Advanced Synthesis of Sulfo-Containing Sulfur Ylides for Scalable Pharmaceutical Intermediate Production
Advanced Synthesis of Sulfo-Containing Sulfur Ylides for Scalable Pharmaceutical Intermediate Production
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for safer, more efficient, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is detailed in patent CN111116441A, which discloses a novel synthesis method and application for sulfo-containing sulfur ylides. This technology represents a significant departure from conventional methodologies that rely on hazardous volatile sulfides, offering a robust pathway for the production of epoxy compounds, which are critical intermediates in the pharmaceutical and agrochemical sectors. By introducing a sulfonic acid group into the sulfur ylide structure, this innovation not only mitigates the toxicity associated with traditional reagents but also enables a closed-loop recycling process for the starting materials. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the mechanistic and economic implications of this patent is essential for optimizing supply chains and reducing overall manufacturing costs.
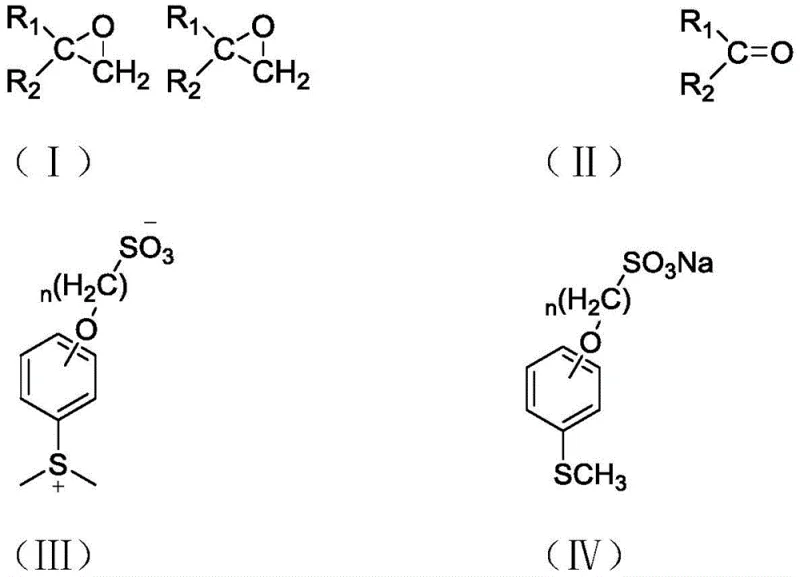
The core of this technology lies in the transformation of a stable sulfonate salt into a reactive sulfur ylide, which subsequently reacts with carbonyl compounds to form ethylene oxide derivatives. The process is characterized by mild reaction conditions for the epoxidation step and the ability to recover the sulfonate precursor from the aqueous phase post-reaction. This dual benefit of operational safety and material efficiency positions this method as a superior alternative for the commercial scale-up of complex polymer additives and active pharmaceutical ingredients. As we delve deeper into the technical specifics, it becomes clear how this approach addresses the longstanding pain points of volatility, odor, and separation difficulties inherent in older sulfur ylide chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfur ylides for epoxidation reactions has heavily relied on dimethyl sulfide or dimethyl sulfoxide as primary starting materials. While chemically effective, these substances present severe logistical and safety challenges in an industrial setting. Dimethyl sulfide, for instance, is a toxic substance with a low boiling point and a notoriously foul smell, making it difficult to handle without specialized containment equipment and posing significant risks to worker health and environmental compliance. Furthermore, when dimethyl sulfide reacts with methylating agents to form the necessary sulfonium salts, the resulting compounds often exhibit low activity, limiting their utility in high-efficiency synthesis. Another critical drawback is the difficulty in separating the byproducts; for example, when propoxy-methyl sulfide is used as a substitute, it shares a high boiling point similar to the target epoxy compound, making distillation and purification energy-intensive and economically inefficient. These factors collectively contribute to higher operational expenditures and increased regulatory scrutiny for manufacturers relying on legacy sulfide chemistry.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a sulfo-containing sulfur ylide that fundamentally alters the physical properties of the reagent system. By synthesizing the ylide from a sulfonate salt, specifically 4-(4-(methylthio)phenoxy)butane-1-sodium sulfonate, the process eliminates the volatility and odor issues associated with dimethyl sulfide. The presence of the sulfonic acid group renders the precursor and its regenerated form highly water-soluble, a property that is ingeniously exploited for product isolation. Instead of complex distillation trains required to separate close-boiling sulfides, the novel approach allows for a simple liquid-liquid extraction where the organic epoxide product is separated from the aqueous layer containing the valuable sulfonate. This structural modification not only enhances the safety profile of the manufacturing process but also drastically simplifies the downstream processing workflow. For a reliable pharmaceutical intermediate supplier, adopting such a route means transitioning from a hazard-heavy operation to a streamlined, green chemistry protocol that aligns with modern sustainability goals.
