Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Scalable Pharmaceutical Manufacturing
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, profoundly influencing the pharmacokinetic profiles of drug candidates. As detailed in patent CN111423381B, a novel and highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds has been developed, addressing critical bottlenecks in the synthesis of these valuable nitrogen-containing five-membered heterocycles. Imidazole derivatives are ubiquitous in bioactive molecules, serving as key structural motifs in drugs such as Alcaftadine and complex natural products like Naamidine A, as well as functioning as essential ligands in coordination catalysis. 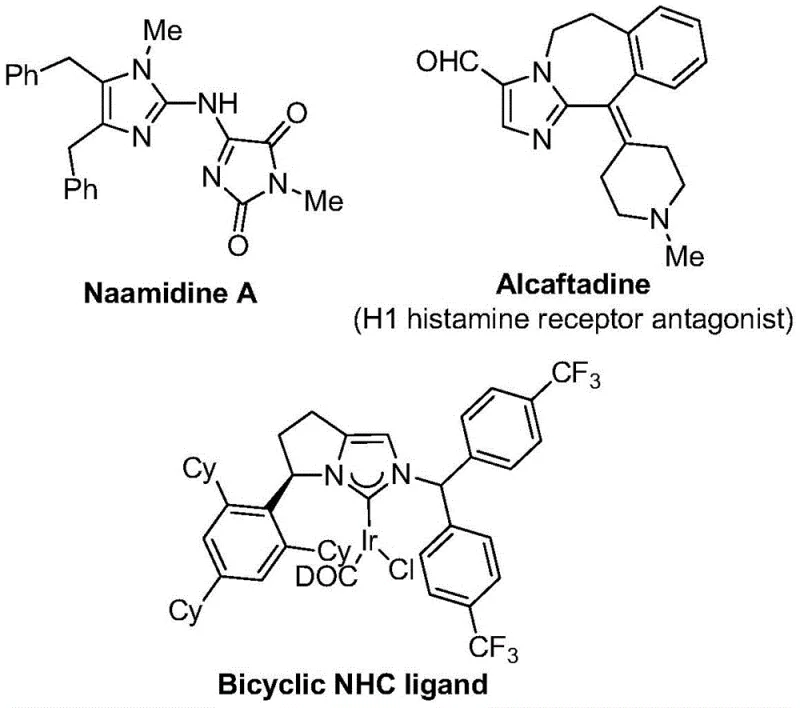 . The introduction of the trifluoromethyl moiety significantly enhances electronegativity, metabolic stability, lipophilicity, and bioavailability, making these compounds highly sought-after intermediates for the development of next-generation therapeutics and functional materials.
. The introduction of the trifluoromethyl moiety significantly enhances electronegativity, metabolic stability, lipophilicity, and bioavailability, making these compounds highly sought-after intermediates for the development of next-generation therapeutics and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the direct reaction of substrates with specialized trifluoromethyl synthons. Common reagents such as trifluorodiazoethane, while effective, pose significant safety hazards due to their explosive nature and require stringent handling protocols that complicate large-scale manufacturing. Furthermore, alternative methods often involve harsh reaction conditions, including elevated temperatures and strong bases, which can lead to poor functional group tolerance and the formation of complex impurity profiles. The limited availability and high cost of certain trifluoromethylating agents further restrict the practical utility of these conventional routes, creating supply chain vulnerabilities for pharmaceutical manufacturers seeking reliable sources of high-purity intermediates. These factors collectively hinder the rapid iteration and scale-up required in modern drug discovery pipelines.
The Novel Approach
The methodology disclosed in patent CN111423381B offers a transformative solution by employing a transition metal palladium-catalyzed carbonylation series reaction. This innovative route utilizes cheap and easily obtained starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, to efficiently construct the 2-trifluoromethyl imidazole core. 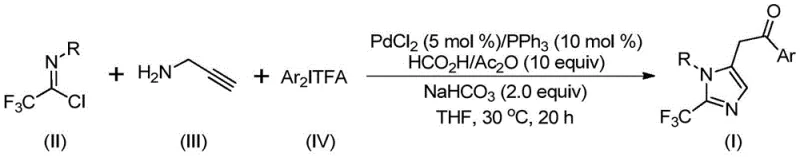 . By operating under remarkably mild conditions at 30°C, this process not only ensures high reaction efficiency but also exhibits exceptional substrate compatibility, allowing for the synthesis of diversely substituted imidazole compounds. The use of in situ generated carbon monoxide from formic acid and acetic anhydride eliminates the need for external CO gas, thereby streamlining the operational workflow and enhancing safety profiles for industrial applications.
