Scalable Metal-Free Synthesis of Diaryl Sulfides for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Diaryl Sulfides for Advanced Pharmaceutical Intermediates
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the realm of pharmaceutical intermediate manufacturing. Patent CN109705006B represents a significant technological breakthrough in this domain by disclosing a novel preparation method for diaryl sulfide compounds. This innovation leverages visible light photocatalysis to couple aryl hydrazines with diaryl disulfides, effectively bypassing the need for traditional transition metal catalysts. For R&D directors and process chemists, this patent offers a compelling alternative to conventional cross-coupling reactions, promising enhanced purity profiles and simplified downstream processing. The method operates under remarkably mild conditions, utilizing an organic dye as a photosensitizer and hydrogen peroxide as a terminal oxidant, which aligns perfectly with modern green chemistry principles. By adopting this technology, manufacturers can achieve substantial improvements in both environmental compliance and economic efficiency, positioning themselves as a reliable diaryl sulfide supplier in the competitive global market.
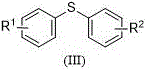
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds to form diaryl sulfides has relied heavily on transition metal-catalyzed coupling reactions, often involving palladium, copper, or nickel complexes. These traditional pathways typically necessitate harsh reaction conditions, including elevated temperatures and the use of strong bases or acids, which can compromise the integrity of sensitive functional groups on the substrate. Furthermore, the reliance on precious metal catalysts introduces a critical bottleneck in pharmaceutical manufacturing: the removal of trace metal residues. Regulatory bodies impose stringent limits on heavy metal content in active pharmaceutical ingredients (APIs), requiring expensive and time-consuming purification steps such as scavenging or recrystallization. Additionally, some conventional methods utilize unstable diazonium salts or require microwave irradiation, which poses safety risks and limits the feasibility of large-scale batch processing. These factors collectively contribute to higher production costs and extended lead times, creating significant pain points for procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN109705006B introduces a metal-free, visible-light-driven protocol that fundamentally reshapes the synthesis landscape. By employing aryl hydrazines and diaryl disulfides as stable, commercially available starting materials, this method eliminates the hazards associated with diazonium salts. The reaction proceeds efficiently at temperatures ranging from 25-45°C, driven by blue or white light irradiation in the presence of air, which drastically reduces energy consumption compared to thermal heating. The use of Eosin Y disodium salt as an inexpensive organic photosensitizer replaces costly noble metals, while hydrogen peroxide serves as a clean oxidant, generating water as the only byproduct. This approach not only simplifies the operational workflow but also ensures that the final product is free from toxic metal contaminants, thereby streamlining the quality control process. The ability to synthesize asymmetric diaryl sulfides with diverse substituents further underscores the versatility of this novel approach for generating complex chemical libraries.
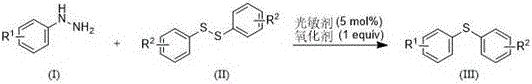
Mechanistic Insights into Visible-Light Organophotocatalysis
The core of this innovative synthesis lies in the intricate interplay between the organic photosensitizer and the reactants under visible light irradiation. Upon absorption of photons, the Eosin Y catalyst enters an excited state, facilitating single-electron transfer (SET) processes that activate the aryl hydrazine substrate. This activation generates reactive aryl radical intermediates, which subsequently attack the disulfide bond of the diaryl disulfide. The presence of hydrogen peroxide as a stoichiometric oxidant is crucial for regenerating the active catalytic species and driving the reaction cycle forward without the accumulation of reduced byproducts. This radical-mediated mechanism operates under ambient atmospheric conditions, leveraging oxygen from the air as a co-oxidant in some pathways, which enhances the atom economy of the process. For process chemists, understanding this mechanism is vital for optimizing reaction parameters such as light intensity and stirring rates to ensure uniform photon flux throughout the reaction mixture, especially when scaling up from milligram to kilogram quantities.
From an impurity control perspective, the metal-free nature of this catalytic system offers distinct advantages for maintaining high-purity diaryl sulfides. Traditional metal-catalyzed routes often suffer from side reactions such as homocoupling of aryl halides or dehalogenation, leading to complex impurity profiles that are difficult to separate. In this photocatalytic system, the selectivity is governed by the redox potentials of the excited catalyst and the specific bond dissociation energies of the disulfide linkage, resulting in a cleaner reaction profile. The absence of metal ions eliminates the formation of metal-organic complexes that can act as nucleation sites for degradation or discoloration of the product. Consequently, the downstream purification burden is significantly reduced, often requiring only standard silica gel column chromatography to achieve analytical purity. This level of control over the impurity spectrum is essential for meeting the rigorous specifications demanded by regulatory agencies for pharmaceutical intermediates.
