Advanced Synthesis of Sugammadex Sodium Intramolecular Thioether Impurity for High-Purity QC Standards
The pharmaceutical industry's relentless pursuit of purity and safety has placed unprecedented scrutiny on the impurity profiles of complex active pharmaceutical ingredients (APIs). In the realm of neuromuscular blockade reversal agents, Sugammadex Sodium stands as a cornerstone therapy, yet its complex cyclodextrin structure presents unique analytical challenges. Patent CN111320712B introduces a groundbreaking synthetic methodology specifically designed to produce the intramolecular thioether impurity of Sugammadex Sodium, a critical reference standard essential for rigorous quality control. This innovation addresses the historical difficulties in obtaining authentic impurity samples, which are requisite for validating self-contrast analytical methods used in regulatory submissions. By leveraging a streamlined two-step process starting from octa-(6-bromo-6-deoxy)-gamma-cyclodextrin, this technology enables the precise construction of the thioether bridge that characterizes this specific degradation product. For R&D directors and quality assurance teams, access to such well-characterized impurities is not merely a regulatory checkbox but a fundamental requirement for ensuring the safety and efficacy of the final drug product. The following analysis dissects the technical merits of this patent, highlighting its potential to redefine supply chain reliability for high-purity pharmaceutical intermediates.
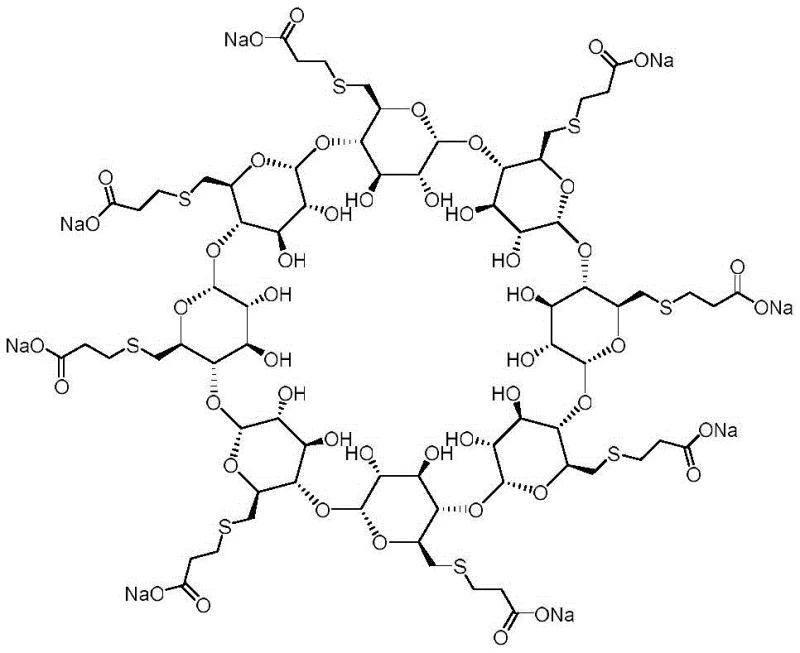
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of specific cyclodextrin-derived impurities has been plagued by low selectivity and arduous purification protocols. Conventional routes often rely on non-specific oxidation or random thioetherification processes that generate a chaotic mixture of regioisomers, making the isolation of the specific intramolecular thioether variant exceptionally difficult. These legacy methods frequently require harsh reaction conditions, including extreme temperatures or aggressive oxidizing agents, which can degrade the sensitive glycosidic bonds of the cyclodextrin backbone. Furthermore, the lack of stereochemical control in older methodologies often results in poor yields, necessitating large-scale reactions to obtain mere milligrams of the target reference standard. This inefficiency translates directly into exorbitant costs and extended lead times for pharmaceutical manufacturers who require these standards for batch release testing. The reliance on such unpredictable chemistry creates a bottleneck in the quality control workflow, potentially delaying the market entry of generic versions or new formulations of Sugammadex.
The Novel Approach
In stark contrast, the methodology disclosed in CN111320712B employs a highly controlled nucleophilic substitution strategy that ensures the precise formation of the intramolecular sulfur bridge. By utilizing octa-(6-bromo-6-deoxy)-gamma-cyclodextrin as a pre-functionalized scaffold, the process eliminates the need for random functionalization, thereby drastically improving regioselectivity. The introduction of a sulfide source under mild conditions allows for the targeted bridging of specific glucose units within the macrocycle, setting the stage for the subsequent attachment of the propionic acid side chains. This approach not only simplifies the synthetic route but also enhances the overall atom economy of the reaction. The use of polar aprotic solvents facilitates the solubility of the bulky cyclodextrin intermediate while maintaining the reactivity of the nucleophiles. Consequently, this novel pathway delivers the target impurity with superior purity profiles, minimizing the burden on downstream purification steps and ensuring a consistent supply of high-quality reference materials for the global pharmaceutical market.
