Revolutionizing Substituted Diphenylamine Production: A Cost-Effective Copper-Catalyzed Route for Industrial Scale-Up
Revolutionizing Substituted Diphenylamine Production: A Cost-Effective Copper-Catalyzed Route for Industrial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable manufacturing processes. A pivotal advancement in this domain is documented in patent CN101570490A, which introduces a highly efficient method for synthesizing substituted diphenylamines. These compounds serve as critical building blocks in the production of advanced functional materials, including hole transport materials for organic light-emitting diodes (OLEDs) and various light- and heat-sensitive dyes. The traditional reliance on harsh thermal conditions or expensive noble metal catalysts has long been a bottleneck for manufacturers seeking to optimize their supply chains. This patent presents a transformative solution by leveraging copper-catalyzed C-N coupling reactions, offering a pathway that balances high purity with economic feasibility.
For R&D directors and technical leaders, the significance of this innovation lies in its ability to bypass the limitations of prior art while maintaining rigorous quality standards. The method utilizes a sophisticated catalytic system comprising copper compounds, nitrogen-containing ligands, and bases to facilitate the coupling of protected aniline derivatives with iodo-benzene derivatives. This approach not only mitigates the formation of unwanted by-products but also ensures that the reaction proceeds under relatively mild conditions. By shifting the paradigm from energy-intensive thermal processes to catalytic precision, this technology opens new avenues for the reliable production of high-purity electronic chemicals and dye intermediates, addressing the growing demand for performance-driven materials in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted diphenylamines has been plagued by significant technical and economic challenges that hinder efficient large-scale production. One of the earliest methods involved passing aniline over alumina at extreme temperatures reaching 450°C. While conceptually simple, this thermal approach suffers from severe drawbacks, including the generation of ammonia gas as a hazardous by-product and incomplete conversion of raw materials. These factors create substantial difficulties in downstream separation and purification, leading to lower overall yields and increased waste management costs. Furthermore, this high-temperature process is restricted to synthesizing symmetrically substituted diphenylamines, limiting its versatility for producing the diverse array of asymmetric derivatives required in modern applications.
More recent advancements have explored the use of palladium-catalyzed C-N coupling reactions, such as the Hartwig-Buchwald reaction. Although these methods offer milder reaction conditions and higher yields, they introduce a different set of constraints related to cost and operational complexity. Palladium catalysts are notoriously expensive and often require specialized, air- and moisture-sensitive ligands. Consequently, reactions must be conducted under strictly anhydrous and oxygen-free conditions, necessitating specialized equipment and rigorous inert atmosphere protocols. For procurement managers and supply chain heads, the reliance on precious metals and the associated safety infrastructure translates into elevated capital expenditure and operational risks, making these routes less attractive for commodity-scale manufacturing of intermediates.
The Novel Approach
The methodology outlined in patent CN101570490A represents a strategic breakthrough by substituting expensive palladium systems with robust copper-based catalysts. This novel approach employs a catalytic amount of copper compounds, such as cuprous iodide (CuI) or cuprous chloride (CuCl), in conjunction with nitrogen-containing ligands like 1,10-phenanthroline or 1,2-cyclohexanediamine. The reaction is conducted in common organic solvents such as toluene, xylene, or dioxane, utilizing bases like potassium tert-butoxide or cesium carbonate to drive the coupling efficiency. This shift to copper catalysis drastically reduces the raw material costs while maintaining high catalytic activity, effectively bridging the gap between the low cost of thermal methods and the high performance of palladium chemistry.
Moreover, the process incorporates a subsequent hydrolysis step using either acid or base to remove protecting groups from the intermediate amides or carbamates, yielding the target substituted diphenylamine with high purity. The reaction conditions are remarkably mild, typically operating between 110°C and 160°C, which significantly lowers energy consumption compared to the 450°C thermal method. The absence of sensitive requirements for water and oxygen exclusion simplifies the operational protocol, allowing for easier handling and safer scale-up. This combination of mild conditions, cost-effective reagents, and a streamlined workup procedure positions this copper-catalyzed route as a superior alternative for the commercial scale-up of complex fine chemical intermediates.
