Revolutionizing Triazole Synthesis: Scalable Catalytic Process for High-Purity Pharmaceutical Intermediates
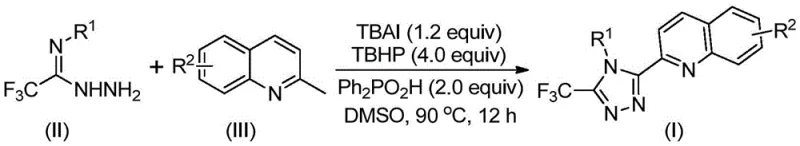
The groundbreaking patent CN113307790B introduces a novel synthetic pathway for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical limitations in traditional heterocyclic chemistry. This method represents a paradigm shift by eliminating transition metal catalysts while achieving high-yield production under ambient conditions, directly responding to the pharmaceutical industry's demand for sustainable and scalable intermediate manufacturing. The process leverages commercially available starting materials including tetrabutylammonium iodide and tert-butyl peroxide aqueous solution, enabling cost-effective production without specialized infrastructure. Crucially, the reaction operates efficiently at 80–100°C for 8–14 hours in standard organic solvents like DMSO, bypassing the stringent anhydrous and oxygen-free requirements that plague conventional triazole syntheses. This innovation significantly enhances the feasibility of large-scale production while maintaining the structural diversity required for next-generation pharmaceutical applications, particularly in bioactive molecular frameworks where triazole moieties serve as essential pharmacophores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of quinolyl-substituted triazoles relies on multi-step sequences starting from quinoline-2-formic acid, typically requiring five reaction stages under severe conditions that yield only 17% of the target compound. These processes demand rigorous anhydrous and oxygen-free environments, specialized equipment for handling sensitive reagents, and extensive purification protocols to remove transition metal residues. The low overall yield stems from multiple intermediate isolations and harsh reaction conditions that promote side reactions and decomposition pathways. Furthermore, the narrow substrate scope restricts structural diversity, limiting the ability to generate tailored analogs for structure-activity relationship studies in drug discovery. The necessity for heavy metal catalysts introduces significant contamination risks that complicate quality control for pharmaceutical applications and necessitate expensive metal removal processes, ultimately increasing production costs and extending lead times for critical intermediates.
The Novel Approach
The patented methodology overcomes these constraints through an iodide-promoted oxidative cyclization that operates under ambient atmospheric conditions without transition metals. By utilizing tetrabutylammonium iodide as a catalytic promoter with tert-butyl peroxide as the oxidant, the process converts readily available 2-methylquinoline into the reactive quinoline aldehyde intermediate in situ. This eliminates the need for pre-synthesized aldehydes and enables direct condensation with trifluoroethylimine hydrazide to form the triazole core through a dehydrated hydrazone intermediate. The reaction achieves high conversion rates within a single vessel at moderate temperatures (80–100°C), with yields ranging from 51% to 97% across diverse substrate combinations as demonstrated in the patent examples. Critically, the absence of heavy metal catalysts removes contamination concerns while simplifying purification protocols, making this approach inherently more suitable for commercial-scale pharmaceutical intermediate production.
Mechanistic Insights into Iodide-Catalyzed Oxidative Cyclization
The reaction proceeds through a well-defined radical-mediated pathway where tetrabutylammonium iodide initiates oxidation of 2-methylquinoline to form the key quinoline aldehyde intermediate. This occurs via iodide oxidation by tert-butyl peroxide to generate iodine radicals that abstract hydrogen from the methyl group, followed by oxygen insertion to yield the aldehyde functionality. The aldehyde then undergoes condensation with trifluoroethylimine hydrazide to form a hydrazone intermediate, which subsequently undergoes oxidative iodination and intramolecular electrophilic substitution to construct the triazole ring system. The final aromatization step completes the formation of the 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole core structure through elimination of water and hydrogen iodide. This mechanism avoids unstable intermediates and side reactions common in traditional approaches, contributing to the high yields observed across various substrate combinations.
Impurity control is achieved through precise stoichiometric management of diphenyl phosphoric acid (2.0 equiv), which serves as both a proton source and stabilizing agent during the cyclization phase. The additive minimizes undesired oxidation pathways by modulating the reaction's acidity profile while facilitating the aromatization step through proton transfer mechanisms. The broad functional group tolerance—accommodating substituents like methyl, methoxy, halogen, and nitro groups at multiple positions—prevents competitive side reactions that could generate impurities. Post-reaction purification via standard column chromatography effectively removes minor byproducts such as unreacted starting materials or trace iodine species, ensuring stringent purity specifications required for pharmaceutical intermediates without requiring specialized equipment or additional processing steps.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl Triazole Efficiently
This innovative synthesis route represents a significant advancement in heterocyclic chemistry manufacturing by replacing multi-step conventional methods with a streamlined single-vessel process that maintains exceptional product quality while reducing operational complexity. The patent demonstrates robust performance across diverse substrate combinations through systematic optimization of reagent ratios and reaction parameters, providing a reliable foundation for commercial implementation. Detailed standardized synthesis procedures have been developed based on the patent's experimental framework to ensure consistent production of high-purity triazole intermediates meeting pharmaceutical industry standards. The following section provides step-by-step guidance for implementing this methodology in manufacturing environments.