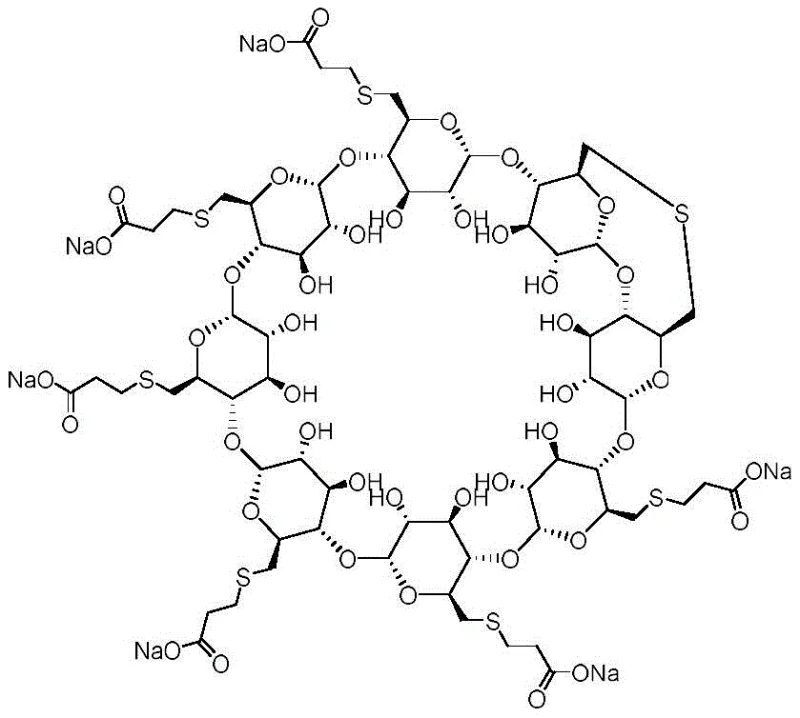
Mechanistic Insights into Sulfide-Mediated Cyclodextrin Functionalization
The core of this synthetic breakthrough lies in the mechanistic elegance of the sulfide-mediated bridging reaction. In the first step, the sulfide ion acts as a potent nucleophile, attacking the electrophilic carbon at the 6-position of the bromo-cyclodextrin. Unlike intermolecular reactions that lead to polymerization or cross-linking, the specific stoichiometry and solvent environment described in the patent favor an intramolecular displacement. This results in the formation of a stable thioether linkage between adjacent or proximal glucose units, effectively locking the conformation of the macrocycle. The reaction temperature, maintained between -5°C and 45°C, is critical; it provides sufficient thermal energy to overcome the activation barrier for the SN2 substitution without promoting elimination side reactions that could compromise the integrity of the sugar ring. The choice of polar solvents such as DMSO or DMF is equally pivotal, as they stabilize the transition state and solvate the sodium cations, thereby increasing the nucleophilicity of the sulfide anion. This precise control over the reaction microenvironment is what allows for the selective generation of the intramolecular impurity structure rather than a statistical distribution of byproducts.
Following the bridge formation, the second step involves the conversion of remaining reactive sites or the completion of the side chain functionality using 3-mercaptopropionic acid. This thioetherification proceeds via a classic Michael-type addition or nucleophilic substitution mechanism, depending on the specific activation state of the intermediate. The presence of sodium hydroxide serves to deprotonate the thiol group of the mercaptopropionic acid, generating a highly reactive thiolate anion. This species then attacks the remaining electrophilic centers on the cyclodextrin scaffold. The careful control of pH and temperature in this stage ensures that the newly formed thioether bridges from the first step remain intact while the desired side chains are installed. This dual-stage mechanism effectively decouples the complex cyclization event from the side-chain functionalization, allowing each step to be optimized independently. For process chemists, this modularity offers significant advantages in troubleshooting and scaling, as parameters can be adjusted for each distinct transformation without affecting the other.
