Advanced Clean Synthesis of H Acid: Enhancing Yield and Sustainability for Global Dye Manufacturers
The chemical industry is currently undergoing a paradigm shift towards greener manufacturing processes, driven by stringent environmental regulations and the economic necessity of resource efficiency. A pivotal development in this arena is documented in patent CN102936214A, which outlines a revolutionary method for the clean preparation of H acid (1-amino-8-naphthol-3,6-disulfonic acid), a critical intermediate in the production of reactive dyes and azo colors. This technology addresses the historical痛点 of the H acid industry: the generation of excessive acidic wastewater and iron sludge associated with traditional reduction methods. By integrating catalytic hydrogenation with a sophisticated mother liquor recycling loop, the process achieves a wastewater reduction of more than 80% compared to existing techniques while simultaneously enhancing product yield and purity. For global procurement leaders and R&D directors, this represents a significant opportunity to secure a reliable H acid supplier capable of meeting both cost and sustainability targets without compromising on the rigorous quality standards required for textile and dye applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of H acid has relied heavily on the diamine method involving iron powder reduction, a process fraught with severe environmental and operational inefficiencies. The traditional route typically involves the reduction of dinitro intermediates using iron powder in acidic media, which generates vast quantities of iron mud sludge that are difficult and costly to dispose of in compliance with modern environmental standards. Furthermore, the conventional process often treats sulfonation and nitration mother liquors as waste streams, leading to substantial loss of valuable sulfuric acid and unreacted organic materials. This linear consumption model not only inflates the cost reduction in dye intermediate manufacturing initiatives but also creates a supply chain vulnerability due to the increasing difficulty of obtaining permits for waste discharge. The accumulation of by-products, particularly the 2,6-naphthalene disulfonic acid isomer, further complicates the purification process, often resulting in lower final yields and inconsistent product quality that fails to meet the high specifications demanded by advanced textile applications.
The Novel Approach
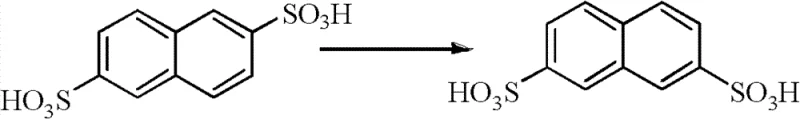
In stark contrast, the methodology disclosed in CN102936214A introduces a circular economy approach to H acid synthesis, fundamentally altering the reaction landscape through catalytic hydrogenation and strategic mother liquor reuse. Instead of employing polluting iron powder, the process utilizes a palladium on carbon (Pd/C) catalyst for the reduction step, which eliminates the formation of heavy metal sludge entirely and allows for the catalyst to be filtered and reused in subsequent batches. A key innovation lies in the direct nitration of the sulfonation mother liquor, where the nitration mother liquor is subsequently recycled back into the sulfonation step, creating a closed loop that maximizes acid utilization. Additionally, the process incorporates a transposition reaction to convert the unwanted 2,6-naphthalene disulfonic acid byproduct back into the useful 2,7-isomer, thereby turning a waste liability into a valuable asset. This holistic integration of reaction steps ensures that the commercial scale-up of complex dye intermediates becomes not only environmentally viable but also economically superior.
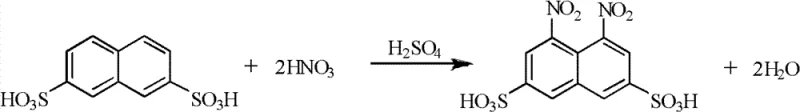
Mechanistic Insights into Catalytic Hydrogenation and Acid Hydrolysis
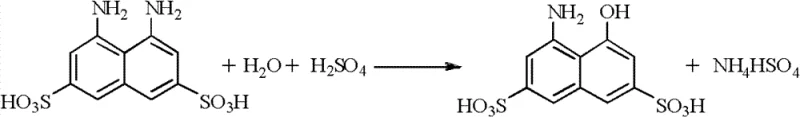
The core of this technological advancement rests on the precise control of reaction conditions during the catalytic hydrogenation and subsequent hydrolysis phases. In the hydrogenation step, the 1,8-dinitro-3,6-naphthalene disulfonic acid is dissolved in water and neutralized before being subjected to hydrogen pressure ranging from 1.0 to 2.5 MPa in the presence of 0.5% to 10% Pd/C catalyst at temperatures between 40°C and 130°C. This specific window of operating parameters is critical for ensuring complete reduction of the nitro groups to amino groups without causing desulfonation or other side reactions that could compromise the molecular integrity of the intermediate. The use of a heterogeneous catalyst like Pd/C facilitates easy separation via filtration, allowing the catalyst cake to be directly recycled for the next batch, which significantly lowers the variable costs associated with precious metal consumption. Following reduction, the resulting 1,8-diamino-3,6-naphthalene disulfonic acid undergoes acidic hydrolysis, where one amino group is selectively converted to a hydroxyl group to form the final H acid structure.
To maintain the efficiency of this cycle over prolonged operation, the patent details a robust mechanism for impurity management within the recycled mother liquors. As the sulfonation and hydrolysis mother liquors are reused, organic impurities and degradation products inevitably accumulate, which can lead to product discoloration and reduced reaction rates if left unchecked. The invention solves this by implementing a periodic extraction protocol using tertiary amines, such as triheptylamine or trioctylamine, dissolved in a diluent like kerosene or xylene. This extraction step is triggered after the mother liquor has been recycled approximately five to ten times, effectively scrubbing the aqueous phase of high-molecular-weight contaminants and reducing the Chemical Oxygen Demand (COD) from levels as high as 50,000 mg/L down to approximately 2,500 mg/L. This purification ensures that the recycled acid retains its reactivity and that the final high-purity H acid meets strict color and content specifications, validating the process for continuous industrial application.