How to Synthesize Diaryl Sulfides Efficiently
Implementing this synthesis route requires careful attention to the molar ratios and reaction environment to maximize yield and reproducibility. The patent specifies a molar ratio of 1:4:1:0.05 for aryl hydrazine, diaryl disulfide, oxidant, and photosensitizer, respectively, which has been optimized to balance reaction kinetics with material costs. Dimethyl sulfoxide (DMSO) is identified as the preferred solvent due to its ability to dissolve both organic substrates and the ionic photosensitizer while remaining stable under oxidative conditions. The reaction is typically conducted in a transparent vessel to allow maximum penetration of visible light, with temperature control maintained between 25-45°C to prevent thermal degradation of the sensitive hydrazine component. Detailed standardized synthesis steps see the guide below.
- Combine aryl hydrazine, diaryl disulfide, and DMSO solvent in a reaction vessel.
- Add Eosin Y disodium salt (5 mol%) as photosensitizer and hydrogen peroxide (1 equiv) as oxidant.
- Stir under visible light irradiation at 25-45°C for 5-12 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The elimination of precious metal catalysts directly addresses one of the most volatile cost drivers in fine chemical manufacturing, as the prices of palladium and other transition metals are subject to significant market fluctuations. By switching to an organic dye-based system, companies can stabilize their raw material costs and reduce dependency on scarce geological resources. Furthermore, the mild reaction conditions imply lower energy expenditures for heating and cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. The simplicity of the workup procedure, which avoids complex metal scavenging steps, shortens the overall production cycle time, enabling faster response to market demands and reducing inventory holding costs.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the substitution of expensive transition metal catalysts with inexpensive organic dyes like Eosin Y. This change not only lowers the direct material cost per kilogram of product but also eliminates the capital expenditure associated with specialized equipment required for metal removal, such as filtration units loaded with scavenger resins. Additionally, the use of hydrogen peroxide as a benign oxidant avoids the generation of hazardous waste streams that incur high disposal fees. The cumulative effect of these factors results in a significantly leaner cost structure, allowing suppliers to offer more competitive pricing without compromising margins. This economic efficiency is critical for maintaining competitiveness in the global supply chain for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as aryl hydrazines and diaryl disulfides ensures a robust and resilient supply chain. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, these bulk chemicals are produced by multiple vendors worldwide, mitigating the risk of supply disruptions. The operational safety of the process, which avoids high pressures and extreme temperatures, further enhances reliability by reducing the likelihood of unplanned shutdowns due to equipment failure or safety incidents. This stability allows for more accurate production planning and forecasting, ensuring consistent delivery schedules to downstream API manufacturers. Reducing lead time for high-purity pharmaceutical intermediates becomes achievable when the synthesis route is devoid of bottlenecks associated with catalyst sourcing and complex purification.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the homogeneous nature of the reaction mixture and the use of common solvents like DMSO. The absence of pyrophoric reagents or sensitive metal complexes simplifies the engineering controls required for large-scale reactors, making the commercial scale-up of complex sulfur-containing compounds more straightforward. From an environmental standpoint, the process adheres to green chemistry principles by minimizing waste generation and avoiding toxic heavy metals. This compliance simplifies the permitting process for new manufacturing facilities and reduces the regulatory burden associated with environmental monitoring. The ability to operate under air atmosphere further reduces the need for inert gas blanketing, lowering operational complexity and utility costs while maintaining a safe working environment for personnel.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The responses highlight the balance between scientific innovation and practical industrial application, ensuring that decision-makers have a comprehensive view of the technology's potential.
Q: What are the primary advantages of this metal-free photocatalytic method over traditional transition metal coupling?
A: The primary advantage is the complete elimination of transition metal catalysts such as palladium or copper, which removes the risk of heavy metal residues in the final API. Additionally, the reaction proceeds under mild temperatures (25-45°C) using visible light, significantly reducing energy consumption compared to high-temperature thermal methods.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method utilizes cheap and readily available starting materials like aryl hydrazines and diaryl disulfides. The operational simplicity, absence of expensive metal catalysts, and mild reaction conditions make it highly amenable to commercial scale-up for producing pharmaceutical intermediates.
Q: What specific substrates are compatible with this visible-light protocol?
A: The protocol demonstrates broad substrate scope, successfully accommodating various functional groups including methoxy, chloro, nitrile, nitro, and amino groups on both the aryl hydrazine and diaryl disulfide components, allowing for the synthesis of diverse asymmetric diaryl sulfides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free photocatalysis in the synthesis of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN109705006B can be seamlessly translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest standards for metal content and impurity profiles. Our state-of-the-art facilities are equipped to handle photochemical reactions safely and efficiently, allowing us to offer this advanced synthesis route to our global clientele with confidence and reliability.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact us to request specific COA data for our diaryl sulfide portfolio and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to innovation, quality, and sustainable growth in the fine chemical sector.
