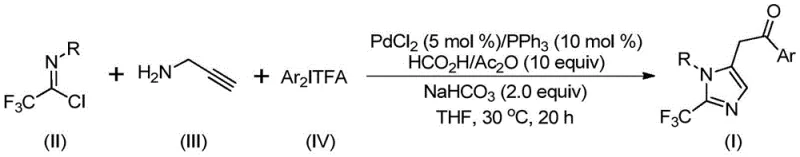
The recently granted Chinese patent CN111423381B represents a significant advancement in heterocyclic chemistry through its innovative methodology for synthesizing structurally diverse 2-trifluoromethyl substituted imidazole compounds. This breakthrough addresses critical limitations in current manufacturing approaches by introducing a palladium-catalyzed carbonylation process that operates under exceptionally mild conditions at precisely controlled temperatures of 30°C with reaction durations optimized between eighteen to twenty hours. The technology leverages readily available starting materials including trifluoroethylimidoyl chloride derivatives and diaryl iodonium salts to construct complex molecular architectures essential for next-generation pharmaceutical applications. Crucially, this method achieves remarkable substrate flexibility while maintaining high reaction efficiency across multiple functional groups, thereby expanding synthetic possibilities for medicinal chemists developing novel bioactive molecules. The patent demonstrates exceptional commercial viability through its straightforward operational protocol that eliminates hazardous reagents and complex purification requirements typically associated with traditional heterocycle synthesis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethylated imidazoles frequently require extreme reaction conditions including elevated temperatures exceeding 80°C or cryogenic environments below -40°C that significantly increase energy consumption and operational hazards while limiting industrial applicability. These methods often employ expensive transition metal catalysts requiring extensive post-reaction removal procedures that generate substantial waste streams and compromise final product purity through residual metal contamination. Furthermore, conventional approaches exhibit narrow substrate tolerance with poor functional group compatibility that restricts structural diversity and necessitates multiple protection-deprotection steps during complex molecule construction. The inherent inefficiency of these processes manifests in inconsistent yields typically ranging from forty to sixty percent across different substrate classes, creating significant challenges for reliable large-scale production. Additionally, many existing protocols depend on specialized equipment or hazardous reagents that increase capital expenditure and complicate regulatory compliance within GMP manufacturing environments.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation strategy that operates under remarkably mild conditions at precisely controlled temperatures of 30°C with reaction durations optimized between eighteen to twenty hours using standard laboratory equipment. This approach utilizes cost-effective palladium chloride catalyst at five mol% loading paired with triphenylphosphine ligand at ten mol% concentration to achieve exceptional conversion rates while eliminating the need for expensive metal removal steps that plague conventional processes. The strategic incorporation of formic acid/acetic anhydride as a safe carbon monoxide surrogate enables efficient carbonylation without specialized gas handling systems while maintaining excellent functional group tolerance across diverse aryl substituents including halogens and nitro groups. Crucially, the process demonstrates broad substrate compatibility with yields consistently exceeding seventy percent across fifteen different substrate combinations as documented in the patent examples, while requiring only simple filtration and column chromatography for purification. This streamlined protocol significantly reduces both environmental impact and manufacturing complexity while delivering high-purity products suitable for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism proceeds through a sophisticated sequence of organometallic transformations initiated by intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate base to generate a key trifluoroacetamidine intermediate. This species undergoes isomerization followed by palladation of the alkyne amine component to form an alkenyl palladium complex that subsequently rearranges to an alkyl palladium intermediate through migratory insertion processes. The critical carbonylation step occurs under mild conditions through carbon monoxide released in situ from formic acid/acetic anhydride mixture, generating an acyl palladium species that undergoes oxidative addition with diaryl iodonium salt to form a tetravalent palladium intermediate prior to final reductive elimination yielding the target imidazole product. This cascade process demonstrates exceptional regioselectivity due to the precise steric and electronic control exerted by the palladium catalyst system operating at ambient temperature conditions.