How to Synthesize H Acid Efficiently
Implementing this clean synthesis route requires careful attention to the sequential integration of sulfonation, nitration, and hydrolysis steps, with particular emphasis on the thermal management of the transposition reaction. The process begins with the sulfonation of refined naphthalene using concentrated sulfuric acid at elevated temperatures of 155°C to 200°C, followed by the separation of the 2,6-isomer which is then subjected to a rearrangement reaction to regenerate the 2,7-isomer. The detailed operational parameters, including the specific molar ratios of naphthalene to sulfuric acid (1:3.5 to 6.0) and the precise temperature ramps for nitration and hydrolysis, are critical for achieving the reported yields of over 90%. For R&D teams looking to adopt this technology, understanding the interplay between acid concentration and reaction kinetics is essential to replicate the high efficiency described in the patent documentation.
- Sulfonate refined naphthalene with concentrated sulfuric acid at 155-200°C to produce 2,7-naphthalene disulfonic acid, separating the 2,6-isomer byproduct.
- Directly nitrate the sulfonation mother liquor with nitric acid to form 1,8-dinitro-3,6-naphthalene disulfonic acid, recycling the nitration mother liquor.
- Perform catalytic hydrogenation using Pd/C at 1.0-2.5 MPa to reduce the dinitro compound to the diamine derivative.
- Hydrolyze the diamine intermediate with sulfuric acid at 105-135°C to yield H acid, recycling the hydrolysis mother liquor.
- Treat accumulated mother liquors with tertiary amine extraction agents after multiple cycles to remove impurities and maintain reaction efficiency.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this clean preparation method offers transformative benefits that extend far beyond simple regulatory compliance. The elimination of iron powder reduction removes the logistical burden and cost associated with the disposal of hazardous iron sludge, a significant overhead in traditional dye intermediate manufacturing. Furthermore, the closed-loop recycling of sulfuric acid mother liquors drastically reduces the consumption of fresh acid, insulating the production process from volatility in raw material pricing and ensuring a more stable cost structure. By converting the 2,6-isomer byproduct back into the main reaction stream, the process maximizes the yield per ton of naphthalene feedstock, effectively lowering the unit cost of production without requiring expensive new raw material sources. These efficiencies collectively contribute to a substantial cost savings profile that makes the final H acid product highly competitive in the global market.
- Cost Reduction in Manufacturing: The transition from stoichiometric iron reduction to catalytic hydrogenation eliminates the need for purchasing large quantities of iron powder and the subsequent costs of sludge treatment and disposal. Additionally, the ability to recycle the Pd/C catalyst multiple times minimizes the consumption of precious metals, while the recovery of sulfuric acid from mother liquors reduces the demand for fresh acid procurement. This multi-faceted approach to cost optimization ensures that the manufacturing process remains economically resilient even when facing fluctuations in commodity prices, providing a distinct financial advantage over legacy production methods.
- Enhanced Supply Chain Reliability: By implementing a continuous recycling loop for mother liquors, the process reduces the dependency on external waste treatment facilities, which can often become bottlenecks during periods of strict environmental enforcement. The internal management of impurities through tertiary amine extraction ensures that production can continue uninterrupted for extended periods without the need for frequent shutdowns to clean reactors or replace degraded acid baths. This operational stability translates directly into reducing lead time for high-purity H acid deliveries, allowing suppliers to meet tight customer deadlines and maintain consistent inventory levels for downstream dye manufacturers.
- Scalability and Environmental Compliance: The modular nature of the extraction and recycling units allows for seamless scalability from pilot plant operations to full-scale commercial production capacities ranging from 100 kgs to 100 MT annually. The significant reduction in wastewater volume and COD load simplifies the permitting process for new facilities and reduces the risk of fines or shutdowns due to non-compliance with local environmental regulations. This alignment with green chemistry principles not only future-proofs the supply chain against tightening legislation but also enhances the brand reputation of companies adopting this technology as leaders in sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean H acid synthesis technology. These insights are derived directly from the experimental data and process descriptions found in patent CN102936214A, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is crucial for making informed decisions about technology transfer and process optimization.
Q: How does this method reduce wastewater compared to traditional iron powder reduction?
A: Traditional methods generate massive amounts of iron mud sludge and acidic wastewater. This patented process replaces iron powder with Pd/C catalytic hydrogenation and implements a closed-loop mother liquor recycling system for sulfonation, nitration, and hydrolysis steps, reducing wastewater discharge by over 80%.
Q: What is done with the 2,6-naphthalene disulfonic acid byproduct?
A: Instead of discarding the 2,6-isomer, the process utilizes a transposition reaction where the 2,6-naphthalene disulfonic acid is heated with sulfuric acid to rearrange it back into the desired 2,7-isomer, significantly improving overall atom economy and raw material utilization.
Q: How are impurities managed during long-term mother liquor recycling?
A: To prevent impurity accumulation that degrades product quality, the process employs a solvent extraction step using tertiary amines (such as triheptylamine) after the mother liquors have been recycled 5 to 10 times, effectively lowering COD levels and ensuring consistent product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable H Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to cleaner, more efficient chemical processes is not just an environmental imperative but a strategic business advantage. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated mother liquor recycling and catalytic hydrogenation techniques described in CN102936214A can be successfully translated from the laboratory to the plant floor. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of H acid performs consistently in your dye formulation processes. Our infrastructure is designed to handle complex organic syntheses with the highest regard for safety and environmental stewardship, making us an ideal partner for long-term supply agreements.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits of switching to this cleaner production route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make a data-driven decision that enhances both your supply chain resilience and your corporate sustainability goals.
