Advanced Copper-Catalyzed Synthesis of Substituted Diphenylamines for High-Purity Electronic Materials
Advanced Copper-Catalyzed Synthesis of Substituted Diphenylamines for High-Purity Electronic Materials
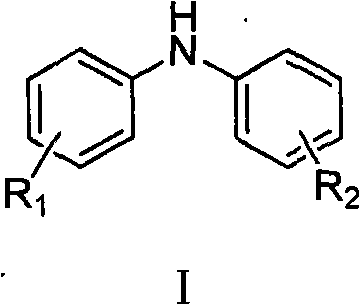
The landscape of organic synthesis for electronic materials and fine chemicals is constantly evolving, driven by the need for more efficient, cost-effective, and scalable processes. A pivotal advancement in this domain is documented in patent CN101570490A, which introduces a robust method for synthesizing substituted diphenylamines using copper catalysis. This technology addresses critical bottlenecks in the production of hole transport materials and specialized dyes, offering a pathway that balances high yield with operational simplicity. Substituted diphenylamines are not merely commodity chemicals; they are the backbone of advanced optoelectronic devices, serving as precursors for triphenylamine derivatives used in OLED displays. The ability to produce these intermediates with high purity and structural precision is paramount for downstream performance. This patent outlines a methodology that replaces harsh thermal conditions and expensive noble metal catalysts with a accessible copper-based system, marking a significant shift towards sustainable and economically viable manufacturing practices for complex aromatic amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diphenylamine derivatives has been plagued by severe operational constraints and economic inefficiencies. Traditional gas-phase methods, such as those described in older literature, often require extreme temperatures exceeding 450°C over alumina catalysts. These conditions are not only energy-intensive but also limit the scope of substrates, typically yielding only symmetrical products while generating hazardous ammonia by-products that complicate separation and purification. Furthermore, alternative solution-phase methods relying on palladium-catalyzed C-N coupling, while effective for laboratory-scale synthesis, present formidable challenges for industrial scale-up. The reliance on specialized palladium complexes, which are sensitive to air and moisture, necessitates expensive inert atmosphere equipment and rigorous exclusion of oxygen, driving up capital and operational expenditures. Additionally, methods utilizing phenylboronic acids introduce another layer of cost due to the high price of boronic acid reagents and the stoichiometric amounts of copper salts required, rendering them unsuitable for large-volume commercial production where margin compression is a constant threat.
The Novel Approach
The methodology presented in patent CN101570490A offers a transformative solution by leveraging a copper-catalyzed Ullmann-type coupling reaction under significantly milder conditions. This novel approach utilizes readily available aryl iodides and N-acyl aniline derivatives as starting materials, reacting them in common organic solvents like toluene or xylene. The core innovation lies in the specific combination of a catalytic amount of copper compounds, such as cuprous iodide or cuprous chloride, paired with inexpensive nitrogen-containing ligands like 1,10-phenanthroline or cyclohexanediamine. This catalytic system facilitates the formation of the carbon-nitrogen bond at temperatures ranging from 110°C to 160°C, which is drastically lower than traditional thermal methods and compatible with standard industrial reactors. By avoiding the use of precious metals and eliminating the need for strictly anhydrous conditions, this process drastically simplifies the workflow, reduces raw material costs, and enhances the safety profile of the manufacturing operation, making it an ideal candidate for the commercial scale-up of complex electronic chemical intermediates.
Mechanistic Insights into Copper-Catalyzed C-N Coupling
The success of this synthetic route hinges on the intricate interplay between the copper catalyst and the nitrogen ligand, which together facilitate a catalytic cycle capable of activating the aryl iodide bond. In this mechanism, the copper species, initially in the +1 oxidation state, undergoes oxidative addition with the aryl iodide to form a transient copper(III) intermediate. The presence of the bidentate nitrogen ligand is critical here, as it stabilizes the copper center, preventing aggregation into inactive metallic copper and maintaining the catalyst's solubility and activity throughout the reaction duration. The N-acyl aniline then coordinates to the copper center, followed by reductive elimination to forge the new C-N bond, regenerating the active copper(I) species. This cycle allows for the efficient conversion of starting materials into the protected diphenylamine intermediate with high turnover numbers. The choice of base, such as potassium tert-butoxide or cesium carbonate, plays a dual role in neutralizing the hydrogen iodide by-product and deprotonating the amine nitrogen to facilitate nucleophilic attack, ensuring the reaction proceeds to completion without stalling due to acid accumulation.
