Revolutionizing Aziridine Production: A Safe and Scalable Base-Mediated Cyclization Strategy
Revolutionizing Aziridine Production: A Safe and Scalable Base-Mediated Cyclization Strategy
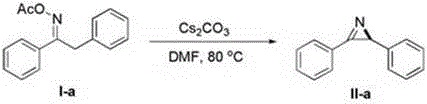
The synthesis of nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, with aziridine compounds serving as pivotal building blocks for complex drug molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN108689901B, which discloses a novel and efficient method for preparing aziridine derivatives represented by formula (II). This technology leverages an intramolecular nucleophilic cyclization of oxime ester derivatives (formula I) in the presence of a base, offering a robust alternative to legacy synthetic routes. For R&D directors and procurement specialists seeking reliable aziridine supplier partnerships, this patent outlines a pathway that combines high atom economy with exceptional operational safety. The process eliminates the need for hazardous azide precursors, utilizing instead stable oxime esters that undergo smooth cyclization under mild thermal conditions, typically between 60°C and 80°C.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the strained three-membered aziridine ring has been fraught with significant safety and efficiency challenges that hinder large-scale adoption. Traditional synthetic strategies predominantly rely on two problematic approaches: the thermal or photocatalytic rearrangement of alkenyl azides and the intramolecular cyclization of sulfonyl oximes. The former method is particularly dangerous due to the inherent instability and explosive nature of azide compounds, posing severe risks during handling and storage in a manufacturing environment. Furthermore, the preparation of these azide precursors often involves multi-step sequences with poor overall yields. The latter approach, involving sulfonyl oximes, frequently necessitates the use of highly toxic reagents and generates substantial amounts of sulfurous waste, creating heavy burdens on environmental compliance and waste treatment infrastructure. These conventional pathways are often incompatible with green chemistry principles, suffering from harsh reaction conditions, strong corrosivity to reactor equipment, and limited substrate tolerance, making them unsuitable for the cost-sensitive production of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these hazardous legacy methods, the technology described in patent CN108689901B introduces a streamlined, base-mediated cyclization strategy that fundamentally reshapes the production landscape for aziridine compounds. By employing readily accessible oxime ester derivatives as the starting materials, this novel approach bypasses the need for explosive azides entirely. The reaction proceeds efficiently in polar aprotic solvents such as DMF, DMA, or DMSO, with DMF being the preferred medium for optimal results. The use of inorganic bases, specifically cesium carbonate, acts as a mild yet effective promoter for the intramolecular nucleophilic attack, driving the formation of the aziridine ring with high selectivity. This method not only simplifies the operational procedure by reducing the number of synthetic steps but also drastically improves the safety profile of the manufacturing process. The mild reaction temperatures, ranging from 60°C to 80°C, further contribute to energy efficiency and reduce the thermal stress on processing equipment, facilitating a smoother transition from laboratory benchtop to commercial scale-up.

The versatility of this synthetic route is exemplified by its broad substrate scope, accommodating a wide array of electronic and steric environments. As illustrated in the specific transformation of 1,2-diphenylethanone oxime ester to 2,3-diphenyl-2H-aziridine, the system tolerates various substituents on the aromatic rings, including halogens, alkyl groups, and alkoxy moieties. This flexibility is crucial for medicinal chemists who require diverse libraries of aziridine intermediates for structure-activity relationship (SAR) studies. The ability to synthesize these complex heterocycles in high yields, often exceeding 80% and reaching up to 93% in optimized cases, underscores the efficiency of this catalytic system. Moreover, the reaction demonstrates excellent functional group tolerance, allowing for the preservation of sensitive moieties that might otherwise be degraded under the harsh acidic or oxidative conditions typical of older methodologies.
Mechanistic Insights into Base-Mediated Intramolecular Cyclization
The core of this technological advancement lies in the elegant mechanism of base-promoted intramolecular nucleophilic cyclization. Upon the addition of cesium carbonate to the reaction mixture containing the oxime ester derivative, the base likely facilitates the generation of a nucleophilic species, potentially through the deprotonation of an alpha-carbon or activation of the oxime nitrogen, although the exact mechanistic pathway may involve a concerted displacement of the acetate leaving group. The proximity of the nucleophilic center to the electrophilic carbon within the oxime ester framework drives the formation of the strained three-membered ring. The choice of cesium carbonate is critical; its large cation radius and moderate basicity provide the ideal balance to promote cyclization without inducing unwanted side reactions such as hydrolysis or elimination. This precise control over reactivity ensures that the desired aziridine product is formed with high regioselectivity and stereochemical integrity, minimizing the formation of isomeric impurities that are difficult to separate downstream.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of heavy metal catalysts eliminates the risk of metal contamination in the final product, a critical quality attribute for pharmaceutical intermediates intended for human consumption. Traditional metal-catalyzed routes often require rigorous purification steps, such as scavenging or recrystallization, to meet strict residual metal limits imposed by regulatory bodies like the FDA or EMA. In this base-mediated process, the primary byproducts are inorganic salts and acetate species, which are easily removed during the aqueous workup phase. The simplicity of the workup procedure—typically involving extraction with ethyl acetate and water followed by drying and concentration—further enhances the purity profile of the crude product. This streamlined purification workflow not only reduces processing time but also minimizes product loss, contributing to the overall high isolated yields reported in the patent examples.
