Advanced Catalyst-Free Synthesis of Spiro Cyclopropane Indenediones for Pharmaceutical Applications
Advanced Catalyst-Free Synthesis of Spiro Cyclopropane Indenediones for Pharmaceutical Applications
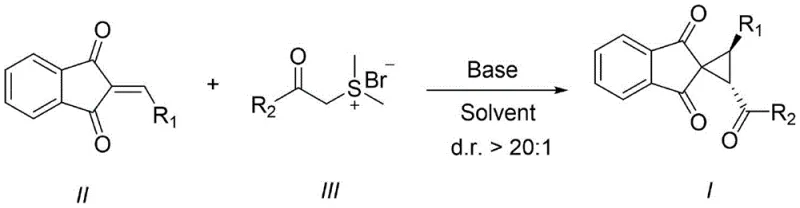
The pharmaceutical and agrochemical industries continuously demand novel scaffolds that offer enhanced metabolic stability and bioactivity, driving the need for efficient synthetic methodologies. Patent CN113264816B introduces a groundbreaking approach for constructing spiro[cyclopropane-1,2'-indene]-1',3'-diketone derivatives, a privileged structural motif found in numerous bioactive natural products and drug candidates. This technology leverages a highly diastereoselective [2+1] cycloaddition between substituted 2-ethylidene-1H-indene-1,3(2H)-diones and sulfur ylides, eliminating the need for complex transition metal catalysts. By utilizing a simple base-mediated protocol, this invention addresses critical pain points in modern organic synthesis, such as catalyst toxicity, high costs, and difficult purification processes. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, this method represents a significant leap forward in process chemistry, offering a robust pathway to high-value spirocyclic cores with exceptional stereocontrol.
![General chemical structure of spiro[cyclopropane-1,2'-indene]-1',3'-diketone derivatives showing variable R1 and R2 substituents](/insights/img/spiro-cyclopropane-indenedione-synthesis-supplier-pharma-20260306144246-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of cyclopropane rings, particularly spiro-fused systems, has relied heavily on classical strategies involving diazo compounds or carbene insertion reactions, which often pose significant safety hazards due to the explosive nature of diazo precursors. Previous literature, such as the work reported by Roy, utilized sulfur ylides for similar transformations but suffered from notoriously low diastereoselectivity, resulting in complex mixtures of isomers that are costly and time-consuming to separate. Furthermore, alternative stereoselective approaches, like those developed by Maleki, necessitated the use of sophisticated nanomagnetic organocatalysts functionalized with L-proline, which introduce substantial complexity to the supply chain and increase the overall cost of goods sold (COGS). These conventional limitations create bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, as the removal of heavy metal residues or complex organic catalysts requires additional downstream processing steps that reduce overall yield and throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN113264816B utilizes a streamlined, catalyst-free strategy that relies solely on the intrinsic reactivity of sulfur ylides in the presence of a mild inorganic or organic base. This novel approach facilitates a formal [2+1] cyclization that proceeds with remarkable efficiency, achieving diastereomeric ratios (d.r.) greater than 20:1 without the need for chiral ligands or expensive metal complexes. The reaction operates under mild thermal conditions, typically ranging from 0°C to 100°C, allowing for precise control over the reaction kinetics and minimizing the formation of unwanted byproducts. By simplifying the reaction matrix to just the substrate, the ylide, a base, and a solvent, this method drastically reduces the environmental footprint and operational complexity, positioning it as an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The ability to access these complex spiro-architectures through such a direct route empowers medicinal chemists to rapidly explore structure-activity relationships (SAR) with diverse substitution patterns.
Mechanistic Insights into Base-Mediated Sulfur Ylide Cycloaddition
The core of this technological breakthrough lies in the precise mechanistic orchestration of the sulfur ylide attack on the electron-deficient olefinic bond of the indanedione derivative. Upon deprotonation by the base, the sulfur ylide generates a nucleophilic carbanion species that selectively attacks the exocyclic double bond of the 2-ethylidene-1H-indene-1,3(2H)-dione. This initial Michael-type addition is followed by an intramolecular nucleophilic substitution where the displaced sulfide leaving group facilitates the closure of the strained three-membered cyclopropane ring. The high diastereoselectivity observed (d.r. ≥ 20:1) suggests a highly ordered transition state, likely governed by steric interactions between the bulky substituents on the ylide and the indanedione framework, which favors the formation of the thermodynamically more stable trans-configured spiro-isomer. This level of stereocontrol is critical for high-purity pharmaceutical intermediates, as it minimizes the burden on chiral separation technologies and ensures consistent quality in the final active pharmaceutical ingredient (API).
