Advanced Rhodium Catalysis for Scalable Moxifloxacin Side Chain Production
Advanced Rhodium Catalysis for Scalable Moxifloxacin Side Chain Production
The pharmaceutical industry constantly seeks more efficient pathways to produce critical antibiotic intermediates, and the synthesis of moxifloxacin side chains represents a significant area of innovation. Patent CN108690017B discloses a groundbreaking preparation method for a rhodium-catalyzed moxifloxacin side chain intermediate, specifically targeting the chiral molecule (1S,6R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4,3,0]nonane. This technology addresses long-standing inefficiencies in the production of fourth-generation quinolone antibiotics by replacing wasteful resolution steps with direct asymmetric synthesis. For R&D directors and procurement managers alike, this shift represents a move towards greener chemistry and substantially reduced manufacturing costs. By leveraging a highly selective rhodium catalyst system, this method achieves superior enantioselectivity and substrate universality, positioning it as a preferred route for reliable pharmaceutical intermediate supplier networks aiming to optimize their API supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of the moxifloxacin side chain has been plagued by inherent inefficiencies associated with racemic synthesis followed by chiral resolution. As illustrated in the traditional pathways, such as the route starting from 2,3-dicarboxylpyridine, the process involves multiple steps including anhydride formation, amidation, and non-asymmetric hydrogenation to produce a racemic mixture. 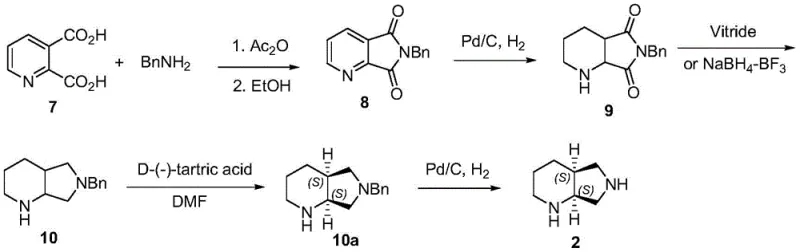 The critical bottleneck in these conventional methods is the necessity to separate the desired (S,S) isomer from its enantiomer, effectively discarding nearly half of the synthesized material as waste. This not only severely caps the theoretical maximum yield at 50% prior to purification losses but also necessitates additional downstream processing steps involving resolving agents and recrystallization. Furthermore, some alternative routes utilize hazardous reagents like sodium hydride in DMF, posing significant safety risks and handling challenges during commercial scale-up of complex pharmaceutical intermediates.
The critical bottleneck in these conventional methods is the necessity to separate the desired (S,S) isomer from its enantiomer, effectively discarding nearly half of the synthesized material as waste. This not only severely caps the theoretical maximum yield at 50% prior to purification losses but also necessitates additional downstream processing steps involving resolving agents and recrystallization. Furthermore, some alternative routes utilize hazardous reagents like sodium hydride in DMF, posing significant safety risks and handling challenges during commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to the multi-step wastefulness of legacy processes, the novel approach described in the patent utilizes a direct asymmetric hydrogenation strategy that fundamentally alters the economic and environmental profile of the synthesis. Instead of generating a racemate and attempting to salvage half of it, this method employs a chiral rhodium catalyst to stereoselectively reduce the double bond in 6-benzyl-1,2,3,4-tetrahydro-pyrrolo[3,4-b]pyridine-5,7-dione. This single catalytic step installs the required chirality with high precision, bypassing the need for any chiral resolution entirely. The result is a streamlined process that maximizes atom economy and minimizes waste generation, aligning perfectly with modern green chemistry principles. By eliminating the resolution step, manufacturers can achieve a drastic simplification of the workflow, leading to faster throughput and reduced consumption of solvents and auxiliary chemicals.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the sophisticated design of the catalytic system, which combines a rhodium precursor with specialized chiral bisphosphine ligands to create a highly active and selective environment for hydrogen transfer. The reaction mechanism involves the in situ formation of a cationic rhodium complex, typically using precursors like bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate or hexafluorophosphate. These metal centers coordinate with chiral ligands from the Duphos or BPE families, such as (S,S)-Me-Duphos or (S,S)-Et-BPE, creating a chiral pocket that dictates the facial selectivity of the hydrogen addition to the substrate. 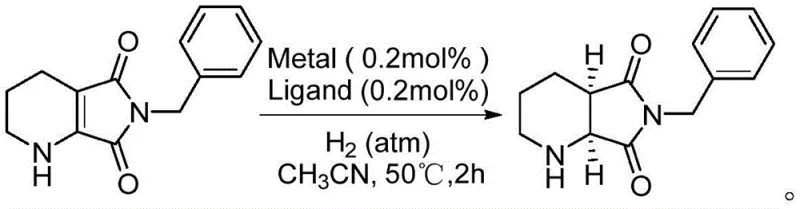 The steric and electronic properties of these ligands are crucial; for instance, ligands with specific alkyl or phenyl substituents on the phospholane rings fine-tune the catalyst's performance, ensuring that the hydrogen atoms are delivered exclusively to one face of the olefinic bond. This precise control allows for the production of the target (1S,6R) configuration with exceptional enantiomeric excess values, often exceeding 90% ee under optimized conditions, thereby ensuring the high optical purity required for subsequent API synthesis.
