Advanced 5-Step Synthesis Strategy for Darolutamide Intermediate Manufacturing
The global pharmaceutical landscape is witnessing a surge in demand for advanced oncology treatments, particularly for prostate cancer therapies like Darolutamide. As a nonsteroidal Androgen Receptor antagonist, Darolutamide represents a critical intervention for non-metastatic castration-resistant prostate cancer. However, the commercial viability of such potent active pharmaceutical ingredients often hinges on the efficiency of their synthesis. Patent CN111116477B introduces a transformative synthesis process that addresses the historical bottlenecks of long reaction sequences and low overall yields. This technical insight report analyzes the novel 5-step pathway, contrasting it with traditional multi-step methodologies to highlight its potential for industrial adoption. By leveraging commercially available starting materials and optimizing reaction conditions, this process offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks seeking to enhance their portfolio with high-value oncology compounds.
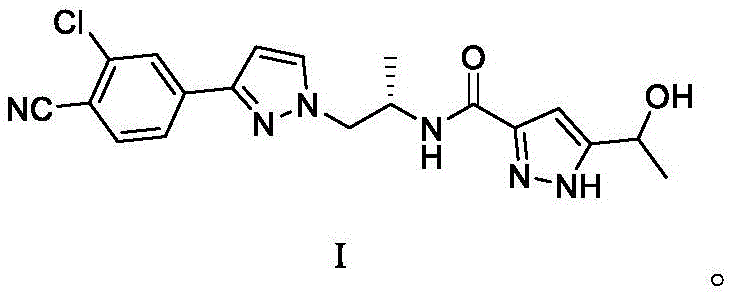
Darolutamide, chemically defined by Formula I, possesses a complex architecture featuring a chlorobenzonitrile moiety linked to a pyrazole ring and a hydroxy-propyl amide side chain. The structural integrity of this molecule is paramount for its biological activity, necessitating a synthesis route that minimizes impurity formation while maximizing throughput. The patent data reveals a strategic approach to constructing this scaffold, moving away from the cumbersome >10 step routes previously documented in the art. Instead, the inventors have curated a concise sequence involving amino protection, halogenation, amidation, and a final stereoselective reduction. This shift not only simplifies the chemical logic but also aligns with modern green chemistry principles by reducing solvent usage and waste generation associated with excessive purification steps between numerous intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex androgen receptor antagonists has been plagued by inefficient linear sequences that exceed 10 discrete reaction steps. These elongated pathways introduce significant cumulative yield losses, where even high-yielding individual steps result in a dismal overall recovery of the final active ingredient. Furthermore, each additional step necessitates isolation, purification, and quality control procedures, drastically inflating the cost of goods sold (COGS) and extending the manufacturing lead time. From a supply chain perspective, reliance on obscure or custom-synthesized precursors for every step creates fragility; a disruption in the supply of a niche intermediate can halt the entire production line. Additionally, traditional routes often employ harsh reagents or transition metal catalysts that require rigorous removal protocols to meet stringent regulatory limits for residual metals in pharmaceutical products, adding further complexity and cost to the downstream processing.
The Novel Approach
The methodology outlined in CN111116477B disrupts this status quo by condensing the synthesis into a highly efficient 5-step sequence. This streamlined approach begins with the strategic protection of a commercially available amine (Compound 7) to prevent side reactions during subsequent alkylation. The core scaffold is assembled through a robust nucleophilic substitution followed by an amide coupling that utilizes an activated acid chloride intermediate. A key innovation lies in the telescoping of the deprotection and coupling steps, which eliminates the need to isolate the unstable free amine intermediate (Compound 4), thereby reducing handling time and exposure to potential degradation. The final transformation employs a mild hydride reduction to install the crucial hydroxyl group with high fidelity. This reduction in step count directly translates to improved industrial operability, allowing for faster batch turnover and a significantly higher total yield compared to legacy processes.
Mechanistic Insights into the 5-Step Synthetic Cascade
The chemical elegance of this process is rooted in its precise control over functional group interconversions. The initial step involves the protection of (R)-1-bromo-3-aminobutane (Compound 7) with di-tert-butyl dicarbonate ((Boc)2O) in the presence of triethylamine. This generates the N-Boc protected alkyl bromide (Compound 2), which serves as a stable electrophile for the subsequent alkylation. The reaction of Compound 2 with the pyrazole derivative (Compound 1) proceeds via an SN2 mechanism, facilitated by DIPEA as a base, to form the C-N bond linking the chiral side chain to the heterocyclic core. Following this, the carboxylic acid precursor (Compound 8) is activated using oxalyl chloride ((COCl)2) with catalytic DMF to generate the reactive acid chloride (Compound 5). This activation is critical for driving the subsequent amide bond formation under mild conditions.