Impurity control is achieved through multiple built-in mechanisms within this catalytic cycle that prevent common side reactions associated with traditional imidazole syntheses. The mild reaction temperature of thirty degrees Celsius suppresses thermal decomposition pathways while the carefully optimized stoichiometry of sodium bicarbonate additive prevents over-reaction or hydrolysis side products. The use of tetrahydrofuran as solvent provides ideal polarity characteristics that facilitate intermediate stabilization without promoting unwanted dimerization or oligomerization reactions. Furthermore, the inherent selectivity of the palladium catalyst system ensures exclusive formation of the desired regioisomer at the two-position of the imidazole ring through precise coordination control during the cyclization step. This multi-layered approach to impurity management consistently delivers products meeting stringent pharmaceutical purity specifications without requiring additional purification steps beyond standard column chromatography.
How to Synthesize High-Purity Trifluoromethyl Imidazoles Efficiently
This patented methodology represents a significant advancement in heterocyclic synthesis technology that enables reliable production of high-purity trifluoromethyl imidazoles through a streamlined process requiring minimal specialized equipment or expertise. The procedure leverages commercially available starting materials and standard laboratory apparatus while maintaining exceptional reproducibility across different production scales. Detailed standardized synthesis steps are provided below to ensure consistent implementation across various manufacturing environments while maintaining optimal product quality and yield characteristics.
- Prepare the reaction mixture by combining palladium chloride catalyst (5 mol%), triphenylphosphine ligand (10 mol%), sodium bicarbonate additive (2.0 equiv), formic acid/acetic anhydride as CO source (10 equiv), trifluoroethylimidoyl chloride substrate (II), propargylamine (III), and diaryl iodonium salt (IV) in tetrahydrofuran solvent under inert atmosphere.
- Stir the homogeneous mixture at precisely controlled temperature of 30°C for exactly twenty hours to ensure complete conversion while maintaining optimal reaction kinetics and minimizing side product formation.
- Perform post-reaction processing by filtration through silica gel followed by column chromatography purification using standard elution protocols to isolate high-purity (>99%) target imidazole compounds with consistent yield profiles.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points faced by procurement and supply chain professionals through its inherently efficient design that eliminates multiple cost drivers while enhancing operational reliability across the entire production continuum. The methodology transforms traditional manufacturing challenges into strategic advantages by leveraging simple chemistry principles that translate into tangible business benefits without requiring significant capital investment or process re-engineering efforts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal procedures and hazardous reagent handling requirements delivers substantial cost savings through reduced waste treatment expenses and simplified facility requirements while maintaining consistent product quality across production scales from laboratory to commercial volumes.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials with multiple global suppliers ensures consistent raw material availability while the robust nature of the process minimizes batch failures and quality deviations that typically disrupt production schedules in traditional heterocycle manufacturing.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from small-scale development to multi-ton production maintains consistent yield profiles without requiring specialized equipment modifications while generating minimal waste streams that align with modern environmental regulations and sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented technology based on detailed analysis of the patent documentation and practical manufacturing considerations relevant to pharmaceutical intermediate production.
Q: How does this method overcome traditional synthesis challenges for trifluoromethyl imidazoles?
A: The patented process eliminates harsh reaction conditions through mild palladium-catalyzed carbonylation at ambient temperature (30°C), avoiding expensive transition metal removal steps while achieving superior substrate compatibility across diverse aryl groups without specialized equipment requirements.
Q: What supply chain advantages does this manufacturing approach provide?
A: The methodology ensures reliable supply through utilization of commercially available starting materials with simplified purification protocols that maintain consistent quality during scale-up from laboratory to industrial production volumes without complex infrastructure modifications.
Q: How does substrate design flexibility impact pharmaceutical intermediate production?
A: The modular system allows precise customization of R and Ar substituents through straightforward variations in precursor selection, enabling tailored synthesis of diverse trifluoromethyl imidazole derivatives with optimized biological activity profiles while maintaining high reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable High-Purity Trifluoromethyl Imidazole Compound Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically designed for complex heterocyclic compounds like trifluoromethyl imidazoles. This patented methodology represents just one example of our commitment to developing innovative solutions that address critical manufacturing challenges in pharmaceutical intermediate production while delivering consistent quality and reliability expected by global pharmaceutical partners.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who can provide specific COA data and route feasibility assessments tailored to your unique production requirements and quality standards.
