Advanced Synthesis of Montelukast Sodium Intermediate for Commercial Scale-up
Advanced Synthesis of Montelukast Sodium Intermediate for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of critical asthma and allergy medications, with Montelukast Sodium standing as a cornerstone therapeutic agent. The patent CN102442947B discloses a highly efficient preparation method for a key Montelukast Sodium intermediate, addressing longstanding challenges in stereochemical control and process complexity. This technical breakthrough centers on a novel nucleophilic substitution strategy that operates under remarkably mild conditions, specifically between 0°C and 30°C, utilizing inert gas protection to maintain integrity. By shifting away from the cumbersome multi-step protection and deprotection sequences characteristic of legacy syntheses, this approach offers a streamlined pathway that is inherently more suitable for industrialized production. For R&D directors and supply chain leaders, understanding this shift is crucial, as it represents a move towards higher atom economy and reduced operational overhead in the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Montelukast intermediates has been plagued by excessive step counts and the reliance on chemically demanding protecting group strategies. As illustrated in the traditional Merck route (CN1061407A), the process necessitates the installation of bulky silyl groups like tert-butyldimethylsilyl (TBS) and acid-labile tetrahydropyranyl (THP) moieties to mask reactive hydroxyl functionalities. 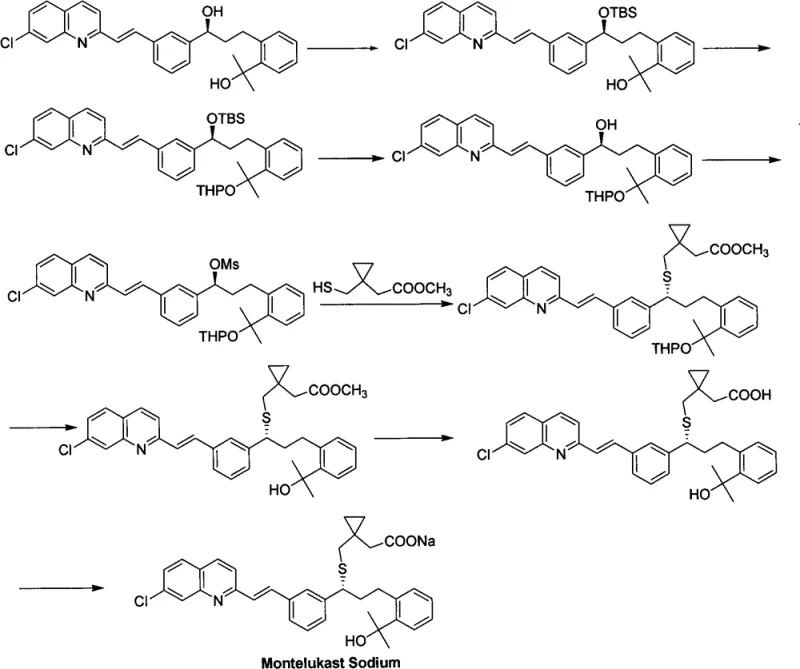 These additional synthetic operations not only extend the overall lead time but also introduce significant opportunities for yield loss at each stage. Furthermore, the removal of these protecting groups often requires harsh acidic or fluoride-based conditions, which can compromise the optical purity of the chiral centers and generate difficult-to-remove impurities. From a procurement perspective, the reagents required for these protection steps add unnecessary cost layers, while the extended reaction timelines create bottlenecks in supply continuity, making the conventional approach less viable for high-volume commercial demands.
These additional synthetic operations not only extend the overall lead time but also introduce significant opportunities for yield loss at each stage. Furthermore, the removal of these protecting groups often requires harsh acidic or fluoride-based conditions, which can compromise the optical purity of the chiral centers and generate difficult-to-remove impurities. From a procurement perspective, the reagents required for these protection steps add unnecessary cost layers, while the extended reaction timelines create bottlenecks in supply continuity, making the conventional approach less viable for high-volume commercial demands.
The Novel Approach
In stark contrast, the methodology outlined in CN102442947B circumvents these inefficiencies by employing a direct nucleophilic substitution on a chloro-intermediate (Compound III). This innovative route eliminates the need for the aforementioned TBS and THP protecting groups entirely, thereby collapsing multiple synthetic steps into a more concise sequence. The reaction proceeds by coupling the chloro-species with a nucleophilic thiol reagent (Compound IV) in the presence of a mild base, such as cesium carbonate, within a polar aprotic solvent like DMF. This strategic simplification results in a single major product with minimal side reactions, drastically easing the purification burden. For manufacturing teams, this translates to a process that is not only faster but also more predictable, ensuring consistent quality output without the variability associated with complex protection chemistry.
Mechanistic Insights into Nucleophilic Substitution and Stereocontrol
The core of this technological advancement lies in the precise control of the nucleophilic substitution mechanism, which is critical for maintaining the stereochemical integrity of the final API. The reaction involves the displacement of a chloride leaving group on the chiral benzylic position of Compound III by the sulfur nucleophile of Compound IV. The selection of cesium carbonate as the base is particularly astute; its moderate basicity is sufficient to activate the thiol nucleophile without inducing elimination side reactions or epimerization of the sensitive chiral center. The use of DMF as the solvent further enhances the reaction kinetics by stabilizing the transition state through solvation of the cationic species, facilitating a smooth SN2-type displacement. This mechanistic elegance ensures that the optical purity, reported with an ee value of approximately 92% in the examples, is preserved throughout the transformation, which is a paramount requirement for regulatory compliance in pharmaceutical manufacturing.
