Scalable Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles via Metal-Free Oxidative Cyclization
Scalable Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles via Metal-Free Oxidative Cyclization
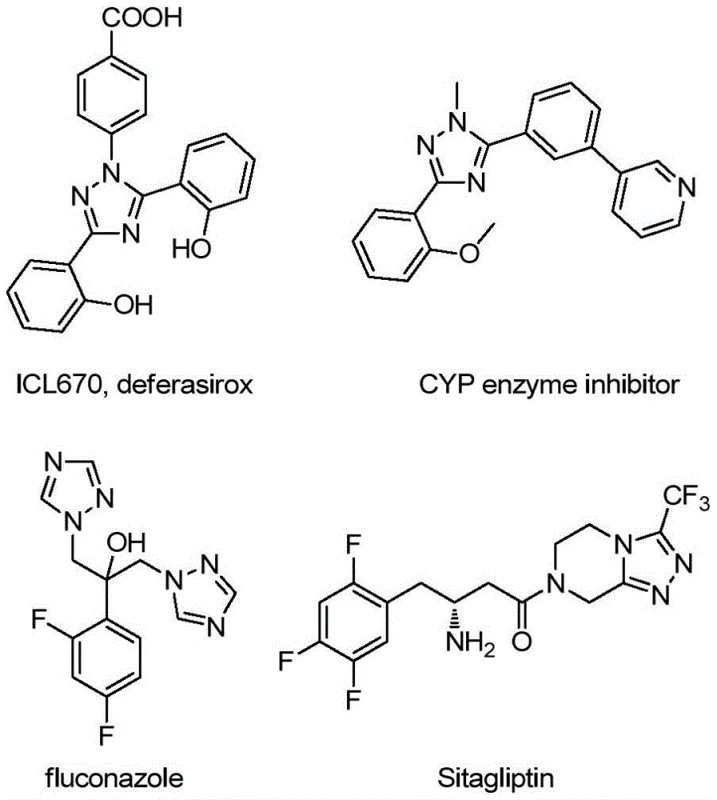
The pharmaceutical and agrochemical industries continuously demand efficient routes to complex heterocyclic scaffolds, particularly those containing nitrogen-rich rings like 1,2,4-triazoles. Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in existing synthetic methodologies. This technology leverages a metal-free oxidative cyclization strategy utilizing tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP) as the primary promoting system. By avoiding toxic heavy metal catalysts and严苛 anhydrous conditions, this invention offers a robust platform for generating diverse bioactive molecules. The core innovation lies in the direct coupling of readily available 2-methylquinoline derivatives with trifluoroacetimidoyl hydrazides, streamlining what was previously a cumbersome multi-step process into a single, operationally simple reaction vessel procedure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional literature methods typically rely on quinoline-2-carboxylic acid as the starting material, necessitating a tedious five-step reaction sequence to construct the target heterocycle. This legacy approach suffers from a dismal total yield of approximately 17%, rendering it economically unviable for large-scale manufacturing. Furthermore, these conventional routes often require severe reaction conditions, including strict moisture control and the use of expensive or hazardous reagents. The reliance on carboxylic acid precursors also limits the structural diversity accessible to medicinal chemists, as functionalizing the quinoline ring prior to cyclization can be synthetically challenging. Consequently, the industry has lacked a reliable, high-yielding method to access this privileged scaffold efficiently.
