Advanced Green Synthesis of Quinoxaline Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Green Synthesis of Quinoxaline Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize critical heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN111072577B, which discloses a novel green synthesis method for efficiently constructing quinoxaline derivatives. This technology leverages a transition metal-catalyzed carbene insertion and cyclization reaction, utilizing pure water as the solvent and sulfur ylides as the carbene donor. Unlike traditional methods that rely on hazardous reagents and toxic organic media, this approach offers a mild, rapid, and environmentally friendly route to the quinoxaline parent ring. For R&D directors and procurement managers, this represents a paradigm shift towards safer and more economical manufacturing processes that do not compromise on yield or purity.
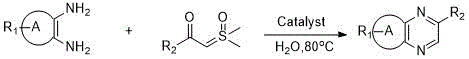
The core innovation lies in the strategic replacement of unstable diazo compounds with stable sulfur ylides, coupled with the use of water as a benign reaction medium. This combination addresses multiple pain points simultaneously: it enhances operational safety, reduces raw material costs, and simplifies downstream processing. The reaction proceeds under relatively mild conditions, typically at 80°C, which minimizes energy consumption and thermal degradation of sensitive functional groups. By adopting this methodology, manufacturers can achieve high-purity quinoxaline intermediates essential for drug synthesis, agrochemicals, and organic materials, while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has relied heavily on the condensation of o-phenylenediamines with dicarbonyl compounds, alpha-hydroxy ketones, or alpha-bromo ketones. While these classical routes are well-documented, they suffer from significant drawbacks that hinder their scalability and economic viability. The starting materials, particularly specialized dicarbonyl compounds, can be difficult to source and expensive to produce. Furthermore, these reactions often require harsh conditions, such as strong acids or high temperatures, which can lead to side reactions and complex impurity profiles. Perhaps most critically, conventional methods frequently necessitate the use of toxic organic solvents, creating substantial environmental liabilities and increasing the cost of waste management and solvent recovery systems.
The Novel Approach
In stark contrast, the method described in patent CN111072577B utilizes a transition metal-catalyzed carbene insertion strategy that fundamentally alters the reaction landscape. By employing sulfur ylides as carbene precursors, the process bypasses the instability and safety risks associated with diazo compounds. The use of water as the sole solvent not only eliminates the need for volatile organic compounds but also leverages the hydrophobic effect to potentially accelerate the reaction rate. This novel approach is characterized by its simplicity, mild reaction conditions, and broad substrate scope. It effectively solves the problems of lengthy synthetic steps, difficult raw material preparation, and high environmental pollution, offering a streamlined pathway that is both economically and ecologically superior to traditional condensation methods.
Mechanistic Insights into Transition Metal-Catalyzed Carbene Insertion
The success of this green synthesis hinges on the precise orchestration of transition metal catalysis to facilitate carbene insertion into N-H bonds followed by cyclization. The mechanism involves the generation of a metal-carbene species from the sulfur ylide precursor, which then undergoes insertion into the amine functionality of the o-diamine. This step is critical for forming the new C-N bonds that construct the quinoxaline ring system. The patent highlights the versatility of the catalyst system, listing a wide array of effective transition metals including Iridium, Rhodium, Ruthenium, Cobalt, and Palladium complexes. For instance, catalysts such as bis(1,5-cyclooctadiene)iridium(I) chloride dimer and dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer have shown excellent efficacy. This mechanistic flexibility allows chemists to tune the electronic and steric properties of the catalyst to match specific substrate requirements, ensuring high conversion rates and selectivity.
From an impurity control perspective, the mild aqueous conditions play a pivotal role in maintaining product integrity. High temperatures and strong acids, common in older methods, often promote polymerization or decomposition of sensitive intermediates. By operating at 80°C in water, the reaction environment is sufficiently gentle to preserve delicate functional groups such as methoxy, chloro, or alkyl substituents on the aromatic rings. The subsequent workup involves a simple extraction with dichloromethane followed by silica gel column chromatography, which effectively removes metal residues and unreacted starting materials. This results in a final product with a clean impurity profile, reducing the burden on downstream purification units and ensuring that the material meets the stringent quality standards required for pharmaceutical applications.
