Scalable Synthesis of Montelukast Sodium: A Breakthrough in High-Purity API Intermediate Manufacturing
Scalable Synthesis of Montelukast Sodium: A Breakthrough in High-Purity API Intermediate Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for producing critical active pharmaceutical ingredients (APIs) and their intermediates. A recent technological advancement detailed in patent CN111892535B introduces a highly efficient synthesis method for Montelukast Sodium, a potent leukotriene receptor antagonist widely used in asthma management. This innovation addresses longstanding challenges in the manufacturing of this complex molecule, specifically targeting the issues of low yield, excessive purification steps, and the reliance on costly reagents found in legacy processes. By leveraging a streamlined two-step reaction sequence involving the coupling of specific chiral intermediates followed by a controlled hydrolysis and salification, this method delivers exceptional purity profiles exceeding 99.80% and yields surpassing 93.5%. For global procurement teams and R&D directors, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates while drastically reducing the environmental footprint associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Montelukast Sodium has been plagued by inefficient synthetic strategies that hinder scalability and inflate production costs. Early methodologies, such as those disclosed in patent EP0737186B1, relied heavily on complex protection group chemistry, necessitating multiple steps to selectively protect diol compounds before coupling and subsequent deprotection. This approach not only extends the manufacturing timeline but also introduces significant difficulties in purifying intermediate compounds, leading to cumulative yield losses. Furthermore, alternative routes reported in literature, such as US2005107612, attempted to utilize Grignard reagents and amine salt formations to bypass certain steps; however, these methods suffered from abysmal total yields, often reported to be less than 20%, rendering them commercially unviable for large-scale operations. Additionally, some modern attempts employed ionic liquids as reaction media, as seen in patents like CN101558042; while chemically feasible, ionic liquids are notoriously expensive, hygroscopic, and difficult to recover, creating harsh operating conditions and troublesome post-treatment workflows that are ill-suited for green manufacturing standards.
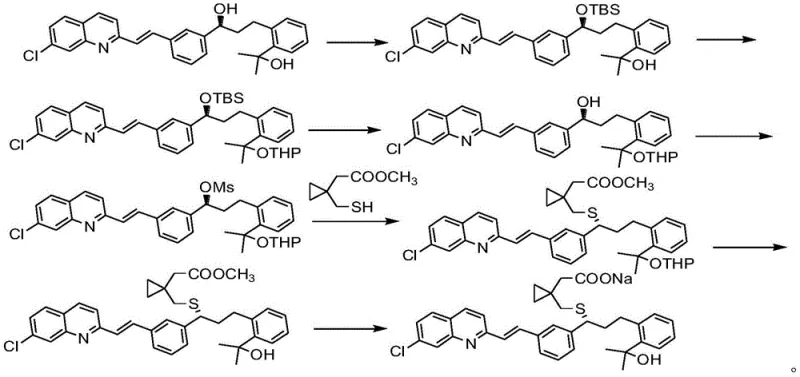
The Novel Approach
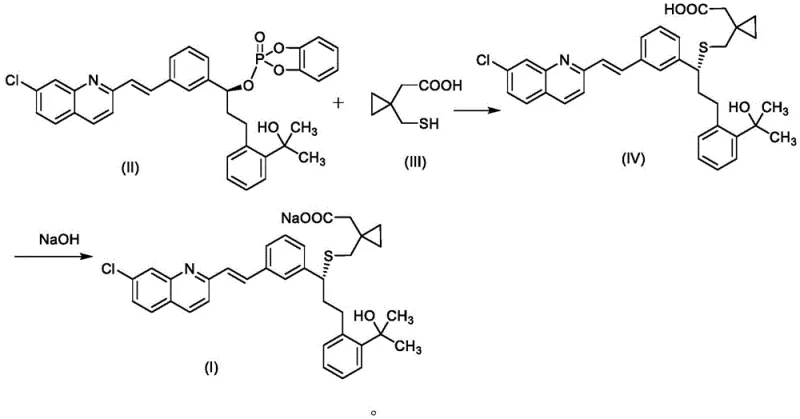
In stark contrast to these cumbersome legacy processes, the novel methodology outlined in CN111892535B offers a paradigm shift towards simplicity and efficiency. This innovative route bypasses the need for extensive protecting group manipulations and avoids the use of prohibitively expensive ionic liquids entirely. Instead, it employs a direct coupling strategy between a phosphoryl-activated chiral alcohol intermediate (Formula II) and a mercapto-acid derivative (Formula III) under mild basic conditions. The reaction is conducted in common, recyclable organic solvents like tetrahydrofuran, facilitating easier workup and solvent recovery. The subsequent conversion to the final sodium salt is achieved through a straightforward hydrolysis and neutralization step using sodium hydroxide in methanol. This streamlined approach not only condenses the synthetic timeline but also inherently improves the impurity profile by minimizing the number of unit operations where side reactions could occur, thereby establishing a new benchmark for the cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into the Coupling and Hydrolysis Strategy
The core of this technological breakthrough lies in the precise control of the nucleophilic substitution reaction between the electrophilic phosphate ester intermediate and the thiol nucleophile. Under the protection of an inert atmosphere, typically nitrogen or argon, the reaction is initiated by dissolving the chiral phosphate intermediate (Formula II) in an organic solvent such as tetrahydrofuran alongside a carefully selected organic base system. The patent specifies a synergistic mixture of bases, preferably 2,6-lutidine and N,N-diisopropylethylamine, which serves to scavenge the acidic byproducts generated during the coupling without promoting unwanted elimination reactions. The reaction temperature is critically maintained between -25°C and -15°C, with an optimal range of -20°C to -15°C, to ensure high stereoselectivity and prevent the racemization of the chiral center. This low-temperature regime is essential for preserving the optical purity of the final product, a critical quality attribute for API intermediates intended for human consumption.