. By operating under remarkably mild conditions at 30°C, this process not only ensures high reaction efficiency but also exhibits exceptional substrate compatibility, allowing for the synthesis of diversely substituted imidazole compounds. The use of in situ generated carbon monoxide from formic acid and acetic anhydride eliminates the need for external CO gas, thereby streamlining the operational workflow and enhancing safety profiles for industrial applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The reaction mechanism involves a sophisticated cascade of organometallic transformations initiated by the interaction of the palladium catalyst with the alkyne substrate. Initially, an intermolecular carbon-nitrogen bond is formed under alkaline promotion to generate a trifluoroacetamidine intermediate, which subsequently undergoes isomerization. The palladium catalyst then facilitates the palladation of the alkyne amine, yielding a crucial alkenyl palladium intermediate. This species undergoes further isomerization to form an alkyl palladium intermediate, setting the stage for the key carbonylation step. Under the influence of carbon monoxide released from the formic acid/acetic anhydride system, an acyl palladium intermediate is generated, effectively inserting the carbonyl group into the growing molecular framework.
Following carbonylation, the diaryl iodonium salt participates in an oxidative addition step, leading to the formation of a high-valent tetravalent palladium intermediate. This transient species is pivotal for the final construction of the heterocyclic ring. The catalytic cycle concludes with a reductive elimination step, which releases the final 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst for subsequent turnover. This intricate mechanism ensures high atom economy and minimizes the formation of side products, resulting in a clean reaction profile that simplifies downstream purification. The robustness of this catalytic cycle allows for the accommodation of various electronic and steric environments on the aryl rings, as evidenced by the successful synthesis of compounds with electron-donating and electron-withdrawing groups.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical execution of this synthesis is designed for operational simplicity and reproducibility, making it highly suitable for both laboratory optimization and commercial production. The process begins with the precise charging of reagents into a reaction vessel, typically a Schlenk tube for small-scale trials or a stirred tank reactor for larger batches. The reaction mixture, comprising the palladium catalyst system, base, CO source, and the three key organic substrates in an aprotic solvent like tetrahydrofuran (THF), is stirred at a controlled temperature. The mild thermal requirement of 30°C significantly reduces energy consumption compared to traditional high-temperature protocols. Detailed standardized synthetic steps for implementing this protocol are provided in the guide below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits centered around cost optimization and supply reliability. The shift away from hazardous and expensive reagents like trifluorodiazoethane to commodity chemicals such as propargylamine and aromatic amines drastically reduces raw material costs. Furthermore, the mild reaction conditions minimize energy expenditures and reduce the wear and tear on processing equipment, contributing to lower overall manufacturing overheads. The simplicity of the post-treatment procedure, involving basic filtration and standard column chromatography, ensures high throughput and reduces the time-to-market for critical intermediates.
- Cost Reduction in Manufacturing: The utilization of palladium chloride, a relatively inexpensive palladium source compared to other complexes, combined with common ligands like triphenylphosphine, optimizes catalyst costs. The elimination of specialized high-pressure equipment for carbon monoxide handling further reduces capital expenditure requirements. By leveraging widely available starting materials, the process mitigates the risk of price volatility associated with niche reagents, ensuring stable pricing structures for long-term supply contracts.
- Enhanced Supply Chain Reliability: The broad substrate scope of this methodology means that a single production line can be adapted to manufacture a wide variety of 2-trifluoromethyl imidazole derivatives by simply changing the aryl substituents on the starting materials. This flexibility allows suppliers to respond rapidly to changing demand patterns without significant retooling. The robustness of the reaction against functional group variations ensures consistent quality and yield, reducing the incidence of batch failures that can disrupt downstream API production schedules.
- Scalability and Environmental Compliance: The avoidance of explosive diazo compounds and toxic gas cylinders significantly improves the safety profile of the manufacturing process, facilitating easier regulatory approval for scale-up. The use of THF as a solvent, which is readily recyclable, aligns with green chemistry principles and simplifies waste management protocols. The high reaction efficiency and selectivity minimize the generation of hazardous waste streams, lowering disposal costs and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, aiming to clarify potential concerns for technical teams evaluating this route for adoption. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride and propargylamine, avoiding the use of hazardous trifluorodiazoethane. It operates under mild conditions (30°C) with high substrate compatibility and excellent yields.
Q: What is the role of formic acid and acetic anhydride in this reaction system?
A: Formic acid and acetic anhydride serve as an in situ carbon monoxide (CO) source. This eliminates the need for handling toxic CO gas cylinders directly, significantly enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: Can this synthesis method tolerate diverse functional groups on the aryl rings?
A: Yes, the protocol demonstrates broad substrate scope. It successfully accommodates various substituents including methyl, tert-butyl, methoxy, chloro, bromo, trifluoromethyl, and nitro groups at ortho, meta, or para positions without compromising reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl imidazole meets the exacting standards required by global regulatory bodies. Our commitment to excellence extends beyond mere compliance; we strive to be a strategic partner in your innovation journey.
We invite you to engage with our technical procurement team to discuss how this advanced palladium-catalyzed technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique project needs, ensuring a secure and efficient supply of these vital pharmaceutical building blocks.
