Advanced Green Synthesis of H Acid: Enhancing Purity and Sustainability for Global Dye Manufacturers
Advanced Green Synthesis of H Acid: Enhancing Purity and Sustainability for Global Dye Manufacturers
The global demand for high-performance azo dyes continues to drive the need for robust and environmentally compliant supply chains for critical intermediates like H acid (1-amino-8-naphthol-3,6-disulfonic acid). Patent CN102936214B introduces a transformative methodology for the clean preparation of H acid, addressing the longstanding industry challenge of excessive wastewater and acidic waste generation. By integrating a closed-loop mother liquor recycling system with catalytic hydrogenation, this technology offers a viable pathway for cost reduction in dye intermediate manufacturing while adhering to stringent environmental regulations. The process utilizes refined naphthalene as the starting material, undergoing sulfonation to produce 2,7-naphthalene disulfonic acid, which is subsequently nitrated directly within the sulfonation mother liquor. This innovative approach not only maximizes atom economy but also significantly mitigates the ecological footprint associated with traditional diazo component production, positioning it as a cornerstone technology for sustainable commercial scale-up of complex dye intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of H acid has been plagued by significant environmental and operational inefficiencies, particularly regarding waste management and byproduct handling. Traditional diamine methods often rely on iron powder reduction to convert nitro groups into amino groups, a process that generates massive quantities of iron sludge that are difficult and costly to dispose of safely. Furthermore, conventional protocols typically treat sulfonation and nitration mother liquors as waste streams, leading to substantial acidic wastewater discharge that requires expensive neutralization and treatment before release. The accumulation of impurities in recycled acids was also a persistent issue in older iterations, often resulting in decreased product purity, darker coloration, and increased viscosity that hampered downstream processing. These factors collectively contributed to high operational expenditures and regulatory risks for manufacturers relying on legacy technologies for high-purity dye intermediates.
The Novel Approach
The methodology disclosed in CN102936214B fundamentally restructures the production workflow to eliminate these bottlenecks through strategic resource integration and advanced separation techniques. Instead of discarding acidic streams, the process recycles nitration mother liquor directly back into the sulfonation reaction step and循环利用 hydrolysis mother liquor for subsequent hydrolysis batches, drastically cutting fresh acid consumption. Crucially, the replacement of iron powder reduction with catalytic hydrogenation using Pd/C removes the source of heavy metal sludge entirely, streamlining the purification process. To address the challenge of impurity buildup during recycling, the patent introduces a specific solvent extraction step using trialkyl tertiary amines after defined cycle counts, effectively resetting the quality of the mother liquor. This holistic design ensures consistent product specifications while achieving a wastewater reduction of over 80% compared to existing processes, making it an ideal solution for reducing lead time for high-purity dye intermediates through streamlined operations.
Mechanistic Insights into Catalytic Hydrogenation and Isomerization
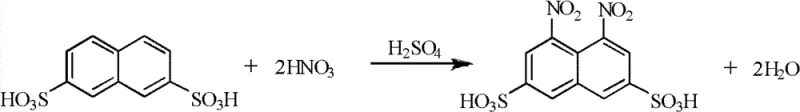
A critical component of this enhanced synthesis route is the precise control over reaction thermodynamics and catalytic efficiency, particularly during the reduction and isomerization phases. The process employs a palladium on carbon (Pd/C) catalyst for the hydrogenation of 1,8-dinitro-3,6-naphthalene disulfonic acid, operating under controlled pressures of 1.0-2.5 MPa and temperatures ranging from 40-130°C. This catalytic environment facilitates the selective reduction of nitro groups to amino groups without affecting the sulfonic acid moieties, yielding 1,8-diamino-3,6-naphthalene disulfonic acid with high selectivity. Unlike stoichiometric reductions, this catalytic cycle allows for the recovery and reuse of the catalyst, further enhancing the economic viability of the process. The subsequent acidic hydrolysis step converts the diamino compound into the final H acid product, where the reaction temperature is carefully maintained between 105-135°C to ensure complete conversion while minimizing degradation. This mechanistic precision is vital for maintaining the structural integrity required for reliable dye intermediate supplier standards.
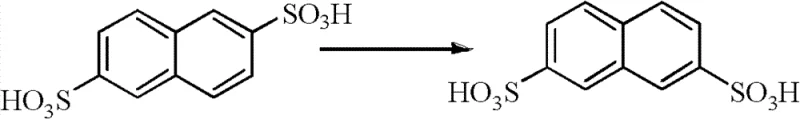
Furthermore, the patent details a sophisticated strategy for managing the 2,6-naphthalene disulfonic acid byproduct generated during the initial sulfonation of naphthalene. Rather than treating this isomer as waste, the process subjects it to a transposition reaction with sulfuric acid at elevated temperatures (155-210°C), converting approximately 35% of the 2,6-isomer back into the desired 2,7-isomer. This recovered material is then mixed with fresh naphthalene and recycled mother liquor for further sulfonation, effectively closing the mass balance loop. The integration of this isomerization step significantly boosts the overall yield of the sulfonation stage to greater than 90%, maximizing raw material utilization. Coupled with the extraction protocol that removes accumulated organics using a three-component oil phase system (extractant, diluent, additive), the mechanism ensures that the reaction medium remains chemically stable over multiple cycles, preventing the quality drift often seen in batch recycling processes.