Mechanistic Insights into Copper-Catalyzed C-N Coupling
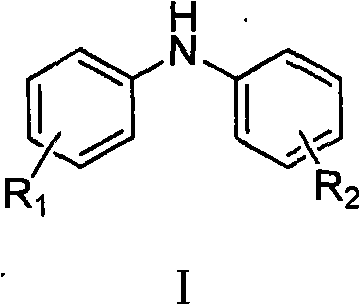
At the heart of this synthesis lies a sophisticated copper-catalyzed C-N bond formation mechanism, often referred to as an Ullmann-type coupling. The reaction initiates with the coordination of the nitrogen-containing ligand to the copper center, forming an active catalytic species that is capable of oxidative addition with the aryl iodide substrate (Formula III). This step is crucial as it activates the carbon-iodine bond, making it susceptible to nucleophilic attack by the nitrogen atom of the protected aniline derivative (Formula II). The presence of the base facilitates the deprotonation of the amine or amide nitrogen, enhancing its nucleophilicity and driving the formation of the copper-amido intermediate. This mechanistic pathway ensures that the coupling occurs selectively at the desired position, minimizing the formation of regio-isomers that could complicate purification.
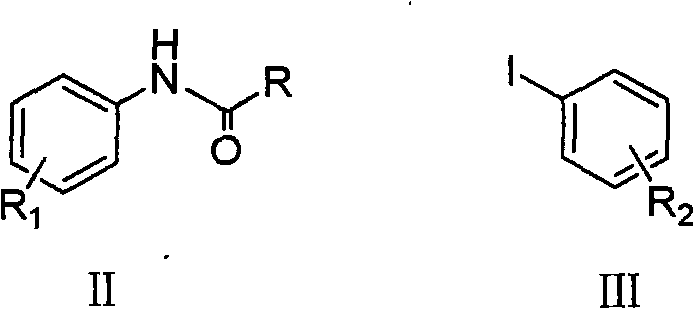
Following the coupling event, the catalytic cycle is completed through reductive elimination, releasing the coupled product and regenerating the active copper catalyst for further turnover. A distinct advantage of this specific patent embodiment is the use of protected amine precursors (amides or carbamates) rather than free amines. This protection strategy is vital for impurity control, as it prevents side reactions such as poly-alkylation or oxidation of the free amine during the harsh coupling conditions. The final hydrolysis step, whether acidic or basic, cleanly cleaves the protecting group (R in Formula II) to reveal the free secondary amine in the target molecule (Formula I). This two-stage process—coupling followed by deprotection—provides a robust mechanism for controlling the impurity profile, ensuring that the final product meets the stringent purity specifications required for electronic and pharmaceutical applications.
How to Synthesize Substituted Diphenylamine Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of operations that can be readily adapted to existing reactor infrastructure. The process begins by charging a reaction vessel with the N-substituted aniline derivative and the iodo-benzene derivative in a suitable solvent such as toluene or xylene. Once the solids are dissolved, the base, copper catalyst, and ligand are added sequentially under stirring. The mixture is then heated to the specified temperature range of 110°C to 160°C and maintained for a duration sufficient to achieve complete conversion, typically around 10 hours as demonstrated in the patent examples. Following the coupling reaction, the mixture is cooled, washed to remove inorganic salts, and subjected to hydrolysis to yield the final product.
- Combine N-substituted aniline derivatives (Formula II) and iodo-benzene derivatives (Formula III) in an organic solvent like toluene or xylene.
- Add a catalytic amount of copper compound (e.g., CuI, CuCl), a nitrogen-containing ligand (e.g., 1,10-phenanthroline), and a base (e.g., t-BuOK, K3PO4).