Mechanistic Insights into Sulfo-Containing Sulfur Ylide Formation and Epoxidation
The chemical mechanism underpinning this invention involves a two-stage sequence that balances thermal activation with selective reactivity. The first stage is the formation of the sulfur ylide itself, which requires the methylation of the sulfonate salt. This step is conducted under solvent-free or melt conditions at elevated temperatures ranging from 220°C to 260°C, typically optimized around 240°C. At these temperatures, the sulfonate salt melts and reacts with a methylating agent such as dimethyl sulfate, methyl iodide, or methyl bromide. The high thermal energy facilitates the nucleophilic attack of the sulfur atom on the methyl group, generating the positively charged sulfonium center essential for ylide formation. This high-temperature methylation is critical for achieving high conversion rates, with experimental data indicating yields exceeding 87% for the ylide intermediate. The stability of the sulfonate backbone during this harsh thermal treatment demonstrates the robustness of the molecular design, ensuring that the functional integrity of the chain is maintained despite the rigorous conditions.
Following the generation of the ylide, the second stage involves the Corey-Chaykovsky type epoxidation reaction with a carbonyl compound, such as acetophenone. This reaction proceeds under much milder conditions, typically between 40°C and 60°C, in the presence of a base like sodium hydroxide or potassium carbonate. The base deprotonates the alpha-carbon adjacent to the sulfonium center, generating the reactive ylide species in situ, which then attacks the carbonyl carbon to form the epoxide ring. A crucial aspect of this mechanism is the fate of the sulfur byproduct; upon ring closure, the sulfur moiety is expelled as the original sulfonate salt. Because this salt is ionic and highly polar, it partitions exclusively into the aqueous phase during workup, while the neutral epoxide product remains in the organic phase. This inherent phase separation driven by the chemical nature of the leaving group is the key to the process's efficiency, allowing for the direct recovery and reuse of the sulfonate without the need for complex chromatographic purification.
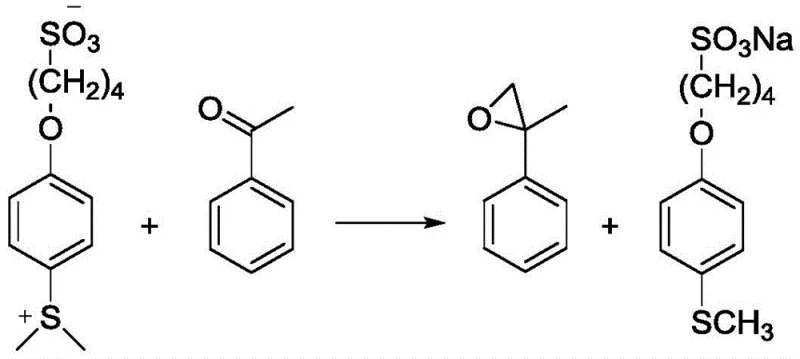
How to Synthesize Sulfo-Containing Sulfur Ylide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the sulfonate precursor via nucleophilic substitution, followed by the high-temperature methylation to generate the active ylide. Finally, the ylide is reacted with the target ketone or aldehyde under basic conditions to afford the desired epoxide. Each step has been optimized in the patent examples to demonstrate reproducibility and scalability, providing a clear roadmap for industrial adoption. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the technical guide below to assist process chemists in replicating these results.
- Synthesize the sulfonate precursor by reacting 1-(4-bromobutoxy)-4-(methylthio)benzene with anhydrous sodium sulfite in a solvent system like water/ethanol at reflux temperatures.
- Convert the sulfonate salt into the sulfur ylide by heating it to 220-260°C and reacting it with a methylating agent such as dimethyl sulfate.