The Novel Approach
The methodology disclosed in CN113307790B represents a paradigm shift by utilizing 2-methylquinoline and trifluoroacetimidoyl hydrazide as direct building blocks. This novel route bypasses the need for pre-functionalized carboxylic acids, instead employing an oxidative activation of the methyl group on the quinoline ring. The reaction proceeds through a cascade involving oxidation, condensation, and cyclization, all facilitated by the TBAI/TBHP system in the presence of diphenylphosphoric acid. This approach not only drastically reduces the step count but also significantly improves atom economy. The tolerance for various substituents on both the quinoline and the hydrazide components allows for the rapid generation of diverse libraries, making it an ideal tool for lead optimization in drug discovery programs.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
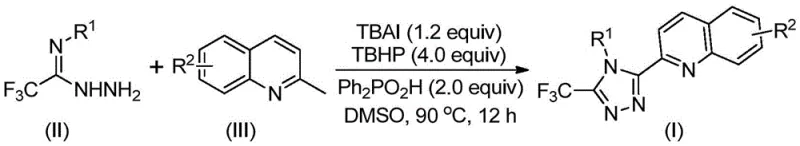
The mechanistic pathway of this transformation is a sophisticated interplay of radical and ionic processes driven by the iodine/peroxide system. Initially, the TBAI and TBHP combination facilitates the oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it in situ into a reactive 2-quinolinecarbaldehyde intermediate. This aldehyde then undergoes a condensation reaction with the trifluoroacetimidoyl hydrazide to form a dehydrated hydrazone species. Subsequently, the system promotes an oxidative iodination followed by an intramolecular electrophilic substitution. This critical cyclization step closes the triazole ring, followed by aromatization to yield the final stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product. The presence of diphenylphosphoric acid is believed to stabilize key intermediates and accelerate the dehydration and cyclization steps, ensuring high conversion rates.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed couplings. Since no palladium, copper, or other heavy metals are employed, the risk of metal contamination in the final Active Pharmaceutical Ingredient (API) is eliminated. This simplifies the downstream purification process, as there is no need for expensive scavenging resins or complex extraction protocols to remove trace metals to ppm levels. The reaction profile suggests that side reactions are minimized due to the specificity of the radical oxidation towards the benzylic methyl position. Furthermore, the use of aqueous TBHP and common organic solvents like DMSO ensures that the reaction medium remains homogeneous, preventing localized hot spots that could lead to decomposition or polymerization byproducts.
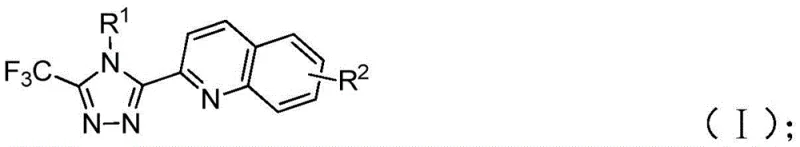
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and temperature control to maximize yield. The standard protocol involves mixing the hydrazide, 2-methylquinoline, TBAI, TBHP, and diphenylphosphoric acid in a polar aprotic solvent. The reaction is typically heated to between 80°C and 100°C for a duration of 8 to 14 hours. Post-reaction workup is straightforward, involving filtration and silica gel treatment followed by standard column chromatography. This simplicity makes the process highly attractive for both laboratory scale-up and pilot plant operations.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphoric acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits regarding cost stability and sourcing reliability. The elimination of precious metal catalysts removes a significant variable cost associated with fluctuating market prices for metals like palladium or rhodium. Additionally, the starting materials, specifically 2-methylquinoline and various aromatic amines used to prepare the hydrazide, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The operational simplicity, which does not require inert atmosphere gloveboxes or specialized dry solvents, further lowers the barrier to entry for contract manufacturing organizations (CMOs).
- Cost Reduction in Manufacturing: The primary driver for cost reduction in pharmaceutical intermediate manufacturing with this technology is the removal of expensive catalytic systems and the reduction of synthetic steps. By consolidating a five-step sequence into a single pot, labor costs, solvent consumption, and energy usage are drastically reduced. The avoidance of heavy metals also eliminates the capital expenditure and operational costs associated with metal removal and validation testing, leading to substantial overall process economics improvements.
- Enhanced Supply Chain Reliability: The robustness of this chemistry against moisture and oxygen means that production schedules are less likely to be disrupted by environmental control failures. The use of widely available reagents like TBAI and TBHP ensures that raw material lead times remain short and predictable. This reliability is crucial for maintaining continuous supply chains for critical API intermediates, preventing stockouts that could delay downstream drug formulation and market launch.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior as it avoids toxic heavy metal waste streams. The reaction can be easily expanded from gram-scale to multi-kilogram production without significant re-optimization, facilitating a smooth technology transfer to commercial manufacturing sites. The use of DMSO, while requiring proper recovery systems, is a well-understood solvent in the industry, simplifying waste management compliance and supporting green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for R&D teams evaluating this route for their specific projects.
Q: What are the advantages of this metal-free synthesis over traditional methods?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid and harsh multi-step conditions yielding only 17%, this novel approach uses cheap 2-methylquinoline and achieves high yields under mild, metal-free conditions without needing anhydrous or oxygen-free environments.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available reagents like TBAI and TBHP, operates in common solvents like DMSO, and has been demonstrated effectively on gram scales, indicating strong potential for commercial scale-up.
Q: What is the role of diphenylphosphoric acid in this reaction?
A: Diphenylphosphoric acid acts as a crucial additive that promotes the oxidative cyclization efficiency, helping to convert the intermediate hydrazone into the final aromatized 1,2,4-triazole ring system with high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative metal-free technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain strategy and accelerate your time to market.