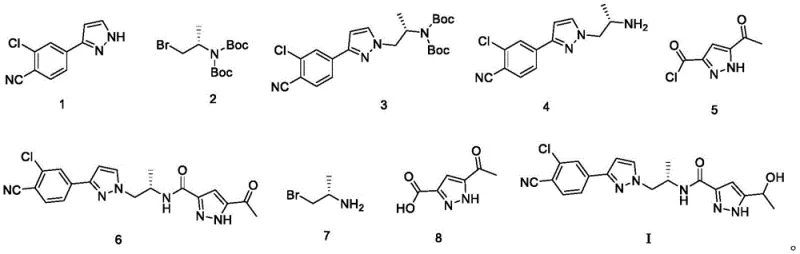
The convergence of the two major fragments occurs in the fourth stage, where the Boc group of Compound 3 is removed using hydrochloric acid in dioxane to reveal the free amine (Compound 4). Without isolation, this amine immediately reacts with the pre-formed acid chloride (Compound 5) to construct the central amide linkage, yielding the keto-intermediate (Compound 6). The final mechanistic event is the chemoselective reduction of the ketone moiety in Compound 6. Using sodium borohydride (NaBH4) in ethanol, the carbonyl group is reduced to the secondary alcohol found in Darolutamide (Formula I). The patent specifies a molar ratio of Compound 6 to NaBH4 of approximately 1:1.3, ensuring complete conversion while minimizing excess reagent waste. This sequence demonstrates a masterful balance of reactivity, where protecting groups are used only when necessary and removed efficiently to drive the synthesis forward.
How to Synthesize Darolutamide Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the exothermic activation and reduction phases. The process is designed to be scalable, utilizing common solvents like dichloromethane and ethanol which are easily recovered and recycled in a GMP environment. The following guide outlines the operational framework derived from the patent examples, providing a roadmap for process chemists to replicate these results. For the detailed standardized synthesis steps including specific quantities and workup procedures, please refer to the structured guide below.
- Protect the amine group of compound 7 using di-tert-butyl dicarbonate to form compound 2.
- Alkylate compound 1 with compound 2 to generate the protected intermediate compound 3.
- Convert carboxylic acid compound 8 into acid chloride compound 5 using oxalyl chloride.
- Deprotect compound 3 to amine 4, then couple with acid chloride 5 to form keto-amide compound 6.
- Reduce the ketone group in compound 6 using sodium borohydride to yield final Darolutamide (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from a 10+ step process to a 5-step protocol represents a paradigm shift in cost structure and risk management. The primary driver of value here is the drastic reduction in unit operations. Fewer steps mean less solvent consumption, lower energy usage for heating and cooling, and reduced labor hours for reactor charging and product isolation. This efficiency gain allows for a substantial reduction in manufacturing costs without compromising the quality of the final API intermediate. Moreover, the reliance on commercially readily available starting materials mitigates the risk of supply chain disruptions. Unlike proprietary intermediates that may be sourced from a single vendor, the precursors identified in this patent (Compounds 1, 7, and 8) are accessible through multiple global suppliers, fostering a competitive pricing environment and ensuring continuity of supply.
- Cost Reduction in Manufacturing: The elimination of five or more reaction steps fundamentally alters the economic model of production. By removing the need for multiple isolation and purification stages, the process significantly reduces the consumption of chromatography media and solvents, which are often the largest variable costs in fine chemical synthesis. Furthermore, the high yield reported in each step (e.g., >90% for the alkylation and >87% for the coupling) ensures that raw material utilization is maximized, minimizing the cost of wasted inputs. The avoidance of expensive transition metal catalysts also removes the necessity for costly scavenging resins or specialized filtration equipment, further driving down the capital and operational expenditure required for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by simplifying the logistics of raw material management. With fewer intermediates to stock and manage, inventory carrying costs are reduced, and the lead time for production batches is shortened. The use of standard reagents like oxalyl chloride and sodium borohydride ensures that the process is not dependent on fragile supply lines for exotic chemicals. This stability is crucial for maintaining the continuous flow of high-purity Darolutamide to downstream formulation partners, preventing stock-outs that could impact patient access to critical medication.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the shorter route generates less chemical waste, aligning with increasingly stringent global regulations on pharmaceutical effluent. The simplified workflow facilitates easier commercial scale-up of complex pharmaceutical intermediates, as the heat transfer and mixing requirements are less demanding than those of a protracted multi-step synthesis. The ability to telescope the deprotection and coupling steps not only saves time but also reduces the volume of hazardous waste generated from intermediate washes. This operational simplicity makes the process highly attractive for large-scale production facilities aiming to optimize their environmental footprint while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on yield expectations, purity profiles, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this new synthesis route compare to conventional methods in terms of step count?
A: Conventional synthesis routes for Darolutamide typically involve more than 10 reaction steps, which increases operational complexity and material loss. The patented process described in CN111116477B streamlines this significantly to just 5 key steps, thereby improving total yield and industrial operability.
Q: What are the critical reaction conditions for the final reduction step?
A: The final reduction of compound 6 to Darolutamide utilizes sodium borohydride (NaBH4) in ethanol. The reaction is initiated at 0°C and stirred at room temperature, followed by quenching with sodium bicarbonate solution to ensure high purity (>99%) and optimal yield.
Q: Are the starting materials for this process commercially available?
A: Yes, the process relies on commercially readily available intermediate compounds such as compound 1, compound 7, and compound 8. This availability supports a robust supply chain and reduces the risk of raw material shortages during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Darolutamide Supplier
The synthesis process detailed in CN111116477B exemplifies the type of innovative chemical engineering that drives value in the modern pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant manufacturing processes for oncology intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific unit operations required for this route, including cryogenic reactions for the acid chloride formation and controlled reductions, all while adhering to stringent purity specifications and rigorous QC labs to ensure every batch meets the highest international standards.
We invite procurement leaders and R&D directors to collaborate with us to leverage this optimized synthesis for your Darolutamide projects. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this 5-step route can improve your margins. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and comprehensive route feasibility assessments. By partnering with us, you secure a supply chain that is not only cost-effective but also technically robust and ready for immediate scale-up.
