Scalable Synthesis of Roflumilast Key Intermediate via Novel Alkylation and Hydroxylation Strategies
The pharmaceutical industry continuously seeks robust manufacturing pathways for Chronic Obstructive Pulmonary Disease (COPD) therapeutics, specifically for the PDE4 inhibitor Roflumilast. Patent CN102633631B introduces a transformative synthetic methodology for producing 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid, a critical building block in this drug's value chain. This invention addresses long-standing inefficiencies in prior art by establishing a linear, high-yield sequence that begins with readily available 3-hydroxy-4-halo-benzoates. By strategically reversing the traditional order of functional group installation, the process achieves exceptional regioselectivity during the alkylation phase, effectively eliminating the formation of difficult-to-separate isomers that have historically plagued production lines. The technical breakthrough lies not merely in the chemical transformations themselves, but in the holistic design of the route which prioritizes operational simplicity and purification ease, ensuring that the final API intermediate meets stringent quality standards without resorting to costly chromatographic techniques.
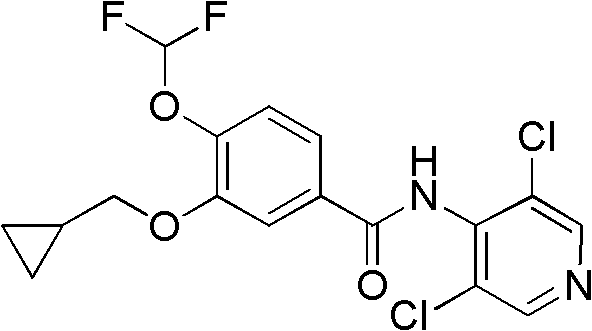
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate has been fraught with significant technical hurdles that impede efficient commercial manufacturing. For instance, the route disclosed in WO2005026095 initiates from 3,4-dihydroxybenzoic acid methyl ester, attempting direct cyclopropylmethylation. This approach suffers from inherent regioselectivity issues, where alkylation occurs indiscriminately at both the 3- and 4-positions, generating a complex mixture of mono-alkylated isomers and double-alkylated by-products. Separating these structurally similar impurities necessitates column chromatography, a technique that is economically prohibitive and operationally cumbersome on a multi-ton scale. Similarly, other reported methods, such as those utilizing catechol precursors, often require hazardous reagents like carbon monoxide for carbonylation steps or demand cryogenic conditions around -60°C, introducing severe safety risks and energy costs that are unsustainable for modern green chemistry initiatives.
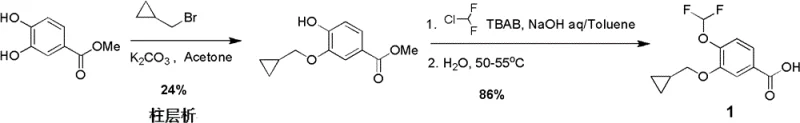
The Novel Approach
In stark contrast, the methodology outlined in CN102633631B leverages a 3-hydroxy-4-halo-benzoate starting material to enforce strict regiocontrol from the outset. By utilizing the steric and electronic directing effects of the halogen atom at the 4-position, the initial alkylation with cyclopropylmethyl bromide proceeds exclusively at the 3-hydroxyl group, achieving yields as high as 99% without detectable isomeric impurities. This strategic inversion of the synthetic logic removes the primary bottleneck of purification. Furthermore, the subsequent introduction of the difluoromethoxy group is performed on a protected ester intermediate under mild basic conditions, avoiding the harsh environments associated with earlier patents. The entire sequence relies on standard unit operations such as filtration, extraction, and recrystallization, making it inherently scalable and perfectly aligned with the requirements of a reliable pharmaceutical intermediate supplier aiming for cost-effective mass production.
Mechanistic Insights into Copper-Catalyzed Hydroxylation and Selective Alkylation
The core of this synthetic innovation rests on two pivotal mechanistic events: the highly selective nucleophilic substitution during alkylation and the transition-metal catalyzed hydroxylation. In the first step, the phenoxide anion generated from the 3-hydroxy-4-halo-benzoate acts as a potent nucleophile. The presence of the halogen (iodine or bromine) at the para-position deactivates the ring towards electrophilic aromatic substitution but facilitates the SN2 attack on the cyclopropylmethyl halide. The reaction is typically conducted in polar aprotic solvents like DMF or DMSO at temperatures between 60°C and 90°C, ensuring complete conversion while maintaining the integrity of the sensitive cyclopropyl ring. This step is crucial because it sets the stereochemical and regiochemical foundation for the entire molecule, preventing the formation of the 4-cyclopropylmethoxy isomer which would be a critical quality attribute failure in the final drug substance.
Following alkylation, the replacement of the halogen with a hydroxyl group represents a sophisticated application of Ullmann-type or Buchwald-Hartwig coupling chemistry. The patent specifies the use of Cuprous Iodide (CuI) or Palladium catalysts such as Pd(dba)2 in conjunction with bidentate ligands like 1,10-phenanthroline or 8-hydroxyquinoline. Mechanistically, the metal center undergoes oxidative addition into the carbon-halogen bond, followed by coordination with the hydroxide source (typically aqueous KOH or NaOH). The subsequent reductive elimination releases the phenolic product and regenerates the active catalyst species. This catalytic cycle is optimized to proceed at temperatures ranging from 80°C to 140°C, delivering yields exceeding 95%. The choice of ligand is paramount here, as it stabilizes the metal complex and accelerates the turnover frequency, thereby minimizing residual metal impurities in the final product, a key concern for regulatory compliance in API manufacturing.
