Scalable Metal-Free Synthesis of Diaryl Sulfides for Advanced Pharmaceutical Applications
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for greener, more sustainable, and cost-effective manufacturing processes. A pivotal advancement in this domain is detailed in Chinese Patent CN109705006B, which discloses a novel preparation method for diaryl sulfide compounds. This intellectual property introduces a groundbreaking metal-free visible-light photocatalytic system that utilizes organic dyes as photosensitizers and hydrogen peroxide as an oxidant. Unlike traditional methods that rely heavily on precious transition metals, this innovation operates under remarkably mild conditions, typically between 25-45°C, and utilizes air as the reaction atmosphere. The strategic shift away from toxic heavy metals addresses one of the most persistent challenges in pharmaceutical intermediate manufacturing: the rigorous and costly removal of metal residues to meet stringent regulatory purity standards. By leveraging cheap and easily available starting materials such as aryl hydrazines and diaryl disulfides, this technology offers a robust pathway for producing high-purity sulfur-containing scaffolds essential for drug discovery.
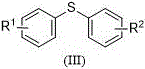
Diaryl sulfides represent a critical structural motif found extensively in bioactive molecules, natural products, and advanced functional materials. Their presence is often correlated with significant biological activities, including potential applications in treating HIV, cancer, and neurodegenerative disorders like Alzheimer's and Parkinson's disease. Consequently, the ability to synthesize these compounds efficiently and cleanly is of paramount importance to R&D directors and procurement strategists alike. The patent highlights a versatile substrate scope where R1 and R2 groups can vary widely, including methoxy, chloro, cyano, nitro, and amino functionalities. This structural diversity allows for the rapid generation of compound libraries for medicinal chemistry campaigns. Furthermore, the method's compatibility with asymmetric synthesis opens new doors for creating chiral sulfur-containing intermediates, which are increasingly demanded in the development of next-generation active pharmaceutical ingredients (APIs). The adoption of such a flexible and environmentally benign protocol positions manufacturers to respond more agilely to market demands for complex fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds to form diaryl sulfides has relied predominantly on transition metal-catalyzed cross-coupling reactions. These conventional pathways typically involve the coupling of halogenated compounds or diazonium salts with thiophenols or disulfides using catalysts based on palladium, copper, or nickel. While effective in many contexts, these methods suffer from inherent drawbacks that severely impact commercial viability and environmental compliance. Firstly, the requirement for high temperatures and harsh reaction conditions often leads to energy inefficiencies and safety hazards in large-scale reactors. Secondly, and perhaps most critically for the pharmaceutical industry, is the issue of metal contamination. Residual transition metals in the final product are strictly regulated due to their toxicity, necessitating additional downstream purification steps such as scavenging or recrystallization. These extra processing stages not only extend the production lead time but also drastically increase the overall cost of goods sold (COGS). Additionally, some alternative methods involving microwave irradiation of aryldiazo compounds pose significant safety risks due to the instability of the diazo precursors, limiting their practical application in industrial settings.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN109705006B represents a paradigm shift towards sustainable organic synthesis. By employing an organic dye, specifically Eosin Y disodium salt, as a photocatalyst, the reaction harnesses visible light energy to drive the transformation at near-ambient temperatures. This approach completely eliminates the need for expensive and toxic transition metals, thereby simplifying the workup procedure and ensuring a cleaner impurity profile from the outset. The use of hydrogen peroxide as a terminal oxidant is another key feature, as it generates water as the only byproduct, aligning perfectly with green chemistry principles. The operational simplicity is further enhanced by the fact that the reaction can be conducted directly in air, removing the need for inert gas protection systems which are costly to maintain. This novel strategy not only reduces the environmental footprint of the manufacturing process but also significantly lowers the barrier to entry for producing high-value sulfur-containing intermediates. The ability to conveniently synthesize asymmetric diaryl sulfides adds another layer of value, enabling the production of more complex molecular architectures without compromising on efficiency or safety.
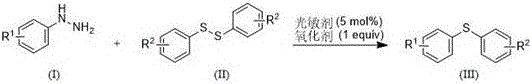
Mechanistic Insights into Visible-Light Photocatalytic Coupling
The core of this innovative synthesis lies in the intricate interplay between the organic photosensitizer and the oxidant under visible light irradiation. When Eosin Y absorbs photons from the visible light source, it transitions to an excited state capable of engaging in single-electron transfer (SET) processes. In the presence of hydrogen peroxide and the aryl hydrazine substrate, a radical cascade is initiated. The excited photocatalist likely facilitates the oxidation of the aryl hydrazine to generate an aryl radical species, while simultaneously activating the disulfide bond or interacting with the oxidant to propagate the radical chain. This radical-mediated mechanism bypasses the traditional oxidative addition and reductive elimination cycles associated with transition metal catalysis. Instead, it relies on the high reactivity of transient radical intermediates to forge the C-S bond efficiently. The mild thermal conditions (25-45°C) are sufficient to sustain this radical flux without causing thermal degradation of sensitive functional groups, which is a common issue in high-temperature metal-catalyzed reactions. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters such as light intensity, wavelength, and reagent stoichiometry to maximize throughput and yield.