How to Synthesize 2,3-Diphenyl-2H-Aziridine Efficiently
To implement this synthesis effectively, operators should adhere to the standardized protocol outlined in the patent examples, which emphasizes the importance of an inert atmosphere and precise temperature control. The process begins with the charging of the oxime ester substrate and cesium carbonate into a dry reaction vessel under argon protection to prevent moisture interference. Following the addition of anhydrous DMF, the mixture is heated to the optimal range of 60°C to 80°C, where it is maintained until thin-layer chromatography indicates complete consumption of the starting material. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Charge a reaction vessel with the oxime ester derivative substrate and cesium carbonate base (20-120 mol%) in an inert atmosphere.
- Add anhydrous DMF solvent and heat the mixture to 60-80°C while monitoring reaction progress via TLC.
- Upon completion, cool the mixture, extract with ethyl acetate and water, dry the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxime ester cyclization technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift away from hazardous azide chemistry significantly lowers the barrier to entry for manufacturing these valuable intermediates, as it removes the need for specialized explosive-handling facilities and extensive safety protocols associated with high-energy compounds. This reduction in operational complexity directly correlates to lower capital expenditure and reduced insurance costs, making the production of aziridine derivatives more economically viable. Furthermore, the reliance on commodity chemicals like cesium carbonate and DMF ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or single-source catalysts that are prone to market volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive transition metal catalysts and the simplification of the purification train. By avoiding the use of precious metals such as palladium or rhodium, which are often required in alternative cyclization methods, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure processes, further driving down utility costs. The high selectivity of the reaction minimizes the formation of byproducts, thereby reducing the volume of solvents and adsorbents required for chromatographic purification, leading to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent supply continuity, a critical factor for pharmaceutical companies managing tight production schedules. The starting materials, oxime ester derivatives, are generally stable and can be synthesized from readily available ketones and hydroxylamine derivatives, creating a secure upstream supply base. Unlike processes dependent on unstable intermediates that require immediate use or cold chain logistics, the reagents in this method can be stored and handled under standard conditions. This stability allows for better inventory management and reduces the risk of production delays caused by reagent degradation or supply shortages, ensuring that downstream API synthesis timelines are met reliably.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is straightforward due to the use of standard industrial solvents and equipment. The reaction does not generate noxious gases or highly toxic waste streams, aligning well with increasingly stringent environmental regulations. The aqueous workup generates waste streams that are easier to treat compared to those containing heavy metals or sulfur byproducts. This environmental compatibility not only simplifies permitting for new manufacturing lines but also enhances the corporate sustainability profile of the producer, a growing priority for global pharmaceutical buyers who are scrutinizing the green credentials of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aziridine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this oxime ester method improve safety compared to traditional aziridine synthesis?
A: Traditional methods often rely on explosive alkenyl azides or toxic sulfonyl reagents. This patented process utilizes stable oxime ester derivatives and mild inorganic bases like cesium carbonate, significantly reducing explosion risks and toxicity hazards.
Q: What is the substrate scope for R1 and R3 groups in this cyclization reaction?
A: The method demonstrates broad compatibility. R1 can be alkyl, phenyl, substituted phenyl, or naphthyl groups. R3 substituents on the aromatic ring can include halogens, alkyls, and alkoxy groups, allowing for diverse functionalization of the final aziridine product.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction operates under mild temperatures (60-80°C) using common industrial solvents like DMF. The absence of expensive transition metal catalysts and the use of simple workup procedures make it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aziridine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality aziridine intermediates play in the development of next-generation therapeutics. Our technical team has extensively analyzed the potential of the base-mediated cyclization route described in CN108689901B and is fully equipped to leverage this chemistry for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our facility is outfitted with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for aziridine-based projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your project goals,索取 specific COA data for our catalog products, or request route feasibility assessments for custom synthesis campaigns. Let us help you accelerate your drug development timeline with safe, scalable, and cost-effective chemical solutions.