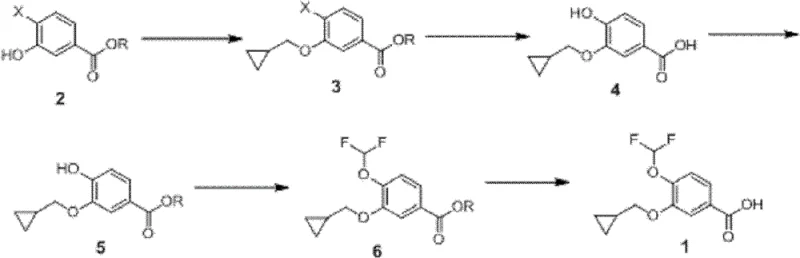
How to Synthesize 3-Cyclopropylmethoxy-4-Difluoromethoxybenzoic Acid Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize the economic and technical benefits described in the patent. The process flow is designed to minimize intermediate isolation steps where possible, although the patent exemplifies isolating the stable ester intermediates to ensure purity. Operators should focus on maintaining strict stoichiometric ratios, particularly during the difluoromethylation step where the ratio of base to difluoromethylating agent dictates the completeness of the reaction. The detailed standardized operating procedures, including specific solvent volumes, addition rates, and workup protocols necessary to replicate the high yields reported in the examples, are essential for technology transfer.
- Alkylate 3-hydroxy-4-halo-benzoate with cyclopropylmethyl bromide in DMF using potassium carbonate at 70°C to yield the cyclopropylmethoxy intermediate.
- Perform catalytic hydroxylation using Cuprous Iodide (CuI) or Palladium catalysts with ligands like 8-hydroxyquinoline to replace the halogen with a hydroxyl group.
- Esterify the resulting hydroxybenzoic acid using thionyl chloride in methanol to protect the carboxylic acid group for subsequent reactions.
- Conduct difluoromethylation using sodium chlorodifluoroacetate or chlorodifluoromethane gas under basic conditions to install the difluoromethoxy moiety.
- Hydrolyze the final ester intermediate using aqueous sodium hydroxide or potassium hydroxide to obtain the target 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented route translates directly into enhanced supply security and reduced total cost of ownership. The elimination of column chromatography is perhaps the most significant economic driver; chromatographic purification is not only expensive in terms of silica gel and solvent consumption but also creates a major throughput bottleneck that limits batch size and extends production cycles. By replacing this with crystallization and extraction, the manufacturing capacity can be significantly increased without additional capital expenditure on specialized equipment. Furthermore, the high yields reported in each step—often exceeding 95%—mean that less raw material is wasted, directly improving the atom economy and reducing the environmental footprint associated with waste disposal, which is increasingly critical for maintaining environmental compliance in chemical manufacturing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chromatographic purification media and the associated large volumes of elution solvents, which drastically lowers the variable cost per kilogram of the intermediate. Additionally, the use of commercially available starting materials like 3-hydroxy-4-iodobenzoic acid derivatives ensures that raw material sourcing is stable and not subject to the volatility of custom-synthesized precursors. The high convergence of the synthesis means fewer overall processing hours are required per batch, optimizing labor and utility costs while maximizing the utilization of existing reactor trains.
- Enhanced Supply Chain Reliability: By avoiding cryogenic conditions and toxic gases like carbon monoxide, the process reduces the risk of unplanned shutdowns due to safety incidents or equipment failures associated with extreme operating conditions. The robustness of the reaction conditions, which tolerate standard industrial heating and stirring capabilities, ensures that production can be maintained consistently across different manufacturing sites. This reliability is crucial for securing long-term contracts with downstream API manufacturers who require guaranteed delivery schedules to meet their own regulatory filing timelines.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, utilizing aqueous workups and recyclable solvents where possible. The absence of heavy metal contamination risks, thanks to efficient catalyst removal protocols, simplifies the wastewater treatment process. This makes the technology highly scalable from pilot plant quantities to multi-ton commercial production without the need for complex engineering controls, facilitating a smoother path to market for generic versions of Roflumilast and ensuring a steady supply of high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. Understanding these details is vital for quality assurance teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and claims within the patent specification, providing a factual basis for process assessment.
Q: How does this new method improve purification compared to WO2005026095?
A: Unlike the prior art which suffers from poor regioselectivity requiring column chromatography, this method utilizes selective alkylation of a 4-halo precursor, allowing purification via simple extraction and crystallization.
Q: What catalysts are used for the hydroxylation step?
A: The process employs transition metal catalysts such as Cuprous Iodide (CuI) or Palladium complexes like Pd(dba)2, paired with ligands like 1,10-phenanthroline or 8-hydroxyquinoline to ensure high conversion rates.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method avoids toxic reagents like carbon monoxide and extreme low temperatures, utilizing robust conditions and high-yield steps that are ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyclopropylmethoxy-4-Difluoromethoxybenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex respiratory drugs depends on the availability of high-quality key intermediates. Our technical team has thoroughly analyzed the pathway described in CN102633631B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are equipped with rigorous QC labs and adhere to stringent purity specifications to ensure that every batch of 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence ensures that potential impurities, such as regioisomers or residual halogens, are controlled well below threshold limits.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for COPD therapeutics is built on a foundation of scientific innovation and operational reliability.