How to Synthesize Quinoxaline Derivatives Efficiently
The operational protocol for this synthesis is designed for simplicity and reproducibility, making it highly suitable for both laboratory optimization and industrial scale-up. The process begins with the sequential addition of the aromatic o-diamine, the sulfur ylide compound, and the selected transition metal catalyst into a clean reactor containing pure water. The mixture is then heated in an oil bath at 80°C and stirred for approximately 24 hours to ensure complete conversion. Following the reaction, the product is extracted using dichloromethane, the solvent is removed under reduced pressure, and the crude residue is purified via standard chromatographic techniques. Detailed standardized synthesis steps and specific parameter optimizations are provided in the guide below.
- Charge aromatic o-diamine, sulfur ylide compound, and transition metal catalyst into a reactor with pure water.
- Heat the reaction mixture in an oil bath at 80°C and stir continuously for 24 hours to ensure complete conversion.
- Extract the product with dichloromethane, remove solvent under reduced pressure, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this green synthesis method translates into tangible strategic benefits beyond mere technical feasibility. The shift from toxic organic solvents to water significantly reduces the cost of goods sold (COGS) by eliminating expensive solvent purchase and recovery cycles. Moreover, the use of stable sulfur ylides instead of hazardous diazo compounds lowers insurance premiums and safety compliance costs. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or extensive rework. This reliability is crucial for maintaining uninterrupted supply chains in the fast-paced pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The elimination of toxic organic solvents and the use of readily available, stable sulfur ylides drastically simplify the production process. By removing the need for complex solvent recovery infrastructure and hazardous waste disposal protocols, manufacturers can achieve substantial cost savings. The mild reaction conditions also reduce energy consumption, further contributing to a lower overall production cost structure compared to traditional high-temperature or high-pressure methods.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically aromatic o-diamines and sulfur ylides, are commercially accessible and stable for long-term storage. This stability mitigates the risks associated with the degradation of sensitive reagents during transport or warehousing. Consequently, manufacturers can maintain leaner inventory levels without compromising production readiness, leading to a more agile and responsive supply chain capable of meeting fluctuating market demands.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes it inherently safer and easier to scale from kilogram to multi-ton quantities. Water acts as an excellent heat sink, reducing the risk of thermal runaway incidents that are common in exothermic organic reactions. Furthermore, the process aligns perfectly with global green chemistry initiatives, facilitating easier regulatory approval and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel quinoxaline synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What are the safety advantages of using sulfur ylides over diazo compounds?
A: Sulfur ylides offer significantly improved stability and safety profiles compared to traditional diazo compounds, which are often unstable, difficult to store, and potentially explosive. This substitution eliminates the hazards associated with handling sensitive diazo precursors, making the process safer for large-scale industrial operations.
Q: How does the use of water as a solvent impact the environmental footprint?
A: Replacing toxic organic solvents with pure water drastically reduces volatile organic compound (VOC) emissions and simplifies waste treatment protocols. This aligns with green chemistry principles, lowering the environmental burden and reducing the costs associated with solvent recovery and hazardous waste disposal.
Q: Which transition metal catalysts are compatible with this synthesis method?
A: The method demonstrates broad compatibility with various transition metal catalysts including Iridium, Rhodium, Ruthenium, Cobalt, and Palladium complexes. This flexibility allows manufacturers to optimize catalyst selection based on cost, availability, and specific substrate requirements to maximize yield and efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of transition metal-catalyzed reactions, ensuring that every batch of quinoxaline derivatives meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify product identity and purity, guaranteeing that our clients receive materials that are ready for immediate use in downstream drug synthesis or material science applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your time to market.
