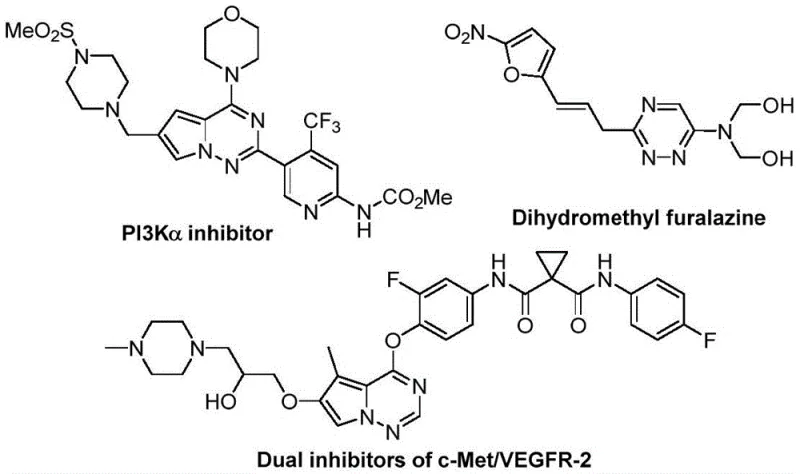
Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating trifluoromethyl groups which enhance metabolic stability and lipophilicity. A significant breakthrough in this domain is documented in patent CN116253692A, which discloses a novel preparation method for trifluoromethyl substituted 1,2,4-triazine compounds. These scaffolds are critical building blocks in modern drug discovery, serving as key motifs in potent bioactive molecules ranging from anticancer agents to antifungal therapies. The strategic incorporation of the trifluoromethyl group into the 1,2,4-triazine core offers substantial improvements in physicochemical properties, making these compounds highly desirable for the development of next-generation active pharmaceutical ingredients (APIs). This report analyzes the technical merits and commercial viability of this metal-free synthetic route.
Historically, the construction of 1,2,4-triazine rings has relied on conventional condensation reactions involving amidrazones and 1,2-diketones or alkynes, often necessitating harsh conditions and complex substrate pre-functionalization. Traditional multicomponent reactions frequently suffer from limited structural diversity and poor atom economy, while cyclization methods using hydrazine with oxazolone or oxadiazole derivatives often require elevated temperatures and extended reaction times. Furthermore, many existing protocols depend on stoichiometric amounts of oxidants or sensitive reagents that complicate the operational workflow and increase safety hazards in a manufacturing environment. These limitations have historically constrained the widespread adoption of 1,2,4-triazine derivatives in cost-sensitive commercial applications, prompting the need for more efficient and sustainable synthetic alternatives.
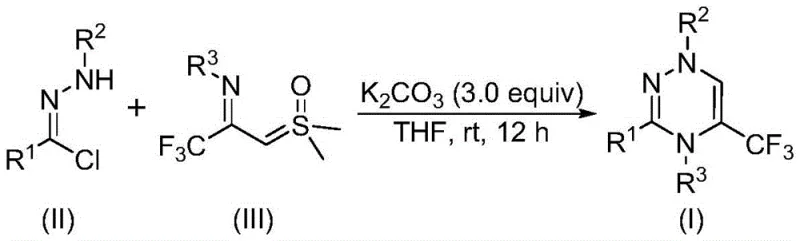
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a synergistic [3+3] cycloaddition strategy that operates under remarkably mild conditions. By employing chlorohydrazone and trifluoroacetyl sulfur ylide as readily available starting materials, the process bypasses the need for pre-synthesized complex substrates. The reaction is promoted by potassium carbonate, an inexpensive and non-toxic inorganic base, and proceeds smoothly in common organic solvents like tetrahydrofuran at temperatures between 20°C and 40°C. Crucially, the transformation occurs in an air atmosphere without the need for inert gas protection, drastically simplifying the engineering requirements for reactor setup. This methodological shift represents a paradigm change in heterocyclic synthesis, prioritizing operational simplicity and safety without compromising on yield or product quality.
Mechanistic Insights into Potassium Carbonate-Promoted Cycloaddition
The mechanistic pathway of this transformation involves the generation of a reactive nitrile imine intermediate, which serves as the 1,3-dipole in the subsequent cycloaddition event. Under the promotion of potassium carbonate, one molecule of hydrogen chloride is eliminated from the chlorohydrazone precursor to generate this transient species in situ. This nitrile imine then undergoes a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, leading to the formation of the six-membered 1,2,4-triazine ring system. The reaction concludes with the elimination of one molecule of dimethyl sulfoxide, driving the equilibrium towards the desired product. Understanding this mechanism is vital for process chemists, as it highlights the importance of base selection and solvent compatibility in maintaining the stability of the reactive intermediates throughout the 10 to 14 hour reaction window.
