Advanced Synthesis of Sugammadex Sodium Intramolecular Thioether Impurities for Global Pharma Supply Chains
Advanced Synthesis of Sugammadex Sodium Intramolecular Thioether Impurities for Global Pharma Supply Chains
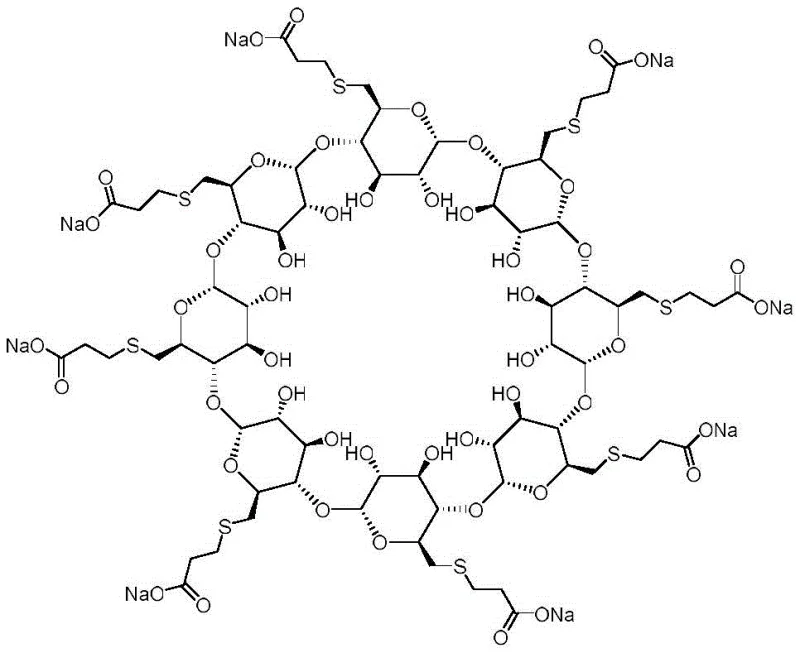
The pharmaceutical industry continuously demands higher purity standards for complex active pharmaceutical ingredients (APIs), particularly for critical care drugs like Sugammadex Sodium. Patent CN111320712A introduces a robust and efficient synthetic methodology specifically designed for producing intramolecular thioether impurities of Sugammadex Sodium. This innovation addresses a significant gap in quality control by providing a reliable route to generate authentic reference standards, which are indispensable for accurate impurity profiling. The ability to synthesize these specific structural variants allows manufacturers to move beyond approximate self-control methods, thereby enhancing the safety and efficacy verification of the final drug product. By establishing a clear pathway to these challenging molecules, the technology supports stricter regulatory compliance and more rigorous batch release testing protocols globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the analysis of impurities in Sugammadex Sodium has relied heavily on self-control methods, which often suffer from significant limitations in both qualitative and quantitative accuracy. Without access to authentic impurity standards, analytical chemists are forced to estimate impurity levels based on relative response factors that may not accurately reflect the true concentration of specific degradation products or synthetic by-products. This lack of precision poses a risk to patient safety, as unidentified or misquantified impurities could potentially exhibit toxicity or alter the pharmacokinetic profile of the medication. Furthermore, conventional synthesis routes for cyclodextrin derivatives often involve harsh conditions that can lead to complex mixtures, making the isolation of a single, pure intramolecular thioether species extremely difficult and cost-prohibitive for routine quality assurance laboratories.
The Novel Approach
The patented methodology offers a transformative solution by utilizing octa-(6-bromo-6-deoxy)-gamma-cyclodextrin as a strategic starting material, enabling a controlled and selective functionalization process. This novel approach leverages a two-step sequence that avoids the chaotic reaction profiles typical of direct modification on fully substituted cyclodextrins. By first introducing a sulfide bridge under controlled conditions and subsequently conjugating with 3-mercaptopropionic acid, the process ensures high regioselectivity and yield. The use of mild reaction temperatures and common polar solvents simplifies the operational complexity, allowing for easier scale-up and purification. This streamlined workflow not only improves the availability of high-purity reference materials but also significantly reduces the time and resources required for method validation in pharmaceutical quality control departments.
Mechanistic Insights into Nucleophilic Substitution and Thioether Formation
The core of this synthetic strategy relies on a precise nucleophilic substitution mechanism facilitated by the unique reactivity of the cyclodextrin scaffold. In the initial step, the bromine atoms at the 6-position of the gamma-cyclodextrin serve as excellent leaving groups, susceptible to attack by sulfide ions generated from sources like sodium sulfide or potassium sulfide. This reaction is conducted in polar aprotic solvents such as DMSO, DMF, or NMP, which stabilize the transition state and enhance the nucleophilicity of the sulfide species. The temperature is carefully maintained between -5°C and 45°C to prevent over-reaction or degradation of the macrocyclic ring, ensuring the formation of the desired intermediate with minimal side products. This controlled environment is critical for preserving the structural integrity of the cyclodextrin cavity while introducing the necessary sulfur functionality for subsequent modifications.
Following the formation of the intermediate, the second stage involves a conjugate addition or substitution reaction with 3-mercaptopropionic acid under alkaline conditions. The presence of sodium hydroxide deprotonates the thiol group of the mercaptopropionic acid, generating a highly reactive thiolate anion that attacks the electrophilic centers on the intermediate. This step is performed at temperatures ranging from 25°C to 80°C, providing sufficient thermal energy to drive the reaction to completion without compromising the stability of the newly formed thioether linkages. The stoichiometry is carefully optimized, with a molar ratio of intermediate to 3-mercaptopropionic acid between 1:6 and 1:30, ensuring that the reaction proceeds to full conversion. The final purification involves crystallization techniques using 2-butanone and water, which effectively separate the target impurity from unreacted starting materials and inorganic salts, yielding a product suitable for analytical certification.
