Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to bioactive scaffolds. A pivotal development in this domain is documented in Chinese Patent CN113307790B, which discloses a robust preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are critical in the design of next-generation pharmaceuticals and functional materials, particularly in the realm of organic light-emitting diodes (OLEDs) where they serve as potent ligands. The patent introduces a transformative oxidative cyclization strategy that bypasses the limitations of classical synthesis, utilizing a metal-free catalytic system composed of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP). For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process chemistry, offering a route that is not only high-yielding but also operationally simple and environmentally benign.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and complexity. Traditional methodologies often rely on quinoline-2-formic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to arrive at the target molecule. This multi-step approach suffers from a dismal cumulative yield of approximately 17%, rendering it economically unviable for large-scale applications. Furthermore, these conventional routes typically demand severe reaction conditions, including strict anhydrous and oxygen-free environments, which impose heavy burdens on facility infrastructure and operational safety. The reliance on such苛刻 conditions not only drives up energy consumption but also complicates the supply chain by requiring specialized handling and storage of sensitive reagents, ultimately leading to higher production costs and longer lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN113307790B revolutionizes the synthesis by employing readily available 2-methylquinoline and trifluoroethylimide hydrazide as starting materials. This innovative route collapses the synthetic timeline into a single operational unit, eliminating the need for intermediate isolation and purification steps that characterize the older five-step processes. The reaction proceeds efficiently in common organic solvents like DMSO at moderate temperatures between 80°C and 100°C, without the necessity for inert atmosphere protection. By leveraging an oxidative cyclization mechanism promoted by TBAI and TBHP, the process achieves exceptional conversion rates, with specific examples demonstrating yields as high as 97%. This dramatic improvement in efficiency directly translates to substantial cost savings in fine chemical manufacturing, making it an attractive option for commercial scale-up of complex heterocyclic intermediates.
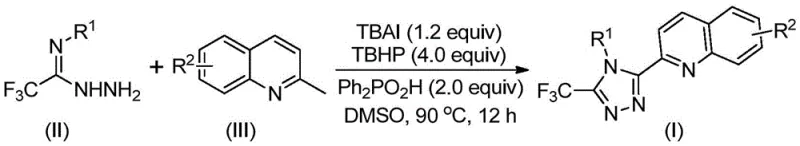
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological breakthrough lies in the elegant interplay between the iodide promoter and the peroxide oxidant. Mechanistically, the tetrabutylammonium iodide acts as a catalyst that facilitates the oxidation of the methyl group on the 2-methylquinoline substrate. In the presence of tert-butyl peroxide, the methyl group is selectively oxidized in situ to generate a reactive 2-quinoline carbaldehyde intermediate. This transient aldehyde species immediately undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution reactions drive the cyclization process, followed by aromatization to yield the final stable 1,2,4-triazole ring system. This cascade reaction is highly atom-economical and avoids the generation of stoichiometric heavy metal waste, aligning perfectly with modern green chemistry principles.
From an impurity control perspective, the absence of transition metal catalysts is a decisive advantage for pharmaceutical applications. Traditional cross-coupling reactions often leave behind trace amounts of palladium, copper, or nickel, which require expensive and time-consuming scavenging steps to meet stringent regulatory limits (e.g., ICH Q3D guidelines). Since this novel method relies solely on organic promoters and oxidants, the resulting crude product is inherently cleaner, significantly reducing the burden on downstream purification processes. The use of diphenylphosphoric acid as an additive further enhances the reaction selectivity, minimizing the formation of side products and ensuring a narrow impurity profile. This level of chemical purity is essential for R&D teams focused on developing drug candidates where even trace impurities can alter biological activity or toxicity profiles.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized molar ratios and reaction parameters defined in the patent. The process is designed to be user-friendly, utilizing standard glassware and heating equipment without the need for specialized gloveboxes or Schlenk lines. The key to success lies in the careful selection of the solvent system, with dimethyl sulfoxide (DMSO) proving to be the superior medium for dissolving both the organic substrates and the ionic catalyst, thereby maximizing reaction kinetics. Operators should monitor the reaction progress via TLC or HPLC to determine the optimal endpoint, typically achieved within 8 to 14 hours depending on the specific substituents on the aromatic rings. The detailed standardized synthesis steps see the guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP) aqueous solution, diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion via oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the target 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The shift from a five-step low-yield process to a one-pot high-yield reaction fundamentally alters the cost structure of the final intermediate. By eliminating multiple unit operations, the process drastically reduces labor costs, solvent consumption, and waste disposal fees. Furthermore, the starting materials, specifically 2-methylquinoline and various substituted aromatic amines used to prepare the hydrazide, are commodity chemicals available in bulk quantities from global suppliers. This abundance ensures a stable supply chain and mitigates the risk of raw material shortages that often plague niche synthetic routes relying on exotic or custom-synthesized precursors.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the elimination of expensive transition metal catalysts and the reduction of synthetic steps. In traditional manufacturing, the cost of palladium or copper catalysts, combined with the ligands and scavengers required for their removal, constitutes a significant portion of the COGS (Cost of Goods Sold). By replacing these with inexpensive organic salts like TBAI and common oxidants like TBHP, the direct material costs are significantly reduced. Additionally, the high reaction yield minimizes the amount of starting material required per kilogram of product, further enhancing the overall process economics and allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Because the process does not require strict anhydrous or oxygen-free environments, it can be performed in standard reactors without the need for extensive nitrogen purging or drying of solients. This operational simplicity reduces the likelihood of batch failures due to environmental contamination, ensuring consistent production output. Moreover, the use of DMSO, a high-boiling polar aprotic solvent, allows for flexible temperature control and easy handling, making the process adaptable to various manufacturing scales from kilogram to multi-ton production without significant re-engineering of the facility.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this metal-free protocol offers a clear path to sustainable manufacturing. The absence of heavy metals simplifies the wastewater treatment process, as there is no need for complex metal precipitation or recovery systems. This aligns with increasingly stringent environmental regulations regarding heavy metal discharge in the chemical industry. The scalability of the reaction is evidenced by its successful demonstration in gram-scale experiments with consistent results, indicating a smooth trajectory for commercial scale-up. The ability to produce high-purity intermediates with a lower environmental footprint positions manufacturers favorably in markets that prioritize green chemistry and sustainability certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-formic acid and five synthetic steps with only 17% total yield, this novel approach utilizes cheap 2-methylquinoline in a single pot, achieving yields up to 97% without harsh anhydrous conditions.
Q: Does this process involve toxic heavy metal catalysts?
A: No, the process is entirely metal-free. It employs an organic catalytic system consisting of tetrabutylammonium iodide and tert-butyl peroxide, which significantly simplifies downstream purification and regulatory compliance for pharmaceutical intermediates.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the reaction uses commercially available solvents like DMSO and does not require strict oxygen-free or anhydrous environments, making it highly robust and easily scalable from gram-level laboratory synthesis to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the development of novel therapeutics and advanced materials. Our team of expert chemists has thoroughly analyzed the technology disclosed in Patent CN113307790B and is fully equipped to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify every batch.
We invite pharmaceutical and chemical companies to collaborate with us to leverage this cutting-edge synthesis method for their specific projects. Whether you require custom synthesis services or bulk supply of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole derivatives, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline while optimizing your manufacturing costs.