Furthermore, the precursor synthesis of Compound III itself demonstrates a refined approach to halogenation that supports the overall efficiency of the route. Instead of relying on aggressive halogenating agents that might degrade the molecule, the patent describes a two-step sequence involving sulfonylation followed by halogenation using lithium chloride or tetrabutylammonium chloride. This method allows for the gentle conversion of the secondary alcohol to the corresponding chloride under controlled temperatures ranging from -10°C to 30°C. By avoiding extreme thermal conditions during the activation of the alcohol, the process minimizes the formation of regioisomers and other structural impurities. This level of control over the impurity profile is essential for R&D directors who must validate that the intermediate meets stringent specifications before proceeding to the final drug substance synthesis, ensuring a robust and defensible regulatory filing.
How to Synthesize Montelukast Sodium Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize the benefits of the novel chemistry. The process begins with the preparation of the chloro-intermediate, followed by the critical coupling step with the cyclopropyl thiol ester. Operators must maintain an inert atmosphere, typically using nitrogen or argon, to prevent oxidation of the thiol species, which could lead to disulfide byproducts. The temperature control during the addition of the base and the subsequent stirring period is vital; keeping the reaction between 0°C and 30°C ensures optimal reaction rates while suppressing potential degradation pathways. Detailed standard operating procedures regarding the stoichiometric ratios of the base (preferably 1.5 equivalents) and the solvent volume (approximately 12.5 mL/g of substrate) are essential for reproducibility. For a comprehensive guide on the specific experimental execution and workup protocols, please refer to the standardized synthesis steps provided below.
- Preparation of the chloro-intermediate (Compound III) via sulfonylation of the secondary alcohol followed by halogenation using lithium chloride or tetrabutylammonium chloride.
- Execution of the nucleophilic substitution reaction between Compound III and the thiol-containing cyclopropyl ester (Compound IV) in DMF solvent.
- Utilization of cesium carbonate as the base at controlled temperatures (0-30°C) to ensure high optical purity and yield before final workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic advantages that extend beyond mere technical feasibility. The primary benefit is the significant reduction in manufacturing costs driven by the elimination of expensive protecting group reagents and the associated waste disposal fees. By shortening the synthetic sequence, the facility can achieve higher throughput with the same existing infrastructure, effectively increasing capacity without capital expenditure. Additionally, the simplified purification process, which relies on standard extraction and crystallization rather than complex chromatography, reduces solvent consumption and processing time. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands for Montelukast Sodium with greater agility and lower variable costs per kilogram.
- Cost Reduction in Manufacturing: The economic impact of removing protecting group chemistry cannot be overstated, as it eradicates the cost of purchasing silyl chlorides and THP reagents along with the labor required for their handling. Moreover, the high yield and single-product nature of the reaction minimize the loss of valuable chiral starting materials, ensuring that the cost of goods sold (COGS) is optimized. The ability to use readily available inorganic bases like cesium carbonate instead of specialized organometallic reagents further contributes to substantial cost savings in raw material procurement. This lean manufacturing approach allows for a more competitive pricing structure in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. The reagents involved, such as DMF and lithium chloride, are commodity chemicals with stable global supply lines, mitigating the risk of raw material shortages that often plague specialty reagent-dependent processes. Furthermore, the mild reaction temperatures reduce the energy load on the manufacturing plant, decreasing dependence on intensive cooling or heating utilities and enhancing overall operational stability. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as the exothermic nature of the reaction is manageable even in large reactors due to the mild temperature window. The reduction in the number of synthetic steps directly correlates to a lower E-factor (environmental factor), meaning less waste is generated per unit of product, which simplifies compliance with increasingly strict environmental regulations. The use of DMF, while requiring careful handling, allows for efficient solvent recovery systems that can be integrated into existing waste management infrastructures. Consequently, this method supports sustainable manufacturing goals while enabling the seamless transition from pilot-scale batches to multi-ton commercial production campaigns.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their supply chain, we have compiled answers to common inquiries based on the patent data. These questions address the critical aspects of stereochemistry, process safety, and scalability that are often top of mind for process chemists and engineering leads. Understanding these nuances helps in making informed decisions regarding technology transfer and vendor qualification. The following insights are derived directly from the experimental data and claims of CN102442947B, ensuring accuracy and relevance to your specific manufacturing context.
Q: How does this novel method improve optical purity compared to traditional routes?
A: By utilizing mild reaction temperatures (0-30°C) and specific alkali metal carbonates like cesium carbonate, the process minimizes racemization risks often seen in harsher conventional conditions, achieving ee values around 92%.
Q: What are the scalability advantages of avoiding protecting groups?
A: Eliminating the installation and removal of bulky protecting groups such as TBS and THP significantly reduces the number of unit operations, lowers solvent consumption, and simplifies downstream purification, making large-scale production more efficient.
Q: Is the solvent system suitable for industrial waste management?
A: Yes, the process primarily utilizes DMF, which is a standard industrial solvent with established recovery and recycling protocols, thereby reducing the environmental footprint compared to complex multi-solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving medications like Montelukast Sodium. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. By leveraging the efficient synthesis methods described in recent patents, we can offer a supply solution that balances cost-effectiveness with the highest standards of pharmaceutical quality, positioning us as a strategic partner for your long-term growth.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to review specific COA data for batch validation, we are ready to provide the support you need. Please reach out to us today to request route feasibility assessments and discover how our advanced manufacturing solutions can enhance your operational efficiency and product competitiveness in the global marketplace.