- Perform the epoxidation reaction by mixing the sulfur ylide with a carbonyl compound and a base in a solvent at 40-60°C, followed by aqueous workup to recover the sulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this sulfo-containing sulfur ylide technology offers transformative benefits for procurement managers and supply chain heads focused on cost reduction in fine chemical manufacturing. The primary economic driver is the recyclability of the sulfonate precursor. In traditional processes, the sulfur reagent is often consumed or converted into a waste stream that requires expensive disposal. Here, the sulfonate acts as a carrier that is regenerated at the end of the reaction cycle. This closed-loop capability means that the effective consumption of the sulfur-containing reagent is negligible after the initial charge, leading to substantial raw material savings over time. Furthermore, the elimination of volatile organic compounds (VOCs) like dimethyl sulfide reduces the need for expensive scrubbing systems and specialized storage tanks, lowering capital expenditure for facility upgrades. The simplified workup procedure, which relies on basic phase separation rather than high-vacuum distillation, also translates to lower energy consumption and shorter batch cycle times, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The ability to recycle the sulfonate precursor significantly lowers the variable cost per kilogram of the final epoxide product. By recovering the sulfonate from the aqueous layer and reusing it in subsequent batches, manufacturers can decouple production volume from raw material procurement for this specific component. Additionally, the use of inexpensive bases like sodium hydroxide and common solvents like toluene or DMSO ensures that operating costs remain competitive. The removal of toxic reagents also reduces the hidden costs associated with regulatory compliance, waste disposal fees, and occupational health monitoring, contributing to a leaner and more profitable operational model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the stability and ease of handling of the new reagents. Unlike dimethyl sulfide, which requires pressurized cylinders or低温 storage due to its volatility, the sulfonate salts are stable solids or high-boiling liquids that can be stored in standard containers. This simplifies logistics, reduces shipping hazards, and minimizes the risk of supply disruptions caused by transportation restrictions on hazardous materials. Moreover, the robustness of the synthesis allows for flexible production scheduling, as the intermediates do not degrade rapidly under ambient conditions. This stability ensures a consistent supply of high-quality intermediates, enabling downstream customers to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exothermic runaway risks associated with volatile sulfides. The reaction conditions are well-defined and manageable within standard stainless steel reactors. Environmentally, the process aligns with green chemistry principles by minimizing waste generation and avoiding the release of noxious gases. The aqueous waste stream, containing the recovered sulfonate, is non-toxic and can be treated or recycled easily, reducing the environmental footprint of the facility. This compliance with stringent environmental standards future-proofs the manufacturing site against tightening regulations, ensuring long-term operational viability and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfo-containing sulfur ylide technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, safety profiles, and process flexibility. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using sulfo-containing sulfur ylides over traditional dimethyl sulfide?
A: Traditional dimethyl sulfide is toxic, volatile, and has a foul odor, posing significant safety and environmental hazards. The sulfo-containing sulfur ylide described in patent CN111116441A is non-volatile, has low irritation, and is water-soluble, which greatly simplifies purification and allows for the recycling of the sulfonate precursor.
Q: Can the sulfonate precursor be recycled in this process?
A: Yes, a key feature of this invention is the recyclability of the raw material. After the epoxidation reaction, the generated sulfonate remains in the aqueous layer due to its high water solubility. It can be recovered via reduced pressure distillation and reused for the synthesis of the sulfur ylide, significantly reducing raw material consumption.
Q: What are the typical reaction conditions for the epoxidation step?
A: The epoxidation reaction is typically conducted in solvents such as dimethyl sulfoxide, toluene, or DMF at moderate temperatures ranging from 40°C to 60°C. The reaction utilizes a basic substance like sodium hydroxide or potassium carbonate and generally completes within 1 to 7 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfo-Containing Sulfur Ylide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic routes like the one described in CN111116441A for producing high-value epoxy intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. Whether you require custom synthesis of the sulfonate precursor or the final epoxide derivative, our infrastructure is designed to meet the demanding requirements of the global pharmaceutical and fine chemical markets.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this route can optimize your bill of materials. Please contact us to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules. Together, we can drive efficiency and sustainability in your supply chain while maintaining the highest standards of quality and reliability.