Following the successful formation of the thioether linkage to generate intermediate (Formula IV), the process transitions to the hydrolysis and salification stage. The intermediate is dissolved in methanol, and sodium hydroxide is introduced at a controlled temperature of 10°C to 30°C. This step cleaves the ester moiety and simultaneously forms the stable sodium salt. A crucial aspect of the mechanism here is the in-process purification strategy; the addition of drying agents like anhydrous sodium sulfate and decolorizing agents like activated carbon directly into the reaction mixture allows for the removal of trace metal ions and colored impurities prior to crystallization. The final isolation involves concentrating the methanol solution and inducing crystallization by the dropwise addition of n-hexane at low temperatures (0°C to 5°C). This antisolvent crystallization technique is highly effective at excluding structurally similar impurities, resulting in the reported exceptional purity levels of greater than 99.80%.
How to Synthesize Montelukast Sodium Efficiently
The synthesis protocol described in the patent provides a clear, reproducible pathway for manufacturing Montelukast Sodium with industrial-grade consistency. The process is designed to be robust against minor variations in scale, making it ideal for both pilot plant trials and full commercial production. The key to success lies in the strict adherence to the specified molar ratios, particularly the 1:1.2 ratio between the phosphate intermediate and the thiol reactant, and the precise temperature controls during the exothermic coupling phase. Operators must ensure that the inert gas protection is maintained throughout to prevent oxidation of the thiol species, which could lead to disulfide impurities. For a comprehensive understanding of the exact operational parameters, including stirring speeds, addition rates, and specific drying times, please refer to the detailed standardized synthesis steps provided in the technical guide below.
- Coupling Reaction: Dissolve compound (II) and organic base in THF, cool to -20 to -15°C, add compound (III) solution, and stir to obtain intermediate (IV).
- Hydrolysis and Salification: Dissolve intermediate (IV) in methanol, add NaOH at 10-20°C, treat with activated carbon, concentrate, and crystallize with n-hexane to obtain pure Montelukast Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere chemical elegance. The elimination of expensive and hard-to-source reagents, such as ionic liquids or specialized protecting group reagents, directly impacts the bill of materials, leading to substantial cost savings in the overall production budget. Furthermore, the reduction in the number of synthetic steps from multi-stage sequences to a concise two-step process significantly lowers the operational expenditure (OpEx) related to labor, energy consumption, and equipment occupancy time. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, ensuring a more reliable supply of high-purity pharmaceutical intermediates without the bottlenecks associated with complex purification trains.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of ionic liquids and complex protecting group chemistries from the process flow. By utilizing commodity chemicals like tetrahydrofuran, methanol, and sodium hydroxide, the raw material costs are drastically minimized compared to prior art methods. Additionally, the high yield of over 93.5% for the final sodium salt means that less starting material is wasted, improving the overall atom economy and reducing the cost per kilogram of the finished API intermediate. This economic efficiency is further compounded by the simplified workup procedure, which requires fewer solvent exchanges and distillation cycles, thereby lowering utility costs and waste disposal fees associated with hazardous solvent streams.
- Enhanced Supply Chain Reliability: From a sourcing perspective, this method relies on readily available bulk chemicals that are not subject to the same supply constraints as specialized catalysts or exotic reagents. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require ultra-low temperature cryogenics beyond standard chillers, ensures that production can be sustained across multiple manufacturing sites globally. This geographic flexibility mitigates the risk of supply disruptions caused by regional logistical issues or single-source supplier dependencies. Consequently, pharmaceutical companies can secure a more stable and continuous supply of Montelukast Sodium, safeguarding their downstream drug formulation schedules against raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as liquid-liquid extraction, filtration, and crystallization that are well-understood in chemical engineering. The avoidance of heavy metal catalysts and the use of recyclable solvents align perfectly with increasingly stringent environmental regulations and green chemistry principles. The simplified effluent profile, devoid of persistent ionic liquid residues or complex organic byproducts, facilitates easier wastewater treatment and reduces the environmental compliance burden. This makes the technology not only commercially attractive but also socially responsible, appealing to stakeholders who prioritize sustainable manufacturing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical benefits and operational specifics of the new route. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this manufacturing method.
Q: What are the yield and purity advantages of this new synthesis method?
A: The method described in patent CN111892535B achieves a Montelukast acid yield of over 98% with purity higher than 99.7%, and a final Montelukast Sodium yield of over 93.5% with purity exceeding 99.80%.
Q: Why is this process more cost-effective than ionic liquid methods?
A: Unlike prior art using ionic liquids which are expensive and moisture-sensitive, this novel route utilizes standard organic solvents like tetrahydrofuran and methanol, significantly reducing raw material costs and simplifying post-treatment.
Q: Does this method require complex protecting group strategies?
A: No, the process eliminates the need for multiple protection and deprotection steps found in conventional routes, streamlining the synthesis into just two main reaction stages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume manufacturing. We are committed to delivering Montelukast Sodium and other complex pharmaceutical intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic advantages tailored to your volume requirements. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering high-quality, cost-effective solutions for your API needs.