- Heat the mixture to 110°C-160°C for coupling, followed by acid or base hydrolysis to remove protecting groups and isolate the target diphenylamine (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN101570490A offers compelling strategic advantages that directly impact the bottom line. The most significant benefit is the drastic reduction in raw material costs achieved by eliminating the need for precious metal catalysts. By replacing palladium with copper, manufacturers can avoid the volatility associated with noble metal pricing and reduce the cost of goods sold substantially. Additionally, the use of common, commercially available solvents and reagents ensures a stable supply chain, reducing the risk of production delays caused by the scarcity of specialized chemicals. This stability is crucial for maintaining continuous production schedules and meeting the delivery commitments of downstream customers in the electronics and dye industries.
- Cost Reduction in Manufacturing: The transition from palladium to copper catalysis results in significant cost savings not only in catalyst procurement but also in downstream processing. Noble metal catalysts often require complex and expensive removal steps to meet residual metal specifications, particularly for electronic grade materials. The copper-catalyzed process simplifies purification, as copper residues are generally easier to manage and remove using standard aqueous washes. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product. These cumulative efficiencies translate into a more competitive pricing structure for the final substituted diphenylamine intermediates.
- Enhanced Supply Chain Reliability: The reliance on commoditized reagents such as cuprous iodide, phenanthroline, and toluene enhances the resilience of the supply chain. Unlike specialized palladium ligands that may have limited suppliers and long lead times, the components of this copper system are widely produced and readily accessible in the global chemical market. This accessibility minimizes the risk of supply disruptions and allows for flexible sourcing strategies. Moreover, the robustness of the reaction to ambient moisture and oxygen reduces the need for specialized inert atmosphere equipment, lowering the barrier to entry for contract manufacturing organizations and enabling faster technology transfer between sites.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a greener alternative to traditional high-temperature synthesis. The avoidance of 450°C thermal conditions eliminates the generation of ammonia gas, a hazardous pollutant that requires extensive scrubbing systems. The use of recyclable solvents like toluene and xylene aligns with standard industrial waste management practices, facilitating compliance with environmental regulations. The high yields and selectivity of the reaction minimize waste generation, supporting sustainability goals. The process is inherently scalable, having been designed with industrial production in mind, ensuring that capacity can be expanded from pilot scale to multi-ton production without encountering unforeseen engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the copper-catalyzed synthesis of substituted diphenylamines. These insights are derived directly from the experimental data and claims presented in patent CN101570490A, providing clarity on the feasibility and benefits of this manufacturing route. Understanding these details is essential for stakeholders evaluating the potential integration of this technology into their existing production portfolios.
Q: What are the primary advantages of this copper-catalyzed method over palladium-catalyzed routes?
A: The copper-catalyzed method described in CN101570490A significantly reduces raw material costs by replacing expensive palladium catalysts with abundant copper salts. Furthermore, it eliminates the stringent requirement for anhydrous and oxygen-free conditions often needed for palladium systems, thereby simplifying operational complexity and enhancing safety in large-scale manufacturing environments.
Q: How does this process improve impurity control compared to traditional high-temperature methods?
A: Unlike traditional thermal methods requiring temperatures as high as 450°C which generate ammonia by-products and lead to difficult separations, this novel approach operates at mild temperatures (110°C-160°C). The inclusion of a specific hydrolysis step allows for the clean removal of protecting groups, resulting in a cleaner crude product profile and reducing the burden on downstream purification processes.
Q: Is this synthesis route suitable for large-scale industrial production of hole transport materials?
A: Yes, the patent explicitly highlights the method's suitability for industrialization. The use of common organic solvents like toluene and xylene, combined with commercially available copper catalysts and ligands, ensures that the process is robust and scalable. The high yields reported in the examples demonstrate its viability for producing key precursors for hole transport materials and sensitive dyes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Diphenylamine Supplier
The technological advancements detailed in patent CN101570490A underscore the immense potential of copper-catalyzed coupling in the production of high-value fine chemicals. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative pathways for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch.
We invite partners in the electronic materials and fine chemical sectors to collaborate with us to optimize their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our capabilities can support your project goals. Let us help you secure a reliable supply of high-quality intermediates while achieving your cost and sustainability targets.