- Combine tetrabutylammonium iodide (1.2 equiv), tert-butyl peroxide aqueous solution (4.0 equiv), diphenyl phosphoric acid (2.0 equiv), trifluoroethylimine hydrazide, and 2-methylquinoline in DMSO solvent under ambient conditions.
- Heat the reaction mixture to 80–100°C with continuous stirring for 8–14 hours to achieve complete oxidative cyclization and aromatization.
- Perform post-reaction processing through filtration, silica gel mixing, and column chromatography purification to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
This catalytic process delivers substantial value across procurement and supply chain operations by addressing fundamental pain points in intermediate manufacturing through inherent process improvements rather than incremental optimizations. The elimination of transition metal catalysts removes both capital expenditure requirements for specialized containment systems and recurring costs associated with metal removal protocols, creating immediate cost advantages without requiring new infrastructure investments. Furthermore, the use of commercially available reagents with extended shelf lives enhances supply chain resilience by reducing dependency on specialized chemical suppliers while enabling just-in-time inventory management strategies that minimize working capital requirements.
- Cost Reduction in Manufacturing: The avoidance of expensive transition metal catalysts eliminates multiple cost drivers including catalyst procurement, specialized handling equipment, and rigorous metal residue testing protocols required for pharmaceutical intermediates. The simplified purification process reduces solvent consumption and chromatography media requirements while enabling higher throughput due to shorter cycle times per batch. These combined factors create significant cost savings through reduced raw material expenses and lower operational complexity without compromising product quality or purity specifications.
- Enhanced Supply Chain Reliability: Operating under ambient atmospheric conditions removes critical dependencies on specialized gas handling systems and moisture-sensitive reagents that frequently cause production delays in traditional syntheses. The use of stable, commercially available starting materials with established global supply networks ensures consistent availability regardless of regional disruptions. This robustness translates to predictable lead times and reduced risk of batch failures due to environmental factors, providing procurement teams with greater confidence in delivery schedules while minimizing safety stock requirements.
- Scalability and Environmental Compliance: The reaction's compatibility with standard glass-lined reactors enables seamless scale-up from laboratory to commercial production without requiring specialized equipment modifications. The absence of heavy metals simplifies waste stream management by eliminating hazardous metal-containing byproducts that require special disposal protocols. This environmentally favorable profile supports regulatory compliance while reducing waste treatment costs, making it particularly suitable for manufacturers seeking sustainable production methods that align with green chemistry principles without sacrificing economic viability.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113307790B's experimental data and implementation framework. These insights reflect practical experience from scaling similar catalytic processes to commercial production volumes while maintaining stringent quality standards required for pharmaceutical applications.
Q: How does this method eliminate heavy metal catalysts while maintaining high yield?
A: The iodide-catalyzed oxidative cyclization mechanism converts 2-methylquinoline to quinoline aldehyde in situ, enabling condensation with trifluoroethylimine hydrazide without transition metals. This avoids costly metal removal steps and eliminates contamination risks in pharmaceutical intermediates.
Q: What substrate flexibility advantages does this process offer for API manufacturing?
A: The broad functional group tolerance allows diverse R¹ and R² substitutions on aryl groups, enabling rapid synthesis of customized triazole derivatives. This design flexibility supports accelerated development of novel drug candidates while maintaining consistent purity profiles.
Q: How does the ambient reaction condition improve supply chain reliability?
A: Operating without anhydrous or oxygen-free requirements simplifies equipment needs and reduces process failure points. This robustness ensures consistent batch-to-batch quality and shorter lead times for high-purity pharmaceutical intermediates in global supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl Triazole Supplier
Our patented synthesis methodology represents a significant advancement in triazole intermediate manufacturing that directly addresses the critical challenges faced by modern pharmaceutical developers seeking reliable sources of high-purity building blocks. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities. Our dedicated technical teams specialize in adapting patented processes like this iodide-catalyzed cyclization to meet specific client requirements while ensuring seamless integration into existing manufacturing workflows through comprehensive route feasibility assessments.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative process can optimize your supply chain economics while meeting your exact quality specifications. Please contact us to obtain specific COA data and detailed route feasibility assessments tailored to your production requirements.