Furthermore, the impurity profile of this reaction is exceptionally clean due to the absence of metal catalysts that often lead to trace metal contamination or catalyze side reactions such as polymerization or over-alkylation. The broad substrate tolerance demonstrated in the patent examples indicates that the electronic nature of the substituents (R1 and R2) has a minimal detrimental effect on the reaction outcome, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like nitro, cyano, and halogens. This robustness implies that the reaction mechanism is resilient to variations in electronic density, making it a versatile platform for synthesizing a wide library of analogs. For process chemists, understanding this mechanism allows for the rational optimization of reaction parameters, such as base strength and solvent polarity, to further enhance yields and selectivity during reducing lead time for high-purity spirocyclic compounds.
How to Synthesize Spiro[cyclopropane-1,2'-indene]-1',3'-diketone Efficiently
The practical implementation of this synthesis is designed for operational simplicity, requiring standard laboratory equipment and readily available reagents. The general procedure involves dissolving the substituted 2-ethylidene-indanedione and the sulfur ylide precursor in a polar aprotic or protic solvent, followed by the addition of a stoichiometric amount of base such as cesium carbonate or triethylamine. The reaction mixture is then stirred at ambient or slightly elevated temperatures until conversion is complete, as monitored by TLC or HPLC. Detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup protocols optimized for maximum recovery, are outlined in the guide below.
- Combine substituted 2-ethylidene-1H-indene-1,3(2H)-dione and sulfur ylide derivative in a suitable organic solvent.
- Add an appropriate base (e.g., Cs2CO3) and stir the mixture at a controlled temperature between 0°C and 100°C.
- Upon completion, quench with water or brine, extract with organic solvent, dry, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this catalyst-free synthesis route offers profound advantages for supply chain stability and cost management. Traditional methods relying on precious metal catalysts or custom-synthesized organocatalysts introduce significant volatility into the supply chain, as these reagents are often subject to price fluctuations and long lead times. By eliminating these dependencies, manufacturers can secure a more predictable and resilient supply of critical intermediates. The simplified reaction design also translates to reduced operational expenditures, as there is no need for specialized equipment to handle hazardous diazo compounds or expensive filtration systems to remove metal residues. This efficiency directly contributes to cost reduction in fine chemical manufacturing, allowing companies to allocate resources towards other critical areas of drug development.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and complex chiral ligands removes a major cost driver from the bill of materials. Additionally, the high diastereoselectivity reduces the need for costly chromatographic separations or recrystallizations to remove isomeric impurities, thereby increasing the overall mass balance and yield of the process. The use of inexpensive inorganic bases and common organic solvents further lowers the input costs, making the production of these high-value spiro-intermediates economically viable on a multi-ton scale.
- Enhanced Supply Chain Reliability: The starting materials, specifically the substituted indanediones and sulfur ylides, are derived from commodity chemicals that are widely available from multiple global suppliers. This diversification of the raw material base mitigates the risk of single-source dependency and ensures continuous production even during market disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, reducing the rate of batch failures and ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the waste stream treatment, aligning the process with increasingly stringent environmental regulations regarding metal discharge. The straightforward workup procedure, involving simple aqueous quenching and extraction, is easily adaptable to large-scale reactors without the need for complex engineering controls. This scalability ensures that the technology can seamlessly transition from gram-scale R&D to kilogram and ton-scale commercial production, supporting the growing demand for these bioactive scaffolds in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What is the primary advantage of this sulfur ylide method over traditional cyclopropanation?
A: Unlike traditional methods requiring complex metal catalysts or diazo compounds, this patent describes a catalyst-free, base-mediated approach that achieves exceptional diastereoselectivity (dr > 20:1) under mild conditions.
Q: What is the substrate scope for R1 and R2 groups in this synthesis?
A: The method demonstrates broad adaptability, tolerating diverse substituents including alkyl, aryl, heteroaryl, halogenated phenyls, and electron-withdrawing groups like nitro and cyano, ensuring versatility for drug discovery.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the elimination of expensive transition metal catalysts and the use of simple workup procedures (filtration and extraction) significantly simplify the process, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[cyclopropane-1,2'-indene]-1',3'-diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN113264816B for accelerating drug discovery programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the highest standards of quality and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of diastereomeric ratios and residual solvent levels, guaranteeing that every batch of spiro[cyclopropane-1,2'-indene]-1',3'-diketone delivered is ready for immediate use in sensitive biological assays or subsequent synthetic steps.
We invite pharmaceutical and agrochemical companies to leverage our expertise to optimize their supply chains and reduce development costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, identifying opportunities to streamline your synthesis of complex intermediates. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your journey from bench to market with speed and reliability.
