Advanced Copper-Catalyzed Synthesis of Substituted Diphenylamines for Industrial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient and cost-effective synthetic routes for critical intermediates. Patent CN101570490A introduces a robust methodology for the preparation of substituted diphenylamines, a class of compounds essential for the production of hole transport materials in organic electronics and various pharmaceutical intermediates. This technology leverages a copper-catalyzed coupling reaction that operates under significantly milder conditions compared to traditional high-temperature gas-phase methods or expensive palladium-catalyzed systems. By optimizing the catalytic system with specific nitrogen-containing ligands and bases, the process achieves high yields while minimizing by-product formation, addressing key pain points for both R&D directors and supply chain managers seeking reliable production pathways.
Substituted diphenylamines serve as pivotal building blocks in the synthesis of complex functional materials, such as N,N,N',N'-tetrakis(4-methylphenyl)-[1,1'-biphenyl]-4,4'-diamine, which is crucial for OLED applications. The ability to synthesize these structures efficiently directly impacts the cost structure and scalability of downstream products. The patent outlines a versatile approach where the structural diversity can be tuned by varying the substituents on the aromatic rings, allowing for the customization of electronic and physical properties required by end-users in the semiconductor and agrochemical sectors.
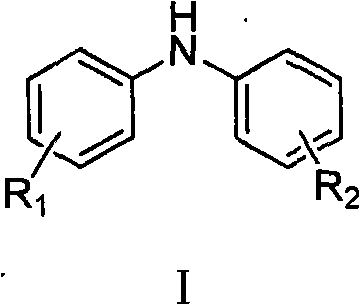
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diphenylamine derivatives has been plagued by significant operational challenges that hinder large-scale adoption. Early methods, such as those described in US2098039, relied on passing aniline over alumina at extreme temperatures reaching 450°C. This gas-phase process not only consumes vast amounts of energy but also suffers from incomplete conversion of raw materials and the generation of ammonia as a hazardous by-product, complicating separation and purification protocols. Furthermore, such harsh thermal conditions often lead to the formation of tars and polymeric side products, drastically reducing the overall yield and purity of the desired symmetric diphenylamines.
More modern approaches utilizing palladium-catalyzed Hartwig-Buchwald coupling have offered milder conditions but introduce their own set of economic and logistical barriers. These reactions typically require specialized palladium complexes that are prohibitively expensive and highly sensitive to moisture and oxygen, necessitating rigorous anhydrous and anaerobic operating environments. The residual palladium in the final product is also a major concern for pharmaceutical and electronic grade materials, requiring additional, costly purification steps to meet stringent metal residue specifications, thereby inflating the total cost of ownership for manufacturers.
The Novel Approach
The methodology presented in CN101570490A represents a strategic shift towards sustainable and economically viable manufacturing by employing earth-abundant copper catalysts. This novel approach utilizes a coupling reaction between an N-acyl aniline derivative and an aryl iodide in the presence of a catalytic amount of copper compounds, such as CuI or CuCl, and nitrogen-based ligands like 1,10-phenanthroline. The reaction proceeds smoothly in common organic solvents like toluene or xylene at moderate temperatures ranging from 110°C to 160°C, eliminating the need for extreme thermal inputs or inert atmosphere gloveboxes.
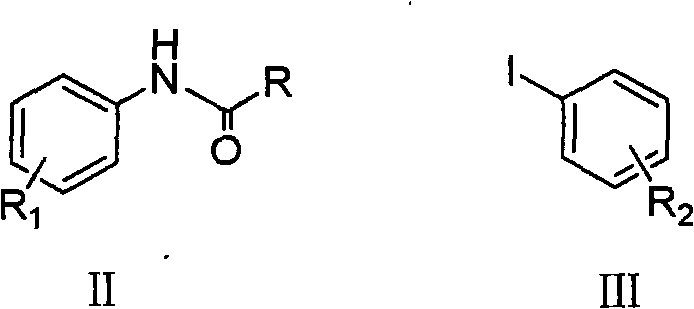
Following the coupling step, a straightforward hydrolysis using acid or base removes the acyl protecting group to reveal the target substituted diphenylamine. This two-step one-pot strategy effectively bypasses the limitations of previous technologies by combining the cost benefits of base metal catalysis with the operational simplicity of open-vessel heating. The use of readily available aryl iodides and protected anilines ensures a flexible substrate scope, enabling the production of asymmetric derivatives that were previously difficult to access via high-temperature condensation methods, thus opening new avenues for material design.