From an impurity control perspective, the metal-free nature of this catalytic cycle offers distinct advantages. In traditional metal-catalyzed processes, side reactions often involve the formation of homocoupled byproducts or metal-complexed intermediates that are difficult to separate. In this photocatalytic system, the primary byproducts are derived from the oxidation of the hydrazine moiety and the reduction of hydrogen peroxide to water, both of which are relatively benign and easier to remove during standard aqueous workups or silica gel chromatography. The absence of metal salts means there is no risk of metal-induced decomposition of the product during storage or subsequent formulation steps. Furthermore, the selectivity of the radical attack can be tuned by adjusting the electronic properties of the substituents on the aryl rings, allowing for precise control over the regioselectivity of the sulfide formation. This level of control is essential for maintaining a narrow impurity profile, which is a key requirement for regulatory approval of pharmaceutical intermediates. The robustness of the radical mechanism against moisture and oxygen (since it runs in air) further enhances its reliability for consistent batch-to-batch quality.
How to Synthesize Diaryl Sulfides Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to the specific ratios of reagents and the configuration of the light source. The patent outlines a straightforward procedure where aryl hydrazine and diaryl disulfide are dissolved in dimethyl sulfoxide (DMSO), followed by the addition of the Eosin Y catalyst and hydrogen peroxide. The reaction mixture is then stirred under visible light, such as blue LEDs or even white light, for a duration of 5 to 12 hours. The simplicity of the setup allows for easy adaptation to flow chemistry systems, which could further enhance photon efficiency and scalability. For R&D teams looking to replicate or adapt this chemistry, it is important to note that the molar ratio of aryl hydrazine to diaryl disulfide is typically maintained around 1:4 to ensure complete conversion of the hydrazine, which acts as the limiting reagent. The detailed standardized synthesis steps, including specific workup procedures and purification techniques to achieve the reported yields (e.g., 68% for diphenyl sulfide), are outlined in the structured guide below.
- Combine aryl hydrazine, diaryl disulfide, and DMSO solvent in a reaction vessel.
- Add Eosin Y disodium salt (5 mol%) as photosensitizer and hydrogen peroxide (1 equiv) as oxidant.
- Stir the mixture under visible light irradiation at 25-45°C for 5-12 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free photocatalytic technology translates into tangible strategic benefits that go beyond mere technical novelty. The most immediate impact is seen in the reduction of raw material costs and processing complexity. By eliminating the need for precious metal catalysts like palladium or specialized ligands, the direct material cost of the reaction is significantly lowered. Moreover, the removal of the heavy metal scavenging step simplifies the manufacturing workflow, reducing the consumption of auxiliary chemicals and solvents associated with purification. This streamlining of the process leads to a substantial reduction in the overall production cycle time, allowing for faster turnaround on customer orders. The use of commodity chemicals such as hydrogen peroxide and DMSO, which are readily available in bulk quantities globally, ensures a stable and resilient supply chain that is less susceptible to the geopolitical fluctuations often affecting the supply of rare earth metals or specialized organometallic reagents.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzed through the lens of total cost of ownership. Traditional methods incur hidden costs related to the disposal of metal-contaminated waste streams and the energy-intensive processes required to strip trace metals from the final product. By adopting a metal-free system, manufacturers can bypass these expensive downstream operations entirely. The catalyst, Eosin Y, is an inexpensive organic dye compared to transition metal complexes, contributing to a lower cost per kilogram of the final API intermediate. Additionally, the mild reaction temperatures reduce the energy load on heating and cooling systems, further driving down utility costs. These cumulative savings can be passed on to customers or reinvested into R&D, providing a competitive edge in pricing strategies for high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is a critical metric for any global chemical supplier. This synthesis route relies on feedstocks that are produced on a massive industrial scale, minimizing the risk of shortages. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this photocatalytic method are commoditized. The robustness of the reaction conditions, which tolerate air and moisture, also reduces the risk of batch failures due to minor deviations in operational protocols. This reliability ensures consistent delivery schedules, which is vital for maintaining just-in-time inventory models for downstream pharmaceutical clients. The ability to scale this reaction from gram to multi-ton quantities without changing the fundamental chemistry provides a seamless path from clinical trial material to commercial launch supply.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to demonstrate a green manufacturing process is a significant commercial asset. This method generates minimal hazardous waste, primarily consisting of aqueous streams that are easier to treat than heavy metal sludge. The absence of toxic metals simplifies the regulatory filing process for new drug applications, as the burden of proof for metal clearance is removed. From a scalability perspective, the reaction can be adapted to continuous flow photoreactors, which offer superior light penetration and heat transfer compared to batch vessels. This technological adaptability ensures that production capacity can be ramped up quickly to meet surges in demand without compromising on safety or environmental standards, making it an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light mediated synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of transition metal catalysts, which removes the need for expensive and complex heavy metal removal steps, ensuring higher purity suitable for pharmaceutical applications.
Q: What are the typical reaction conditions required for this synthesis?
A: The reaction proceeds under mild conditions, specifically at temperatures between 25-45°C, using visible light irradiation in the presence of air, with DMSO as the solvent.
Q: Is this process scalable for industrial production of fine chemicals?
A: Yes, the use of cheap and readily available raw materials like aryl hydrazines and diaryl disulfides, combined with simple operational procedures, makes this method highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfide Supplier
The technological potential of metal-free photocatalysis for synthesizing diaryl sulfides is immense, offering a cleaner and more efficient route to valuable chemical building blocks. At NINGBO INNO PHARMCHEM, we recognize the strategic importance of such innovations in driving the next generation of pharmaceutical development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photoreactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of diaryl sulfide intermediate meets the highest global standards for quality and consistency.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to reach out to request specific COA data for our diaryl sulfide portfolio and discuss route feasibility assessments for your target molecules. Together, we can accelerate the development of life-saving therapies while adhering to the highest principles of sustainability and economic efficiency.