The steric and electronic properties of these ligands are crucial; for instance, ligands with specific alkyl or phenyl substituents on the phospholane rings fine-tune the catalyst's performance, ensuring that the hydrogen atoms are delivered exclusively to one face of the olefinic bond. This precise control allows for the production of the target (1S,6R) configuration with exceptional enantiomeric excess values, often exceeding 90% ee under optimized conditions, thereby ensuring the high optical purity required for subsequent API synthesis.
From an impurity control perspective, this catalytic mechanism offers distinct advantages over chemical resolution by avoiding the formation of diastereomeric salts which can be difficult to separate completely. The high specificity of the rhodium-ligand complex means that side reactions are minimized, and the formation of the unwanted enantiomer is suppressed at the source rather than removed post-reaction. This intrinsic selectivity reduces the burden on downstream purification processes, such as column chromatography or crystallization, which are often required to meet stringent regulatory specifications for chiral drugs. Furthermore, the robustness of the catalyst system allows for operation under relatively mild conditions, typically between 25°C and 80°C, which helps prevent thermal degradation of the sensitive bicyclic intermediate. This stability ensures a cleaner reaction profile and a more consistent quality of the final product batch after batch.
How to Synthesize (1S,6R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4,3,0]nonane Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst preparation and reaction parameters to fully realize the benefits of high efficiency and enantioselectivity. The process begins with the activation of the rhodium precursor in an anhydrous organic solvent, preferably acetonitrile, under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Once the active catalytic species is generated, the substrate is introduced, and the system is pressurized with hydrogen gas to drive the reduction. Detailed standardized synthesis steps for this procedure are provided in the guide below, outlining the precise molar ratios, pressure settings, and workup procedures necessary for reproducible results.
- Prepare the chiral catalyst in situ by mixing a rhodium salt (e.g., Rh(COD)2BF4) with a chiral bisphosphine ligand (such as (S,S)-Me-Duphos) in an organic solvent like acetonitrile under inert atmosphere.
- Charge the reactor with the substrate 6-benzyl-1,2,3,4-tetrahydro-pyrrolo[3,4-b]pyridine-5,7-dione and the prepared catalyst solution, ensuring a molar ratio between 1: 2000 and 1:100.
- Perform asymmetric hydrogenation under hydrogen pressure (5-50 atm) at temperatures between 25°C and 80°C, followed by solvent removal and purification to isolate the high-purity chiral intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed methodology translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. By fundamentally redesigning the synthesis to eliminate the chiral resolution step, the process removes a major bottleneck that traditionally inflates production timelines and material costs. The ability to convert raw materials directly into the desired chiral intermediate without generating a 50% waste stream of the wrong enantiomer means that the effective capacity of existing manufacturing infrastructure is essentially doubled without requiring new capital investment. This efficiency gain is critical for maintaining supply continuity in the face of fluctuating demand for broad-spectrum antibiotics like moxifloxacin.
- Cost Reduction in Manufacturing: The elimination of chiral resolution agents and the associated separation processes leads to substantial cost savings in raw materials and waste disposal. Since the process avoids the use of hazardous reagents like sodium hydride and reduces the total number of unit operations, the overall operational expenditure is significantly lowered. Furthermore, the high atom economy ensures that expensive starting materials are utilized much more effectively, directly contributing to cost reduction in API manufacturing and improving the margin profile for the final drug product.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route reduces the dependency on multiple specialized reagents and intermediates, thereby mitigating supply chain risks associated with sourcing complex resolving agents. The robustness of the catalytic system allows for more predictable production schedules, as there are fewer variables related to resolution efficiency that can cause batch failures or delays. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on time to meet their own production commitments.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard hydrogenation equipment that is common in fine chemical plants, which facilitates the commercial scale-up of complex pharmaceutical intermediates from pilot to full production volumes. Additionally, by generating significantly less chemical waste and avoiding toxic solvents where possible, the method aligns with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint but also lowers the costs associated with effluent treatment and regulatory compliance, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis route. These answers are derived directly from the patent data and are intended to clarify the operational benefits and technical feasibility for potential partners and licensees. Understanding these details is crucial for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: How does this rhodium-catalyzed method improve upon traditional synthesis routes?
A: Traditional routes rely on non-asymmetric hydrogenation followed by chiral resolution, which inherently wastes 50% of the material as the unwanted enantiomer. This novel method uses asymmetric catalysis to directly produce the desired (S,S) configuration with high enantiomeric excess, eliminating the resolution step and significantly improving overall yield and atom economy.
Q: What are the key reaction conditions for optimal enantioselectivity?
A: Optimal results are achieved using a rhodium precursor like bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate paired with chiral ligands such as (S,S)-Me-Duphos or (S,S)-Et-BPE. The reaction typically proceeds in acetonitrile at moderate temperatures (around 50°C) and hydrogen pressures ranging from 20 to 30 bar.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It avoids hazardous reagents like sodium hydride in DMF found in older routes and utilizes standard hydrogenation equipment. The high conversion rates and elimination of resolution steps make it highly efficient for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moxifloxacin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed technology for the global antibiotic market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of moxifloxacin intermediate meets the highest international standards for chiral purity and chemical quality.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain resilience and competitive advantage in the quinolone antibiotic sector.