How to Synthesize Sugammadex Sodium Intramolecular Thioether Impurity Efficiently
The synthesis of this critical reference standard is designed to be operationally simple yet chemically robust, making it accessible for both research and production environments. The process begins with the dissolution of the bromo-cyclodextrin precursor in a polar solvent, followed by the controlled addition of the sulfide reagent. After the intermediate is isolated and purified, it is reacted with 3-mercaptopropionic acid in an aqueous base. The detailed standardized synthesis steps, including specific work-up procedures and purification parameters, are outlined in the guide below to ensure reproducibility and high purity.
- React octa-(6-bromo-6-deoxy)-gamma-cyclodextrin with a sulfide source (such as sodium sulfide) in a polar solvent like DMF or DMSO at temperatures between -5°C and 45°C to form the intermediate.
- Dissolve the intermediate in a polar solvent and react with 3-mercaptopropionic acid in an aqueous sodium hydroxide solution at temperatures ranging from 25°C to 80°C.
- Purify the final product through crystallization using solvents such as 2-butanone and water, followed by filtration, washing, and vacuum drying to obtain the high-purity impurity standard.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents substantial opportunities for optimizing the sourcing of critical quality control materials. The reliance on readily available and cost-effective raw materials, such as octa-(6-bromo-6-deoxy)-gamma-cyclodextrin and commodity chemicals like sodium sulfide and 3-mercaptopropionic acid, drastically reduces the dependency on exotic or proprietary reagents. This accessibility translates directly into enhanced supply chain resilience, as the risk of raw material shortages is minimized. Furthermore, the simplicity of the reaction conditions eliminates the need for specialized high-pressure reactors or cryogenic cooling systems, allowing for production in standard multipurpose facilities. This flexibility enables suppliers to respond more rapidly to fluctuating market demands for reference standards, ensuring continuous availability for pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The streamlined nature of this two-step process significantly lowers the overall cost of goods sold by minimizing unit operations and reducing solvent consumption. By avoiding complex chromatographic separations and utilizing straightforward crystallization for purification, the method reduces both labor and material costs associated with downstream processing. The high yield of the reaction further contributes to economic efficiency, maximizing the output from each batch of starting material. Additionally, the elimination of expensive transition metal catalysts removes the need for costly metal scavenging steps, resulting in a cleaner and more economical production profile that aligns with lean manufacturing principles.
- Enhanced Supply Chain Reliability: The use of stable and commercially abundant starting materials ensures a consistent and reliable supply of the final impurity standard. Unlike processes that rely on unstable intermediates or custom-synthesized reagents with long lead times, this method utilizes off-the-shelf chemicals that can be sourced from multiple global vendors. This diversification of the supply base mitigates the risk of disruption due to geopolitical issues or single-source failures. The robustness of the synthetic route also means that production can be easily scaled up or down to match demand without significant re-engineering of the process, providing procurement teams with greater flexibility in inventory management.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this synthesis offers distinct advantages due to its mild reaction conditions and reduced waste generation. The absence of harsh reagents and the use of recyclable polar solvents contribute to a lower environmental footprint, facilitating easier compliance with increasingly stringent green chemistry regulations. The process generates minimal hazardous waste, simplifying disposal and reducing associated costs. Moreover, the scalability of the crystallization and filtration steps ensures that the process can be seamlessly transferred from laboratory scale to multi-kilogram production, supporting the growing global demand for high-quality pharmaceutical reference materials without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Sugammadex Sodium impurities. These insights are derived directly from the patented technology and are intended to assist technical teams in evaluating the feasibility and benefits of this new supply source. Understanding these details is crucial for integrating these reference standards into your existing quality control frameworks.
Q: Why is the synthesis of intramolecular thioether impurities critical for Sugammadex Sodium production?
A: Accurate quantification of impurities is essential for regulatory compliance and patient safety. Traditional self-control methods often lack the precision required for definitive identification. Having a synthesized reference standard allows for precise calibration of analytical instruments, ensuring that the final API meets stringent pharmacopoeial specifications regarding related substances.
Q: What are the key reaction conditions for this synthesis method?
A: The process utilizes mild reaction conditions to ensure stability and high yield. The first step operates effectively between -5°C and 45°C using polar aprotic solvents like DMF or DMSO. The second conjugation step proceeds efficiently at temperatures between 25°C and 80°C in an alkaline aqueous environment, minimizing degradation of the sensitive cyclodextrin scaffold.
Q: Is this synthetic route suitable for large-scale manufacturing of reference standards?
A: Yes, the methodology is designed for scalability. It employs readily available starting materials such as octa-(6-bromo-6-deoxy)-gamma-cyclodextrin and 3-mercaptopropionic acid. The work-up procedures involve standard unit operations like crystallization and filtration, which are easily transferable from laboratory to pilot and commercial scales without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity reference standards play in ensuring the safety and efficacy of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical market. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to replicate complex synthetic routes like the one described in CN111320712A demonstrates our technical prowess and dedication to quality.
We invite you to collaborate with us to optimize your supply chain for critical impurity standards. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and quality assurance objectives effectively.