Impurity control is another critical aspect where this mechanism offers distinct advantages over transition-metal catalyzed routes. Since the reaction does not involve heavy metal catalysts such as palladium, copper, or nickel, there is no risk of metal leaching into the final product, which is a stringent requirement for pharmaceutical intermediates intended for human consumption. The primary byproducts are inorganic salts and dimethyl sulfoxide, both of which are easily removed during standard aqueous workup and purification procedures. The absence of metal-mediated side reactions, such as homocoupling or over-reduction, ensures a cleaner crude reaction profile. This inherent purity simplifies the downstream purification process, often allowing for high-purity isolation via standard column chromatography or crystallization, thereby reducing the overall processing time and solvent consumption associated with extensive scrubbing steps.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
To implement this synthesis effectively, operators must adhere to precise molar ratios and mixing protocols to maximize conversion efficiency. The patent specifies a preferred molar ratio of chlorohydrazone to trifluoroacetyl sulfur ylide to potassium carbonate of 1:2:3, ensuring that the base is present in sufficient excess to drive the dehydrohalogenation step to completion. The reaction mixture should be stirred uniformly in an aprotic solvent like tetrahydrofuran, which effectively dissolves both organic reactants and the inorganic base. Detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined in the technical guide below to ensure reproducibility across different batch sizes.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran under air atmosphere.
- Maintain the reaction temperature between 20°C and 40°C and stir continuously for a duration of 10 to 14 hours to ensure complete conversion.
- Upon completion, perform post-treatment involving filtration and silica gel mixing, followed by column chromatography purification to isolate the final trifluoromethyl substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers compelling economic benefits driven by the simplification of raw material sourcing and process engineering. The elimination of expensive transition metal catalysts removes a significant cost center, as these metals often account for a substantial portion of the bill of materials in fine chemical synthesis. Furthermore, the ability to run the reaction in air at near-room temperature significantly reduces energy consumption compared to processes requiring cryogenic cooling or high-temperature reflux. These factors collectively contribute to a more favorable cost structure, making the commercial production of these high-value intermediates more accessible for large-scale manufacturing projects without sacrificing quality standards.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with inexpensive potassium carbonate results in direct savings on raw material costs, while the simplified workup reduces solvent usage and waste disposal expenses. The avoidance of specialized equipment for handling air-sensitive reagents further lowers capital expenditure requirements for production facilities. Additionally, the high conversion rates observed across various substrate scopes minimize the loss of valuable starting materials, enhancing the overall material efficiency of the process. These cumulative effects lead to substantial cost optimization in the manufacturing of complex heterocyclic intermediates.
- Enhanced Supply Chain Reliability: The starting materials, including chlorohydrazones and trifluoroacetyl sulfur ylides, are derived from commercially available precursors such as acyl chlorides and hydrazines, ensuring a stable and diversified supply base. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by utility fluctuations or environmental controls, guaranteeing consistent output. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, reducing the risk of stockouts and enabling more accurate demand forecasting. The scalability of the method from gram to kilogram levels further supports flexible production planning to meet market demands.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproducts are non-toxic and easily treatable, aligning with increasingly strict environmental regulations in the chemical industry. The absence of heavy metals simplifies the regulatory approval process for new drug filings, as residual metal testing is less burdensome. Scaling this reaction to multi-kilogram or tonnage levels is straightforward due to the lack of exothermic hazards associated with strong oxidants or unstable intermediates. This environmental and operational safety profile facilitates faster technology transfer from R&D to commercial production sites, accelerating time-to-market for new pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis platform. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers evaluating this technology for adoption. Understanding these specifics helps in assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process explicitly avoids the use of heavy metal catalysts, utilizing inexpensive potassium carbonate instead, which significantly simplifies downstream purification and reduces heavy metal residue risks.
Q: What are the typical reaction conditions for this triazine formation?
A: The reaction proceeds efficiently under mild conditions, specifically at temperatures ranging from 20°C to 40°C in an air atmosphere, eliminating the need for inert gas protection or extreme heating.
Q: Is this method suitable for large-scale pharmaceutical intermediate production?
A: Yes, the use of cheap, non-toxic reagents and the absence of stringent anhydrous or anaerobic requirements make this protocol highly adaptable for commercial scale-up and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocyclic chemistry in the development of advanced pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into reliable industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoromethyl substituted 1,2,4-triazine compounds meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains for critical drug substances.
We invite you to collaborate with us to leverage this innovative metal-free synthesis for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain for high-purity pharmaceutical intermediates.
