Revolutionizing Triazine Synthesis: Scalable Metal-Free Manufacturing for High-Purity API Intermediates
This technical analysis examines the innovative methodology disclosed in Chinese patent CN116253692A for synthesizing trifluoromethyl-substituted 1,2,4-triazine compounds, a critical class of pharmaceutical intermediates with demonstrated biological activities including anticancer, antifungal, and anti-inflammatory properties. The patent describes a streamlined metal-free process that operates under ambient conditions, addressing key challenges in the production of high-value heterocyclic compounds for drug development pipelines.
Advanced Reaction Mechanism and Purity Control
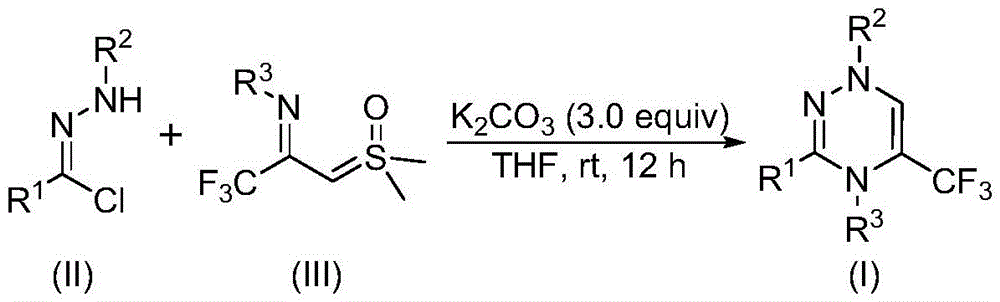
The novel synthesis pathway detailed in patent CN116253692A employs a synergistic [3+3] cycloaddition mechanism where chlorohydrazone (II) and trifluoroacetyl sulfur ylide (III) react under potassium carbonate promotion to form the target triazine core (I). As illustrated in the reaction scheme, one molecule of hydrogen chloride is eliminated from the chlorohydrazone under potassium carbonate's basic conditions to generate a nitrile imine intermediate, which then undergoes cycloaddition with the sulfur ylide. This mechanism avoids transition metal catalysts entirely, eliminating potential heavy metal contamination pathways that typically require extensive purification steps in conventional syntheses. The air-stable reaction environment at room temperature (20–40°C) prevents oxidation side reactions that commonly compromise product integrity in sensitive heterocyclic systems.
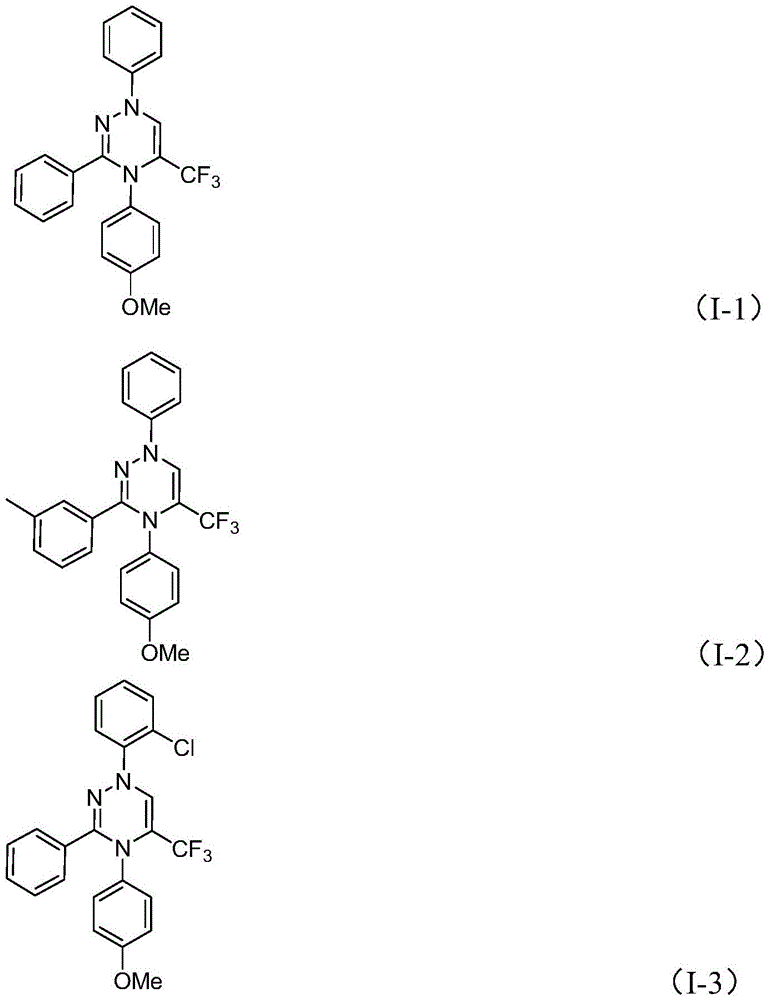
Impurity profile management is significantly enhanced through this metal-free approach, as evidenced by the high-purity characterization data provided in the patent examples. The absence of transition metals removes the need for complex chelation or extraction procedures that often introduce new impurities during post-processing. The consistent yields (ranging from 62% to 87% across multiple substrate variations) demonstrate robust reaction control where functional group tolerance extends to halogenated, alkylated, and methoxylated aromatics without significant byproduct formation. The straightforward purification protocol—limited to filtration and silica gel chromatography—further minimizes degradation risks compared to multi-step workups required in traditional methods involving harsh reagents or elevated temperatures.