Mechanistic Insights into Copper-Catalyzed C-N Coupling
The core of this synthetic innovation lies in the mechanistic efficiency of the copper-catalyzed C-N bond formation, often referred to as an Ullmann-type coupling. In this catalytic cycle, the copper species, stabilized by the bidentate nitrogen ligand, facilitates the oxidative addition of the aryl iodide, forming a reactive organocopper intermediate. The ligand plays a critical role in preventing the aggregation of copper species into inactive clusters, thereby maintaining high catalytic turnover numbers even at relatively low catalyst loadings of 5-10 mol%. This stabilization allows the reaction to proceed with aryl iodides that might otherwise be unreactive under standard Ullmann conditions, expanding the scope of accessible chemical space.
Impurity control is inherently built into the reaction design through the use of the N-acyl protecting group on the aniline starting material. This protection prevents over-alkylation or polymerization of the amine nitrogen during the coupling phase, ensuring that the primary reaction pathway leads exclusively to the mono-coupled intermediate. Subsequent hydrolysis under acidic or basic conditions cleanly cleaves the amide bond without affecting the newly formed C-N linkage or the aromatic substituents. This orthogonal reactivity ensures a high-purity profile for the final product, minimizing the burden on downstream purification units and aligning with the rigorous quality standards required for high-purity electronic chemical manufacturing.
How to Synthesize Substituted Diphenylamine Efficiently
The synthesis protocol described in the patent offers a streamlined workflow suitable for both laboratory optimization and pilot plant operations. The process begins with the precise weighing of the N-acyl aniline and aryl iodide substrates, which are then dissolved in a high-boiling solvent such as xylene or dioxane. The addition of the base, typically potassium tert-butoxide or cesium carbonate, initiates the deprotonation necessary for the catalytic cycle, followed by the introduction of the copper catalyst and ligand system. Detailed standardized synthesis steps see the guide below.
- Combine N-acyl aniline derivatives and aryl iodides in an organic solvent like toluene or xylene with a copper catalyst and nitrogen ligand.
- Heat the reaction mixture to 110°C-160°C in the presence of a base to facilitate the coupling reaction.
- Perform a hydrolysis step using acid or base to remove the acyl protecting group and isolate the target substituted diphenylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed process offers tangible strategic benefits that extend beyond simple reaction yields. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By replacing precious palladium complexes with commodity copper salts, manufacturers can significantly lower the direct material cost per kilogram of product, creating a more resilient cost structure that is less susceptible to fluctuations in the noble metal market. This cost efficiency is compounded by the elimination of expensive ligand systems often required for palladium catalysis, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The economic impact of switching to copper catalysis is profound, as it removes the necessity for costly metal scavenging resins typically used to reduce palladium residues to ppm levels. Since copper is less toxic and easier to remove via standard aqueous workups, the downstream processing costs are substantially reduced. Additionally, the ability to run the reaction at atmospheric pressure without specialized high-pressure equipment lowers capital expenditure requirements for reactor infrastructure, making the technology accessible for a wider range of production facilities.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available starting materials and reagents. Aryl iodides and N-acyl anilines are commodity chemicals with established global supply chains, reducing the risk of bottlenecks associated with specialized reagents. The robustness of the reaction conditions, which tolerate moderate levels of moisture and oxygen compared to palladium systems, also reduces the risk of batch failures due to environmental excursions, ensuring consistent delivery schedules and reliable inventory management for downstream customers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process generates fewer hazardous by-products compared to high-temperature gas-phase methods. The absence of ammonia generation and the use of recyclable organic solvents simplify waste treatment protocols and reduce the environmental footprint of the manufacturing site. The mild reaction temperatures also enhance operational safety by minimizing thermal runaway risks, facilitating safer scale-up from kilogram to multi-ton production scales while maintaining compliance with increasingly stringent industrial safety regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits and scope of the technology for potential partners and licensees.
Q: Why is copper catalysis preferred over palladium for diphenylamine synthesis?
A: Copper catalysts are significantly more cost-effective than palladium systems and avoid the stringent anhydrous and oxygen-free conditions required for many Pd-catalyzed reactions, simplifying industrial operations.
Q: What types of substituents are compatible with this synthesis method?
A: The method supports a wide range of substituents including methyl, methoxy, amino, and halogen groups at ortho, meta, or para positions, allowing for diverse derivative production.
Q: How does this process improve supply chain reliability?
A: By utilizing commercially available raw materials and avoiding expensive noble metals, the process reduces dependency on scarce resources and minimizes purification bottlenecks associated with metal residue removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Diphenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of substituted diphenylamine performs consistently in your downstream applications, whether for OLED fabrication or pharmaceutical synthesis.
We invite you to collaborate with us to leverage this advanced copper-catalyzed technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall production costs.