How to Synthesize H Acid Efficiently
The implementation of this clean preparation method requires strict adherence to the optimized reaction parameters and recycling protocols outlined in the patent data to achieve the reported efficiency gains. The synthesis begins with the sulfonation of refined naphthalene using concentrated sulfuric acid at a molar ratio of 1:3.5-6.0, heated to 155-200°C for 5-7 hours to ensure complete conversion. Following the separation of the 2,6-isomer, the remaining liquor undergoes nitration with 97-99% nitric acid at 0-70°C, followed by the crucial catalytic hydrogenation and hydrolysis steps. Detailed standard operating procedures regarding pressure controls, catalyst loading, and extraction intervals are essential for replicating the >80% waste reduction and high yield performance. For a comprehensive breakdown of the specific operational parameters and safety guidelines, please refer to the standardized synthesis guide below.
- Sulfonate refined naphthalene with concentrated sulfuric acid at 155-200°C to generate 2,7-naphthalene disulfonic acid, separating the 2,6-isomer byproduct.
- Directly nitrate the 2,7-isomer in the sulfonation mother liquor using 97-99% nitric acid at 0-70°C to form 1,8-dinitro-3,6-naphthalene disulfonic acid.
- Perform catalytic hydrogenation using Pd/C followed by acidic hydrolysis at 105-135°C, utilizing extraction to treat recycled mother liquors for impurity removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this patented technology translates into tangible operational improvements and risk mitigation strategies that extend beyond simple unit cost metrics. By eliminating the generation of iron sludge, manufacturers can bypass the complex and costly logistics associated with hazardous heavy metal waste disposal, thereby simplifying the environmental compliance landscape. The ability to recycle acidic mother liquors multiple times significantly reduces the consumption of raw materials like concentrated sulfuric acid and nitric acid, insulating the production cost from volatile commodity price fluctuations. Moreover, the extraction-based purification of recycled streams ensures that product quality remains consistent over long production runs, reducing the risk of batch failures and supply interruptions. These factors collectively contribute to a more resilient and predictable supply chain for critical textile chemicals.
- Cost Reduction in Manufacturing: The elimination of iron powder reduction removes the need for expensive filtration equipment dedicated to sludge removal and the subsequent treatment of iron-containing wastewater. Additionally, the recycling of nitration and hydrolysis mother liquors drastically lowers the volume of fresh acid required per kilogram of product, directly reducing variable production costs. The recovery and isomerization of the 2,6-naphthalene disulfonic acid byproduct further enhance raw material efficiency, ensuring that a higher percentage of the input naphthalene is converted into saleable H acid. This multi-faceted approach to cost optimization creates a leaner manufacturing profile that is less susceptible to margin erosion from waste disposal fees and raw material price hikes.
- Enhanced Supply Chain Reliability: The robust nature of the mother liquor recycling system, supported by periodic extraction cleaning, ensures stable production throughput without the frequent shutdowns required for reactor cleaning in traditional processes. High yields exceeding 90% in the sulfonation step and efficient conversion in subsequent stages mean that less feedstock is needed to meet the same output targets, securing inventory levels against upstream supply constraints. The use of heterogeneous catalysis (Pd/C) allows for easier catalyst separation and reuse compared to homogeneous systems, minimizing downtime associated with catalyst replenishment. This operational stability is crucial for maintaining consistent delivery schedules to downstream dye manufacturers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as sulfonation, nitration, and hydrogenation that can be easily adapted to larger reactor volumes. The significant reduction in wastewater volume (over 80%) simplifies the requirements for effluent treatment plants, allowing facilities to operate within stricter environmental permits without massive capital investment in new treatment infrastructure. By proactively addressing the accumulation of impurities through solvent extraction, the technology prevents the long-term degradation of process streams that often limits the lifespan of recycling loops in other green chemistry applications. This forward-looking design ensures long-term viability and regulatory alignment for large-scale chemical production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this clean H acid synthesis method, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value proposition of suppliers utilizing this route. The answers provided reflect the specific mechanisms of waste reduction, impurity control, and yield optimization that distinguish this process from conventional manufacturing standards.
Q: How does this process reduce wastewater compared to traditional H acid manufacturing?
A: The process achieves over 80% reduction in wastewater discharge by recycling nitration and hydrolysis mother liquors back into the sulfonation and hydrolysis steps respectively, rather than disposing of them as waste acid.
Q: What is the advantage of using catalytic hydrogenation over iron powder reduction?
A: Catalytic hydrogenation using Pd/C eliminates the generation of large quantities of toxic iron sludge, significantly simplifying downstream filtration and reducing environmental compliance costs associated with heavy metal waste disposal.
Q: How are accumulated impurities managed during mother liquor recycling?
A: After multiple recycling cycles (five for nitration liquor, ten for hydrolysis liquor), an extraction process using trialkyl tertiary amines is employed to remove accumulated organic impurities, ensuring consistent product quality and preventing viscosity issues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable H acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient chemical processes is not just an environmental imperative but a strategic business advantage. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the clean preparation of H acid can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of H acid meets the exacting standards required for high-performance reactive and acid dyes. Our commitment to process excellence means we can deliver the consistency and quality necessary to support your most demanding applications.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes translate into value for your organization. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our technical claims and explore the potential for long-term collaboration in the dye intermediate sector.