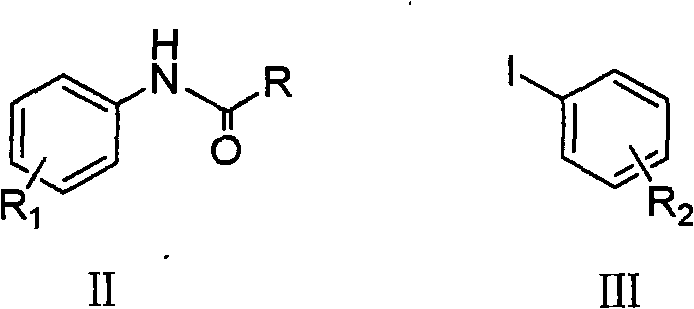
Following the coupling reaction, the process incorporates a strategic hydrolysis step that is vital for impurity control and final product specification. The initial coupling yields an N-acyl protected diphenylamine, which serves to moderate the nucleophilicity of the amine during the coupling phase, thereby minimizing side reactions such as poly-arylation. The subsequent hydrolysis, performed using either acidic conditions like hydrochloric acid or basic conditions like sodium hydroxide, cleanly cleaves the amide bond to reveal the free secondary amine. This two-step sequence—coupling followed by deprotection—provides a powerful handle for purity management. It ensures that any unreacted starting materials or over-arylated by-products can be effectively separated or converted, resulting in a final product with a clean impurity profile. For R&D directors focused on the quality of hole transport materials, this level of control over the molecular architecture is essential, as trace impurities can severely degrade the efficiency and lifespan of OLED devices.
How to Synthesize Substituted Diphenylamine Efficiently
The practical implementation of this copper-catalyzed protocol involves a straightforward sequence of mixing, heating, and workup operations that are well-suited for standard chemical processing equipment. The process begins with the dissolution of the N-acyl aniline derivative and the aryl iodide in a solvent such as toluene or xylene, followed by the sequential addition of the base, copper catalyst, and ligand. The reaction mixture is then heated to reflux or maintained at a controlled temperature between 110°C and 160°C for a period typically around 10 hours, allowing the coupling to reach high conversion. Upon completion, the reaction is cooled, and the crude product is isolated through aqueous washing and solvent recovery. The final deprotection step involves treating the residue with acid or base under reflux conditions, followed by neutralization and extraction to yield the pure substituted diphenylamine. Detailed standardized synthetic steps see the guide below.
- Combine N-acyl aniline derivative (Formula II) and aryl iodide (Formula III) in an organic solvent such as toluene or xylene.
- Add a catalytic amount of copper compound (e.g., CuI, CuCl), a nitrogen-containing ligand (e.g., 1,10-phenanthroline), and a base (e.g., K3PO4, t-BuOK).
- Heat the mixture to 110°C-160°C for coupling, followed by acid or base hydrolysis to remove the acyl protecting group and isolate the target diphenylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101570490A represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with critical electronic chemical intermediates. The shift from noble metal catalysis to base metal catalysis fundamentally alters the cost equation, removing the volatility associated with palladium prices and the logistical burden of recovering expensive catalyst residues. Furthermore, the use of commodity chemicals like aryl iodides and simple nitrogen ligands ensures a stable and diversified supply base, reducing the risk of single-source bottlenecks that can disrupt production schedules. The robustness of the reaction conditions also implies lower maintenance costs for reactor vessels, as the process does not demand the specialized linings or sealing technologies required for ultra-high vacuum or extreme temperature operations.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with abundant copper salts results in a direct and substantial reduction in raw material costs per kilogram of product. Additionally, the elimination of stoichiometric copper waste and the ability to use cheaper solvents like toluene instead of specialized ethers further drives down the overall cost of goods sold. The simplified purification process, which avoids complex chromatography or heavy metal scavenging steps, reduces utility consumption and waste disposal fees, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: By utilizing reagents that are widely available on the global chemical market, this synthesis route minimizes dependency on niche suppliers who may have limited capacity or long lead times. The stability of the copper catalyst system under ambient conditions means that raw materials can be stored and handled with standard protocols, reducing the risk of degradation or spoilage during transit and storage. This reliability translates into more predictable production cycles and the ability to respond rapidly to fluctuations in market demand for high-purity electronic chemicals.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of toxic heavy metals make this process inherently safer and easier to scale from pilot plant to multi-ton production. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for manufacturing sites. The high atom economy of the coupling reaction, combined with efficient solvent recovery systems, supports a sustainable manufacturing footprint that appeals to environmentally conscious stakeholders and end-users in the green electronics sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the copper-catalyzed synthesis of substituted diphenylamines, providing clarity on the process capabilities and product specifications. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this technology for their supply chains.
Q: Why is copper catalysis preferred over palladium for diphenylamine synthesis?
A: Copper catalysts are significantly less expensive than palladium complexes and do not require stringent anhydrous or oxygen-free conditions, simplifying industrial operations and reducing raw material costs.
Q: What are the key applications of substituted diphenylamines produced by this method?
A: These compounds serve as essential precursors for hole transport materials in OLED displays, light-sensitive dyes, and various pharmaceutical intermediates requiring high purity.
Q: How does the hydrolysis step impact the final product quality?
A: The hydrolysis step effectively removes the acyl protecting group introduced during the coupling reaction, ensuring the final amine product is free from acylated impurities and meets high purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Diphenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of advanced material applications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of substituted diphenylamine performs consistently in your downstream formulations. Our infrastructure is designed to handle complex chemistries with the utmost care, providing a secure and reliable source for your most demanding projects.
We invite you to collaborate with us to unlock the full potential of this cost-effective synthesis route for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive value for your organization.