How to Synthesize Sugammadex Sodium Intramolecular Thioether Impurity Efficiently
The practical execution of this synthesis requires strict adherence to the patented parameters to ensure reproducibility and high yield. The process begins with the dissolution of the bromo-cyclodextrin starting material in a polar solvent, followed by the controlled addition of the sulfide source. Maintaining the reaction within the specified temperature window is crucial for maximizing the formation of the bridged intermediate. Once the intermediate is isolated through crystallization and purification, it is subjected to the second reaction with 3-mercaptopropionic acid in an aqueous alkaline medium. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined below to guide laboratory implementation.
- React octa-(6-bromo-6-deoxy)-gamma-cyclodextrin with a sulfide source in a polar solvent to form a bridged intermediate.
- Purify the intermediate via crystallization using 2-butanone and water, followed by vacuum drying.
- React the purified intermediate with 3-mercaptopropionic acid in aqueous sodium hydroxide to yield the final intramolecular thioether impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The simplification of the synthetic pathway directly correlates with reduced operational complexity, which is a key driver for cost containment in the manufacturing of high-value reference standards. By eliminating the need for exotic reagents or multi-step protection-deprotection sequences, the process lowers the barrier to entry for production, thereby enhancing supply security. Furthermore, the mild reaction conditions imply lower energy consumption and reduced wear on processing equipment, contributing to a more sustainable and cost-effective manufacturing footprint. The high yield reported in the patent suggests a more efficient utilization of raw materials, which is particularly important given the specialized nature of cyclodextrin derivatives. These factors collectively position this technology as a robust solution for securing a stable supply of critical quality control materials.
- Cost Reduction in Manufacturing: The streamlined two-step process significantly reduces the number of unit operations required compared to traditional multi-step syntheses. By avoiding the use of expensive transition metal catalysts or hazardous oxidizing agents, the method minimizes both raw material costs and the expenses associated with waste disposal and environmental compliance. The ability to isolate the intermediate through simple crystallization rather than complex chromatography further drives down processing costs. This efficiency allows suppliers to offer competitive pricing for high-purity impurity standards without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as octa-(6-bromo-6-deoxy)-gamma-cyclodextrin and common industrial solvents, mitigates the risk of supply disruptions. Unlike processes dependent on custom-synthesized precursors with long lead times, this route utilizes commodity chemicals that are readily accessible from multiple global vendors. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in utility supplies or environmental conditions, ensuring consistent batch-to-batch production. This reliability is paramount for pharmaceutical companies that require uninterrupted access to reference standards for ongoing quality assurance programs.
- Scalability and Environmental Compliance: The use of water and polar organic solvents that are easily recoverable aligns well with modern green chemistry principles. The absence of heavy metals or persistent organic pollutants in the waste stream simplifies the effluent treatment process, reducing the environmental liability for manufacturers. Moreover, the scalability of the crystallization steps ensures that the process can be seamlessly transferred from gram-scale laboratory synthesis to kilogram-scale commercial production. This scalability ensures that the supply of these critical impurities can grow in tandem with the increasing global demand for Sugammadex Sodium, preventing bottlenecks in the broader pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Sugammadex Sodium impurities. These insights are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity for stakeholders evaluating this technology for their quality control workflows.
Q: What is the primary advantage of this synthesis method for Sugammadex impurities?
A: The method described in patent CN111320712B offers a significantly simplified operational process with mild reaction conditions, resulting in higher yields compared to traditional multi-step derivatization routes.
Q: Why is the intramolecular thioether impurity critical for Sugammadex manufacturing?
A: This specific impurity serves as a vital reference standard for self-contrast analytical methods, ensuring the qualitative and quantitative accuracy required for regulatory compliance and patient safety.
Q: Can this process be scaled for commercial production of reference materials?
A: Yes, the use of readily available starting materials like octa-(6-bromo-6-deoxy)-gamma-cyclodextrin and common polar solvents facilitates straightforward scale-up from laboratory to pilot plant levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Impurity Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your analytical data depends on the quality of your reference standards. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex molecules like cyclodextrin derivatives are manufactured with precision. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the identity and purity of every batch, guaranteeing that our Sugammadex Sodium intramolecular thioether impurity meets the highest global regulatory standards. Our commitment to technical excellence ensures that you receive materials that are fully characterized and ready for immediate use in your validation protocols.
We invite you to collaborate with us to optimize your supply chain for critical pharmaceutical intermediates. By leveraging our advanced synthesis capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your quality control operations are supported by the most reliable and cost-effective sources in the industry.