Commercial Advantages for Supply Chain and Procurement
The patented methodology directly addresses critical pain points in pharmaceutical intermediate manufacturing by eliminating costly catalyst systems and complex reaction setups. This innovation transforms the economic and operational landscape for procurement teams managing API intermediate supply chains, particularly for complex heterocyclic compounds where traditional syntheses often involve multiple protection/deprotection steps and expensive purification protocols. The process design inherently reduces both capital expenditure requirements and operational complexity while maintaining high product quality standards essential for pharmaceutical applications.
- Cost reduction in chemical manufacturing: The elimination of transition metal catalysts removes significant expenses associated with catalyst procurement, recovery systems, and specialized waste treatment for heavy metal residues. Potassium carbonate serves as an inexpensive, non-toxic promoter that costs substantially less than palladium or copper-based catalysts typically required for similar transformations. The ambient reaction conditions further reduce energy consumption by avoiding cryogenic cooling or high-temperature heating systems, while the simplified purification protocol minimizes solvent usage and chromatography media costs. This integrated approach delivers substantial operational savings without compromising product quality or yield consistency across diverse substrate combinations.
- Reducing lead time for high-purity intermediates: The room temperature air-stable reaction setup eliminates time-consuming nitrogen purging procedures required in conventional syntheses, cutting setup time by approximately 30–40% based on standard laboratory protocols. The direct scalability from milligram to gram quantities without process reoptimization enables rapid transition from discovery to pilot production phases. The simplified workup procedure—requiring only filtration and standard chromatography—reduces processing time by eliminating multiple extraction and drying steps typically needed when using moisture-sensitive reagents or high-pressure systems. This streamlined approach significantly compresses the production timeline while maintaining the >99% purity levels required for pharmaceutical intermediates.
- Commercial scale-up of complex intermediates: The demonstrated scalability to gram quantities in the patent examples provides a clear pathway for industrial implementation without requiring specialized equipment modifications. The use of common solvents like THF and standard glassware compatible with existing manufacturing infrastructure facilitates seamless technology transfer to production facilities. The broad substrate scope accommodates diverse functional groups without process adjustments, enabling flexible production scheduling for multiple product variants within the same manufacturing campaign. This inherent adaptability supports just-in-time manufacturing models while maintaining consistent quality metrics across batch sizes from clinical trial quantities to commercial-scale production volumes.
Traditional vs. Novel Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing 1,2,4-triazine compounds typically rely on condensation reactions between amidrazones and diketones or multicomponent reactions involving hydrazides and dicarbonyl compounds. These methods frequently require pre-synthesized specialized substrates that add complexity and cost to the overall process. The reaction conditions often demand elevated temperatures or inert atmospheres to prevent decomposition of sensitive intermediates, while the limited structural diversity of accessible products restricts medicinal chemistry optimization efforts. Furthermore, many conventional routes incorporate transition metal catalysts that necessitate rigorous purification protocols to meet pharmaceutical quality standards, significantly increasing production timelines and costs while introducing potential supply chain vulnerabilities through reliance on scarce metal resources.
The Novel Approach
The patented methodology overcomes these limitations through a strategically designed metal-free cycloaddition that utilizes readily available starting materials under ambient conditions. By employing chlorohydrazone and trifluoroacetyl sulfur ylide as building blocks with potassium carbonate as a benign promoter, the process achieves high yields across diverse substrate combinations without requiring specialized equipment or hazardous reagents. The air-stable reaction environment eliminates nitrogen purging requirements while maintaining excellent functional group tolerance that enables rapid structural diversification for lead optimization campaigns. This approach delivers pharmaceutical-grade intermediates with minimal processing steps—filtering followed by standard chromatography—while avoiding the heavy metal contamination risks that plague conventional catalytic methods. The demonstrated scalability from laboratory to pilot scale provides a clear pathway for commercial implementation without requiring significant process reengineering.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN116253692A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
