Advanced 5-Step Synthesis of Darolutamide for Commercial API Manufacturing
Advanced 5-Step Synthesis of Darolutamide for Commercial API Manufacturing
The pharmaceutical landscape for prostate cancer treatment has been significantly advanced by the introduction of nonsteroidal Androgen Receptor antagonists, specifically Darolutamide. Patent CN111116477A discloses a groundbreaking synthesis process that addresses the critical inefficiencies found in legacy manufacturing routes. While traditional methods often necessitate more than ten discrete reaction steps, resulting in compounded yield losses and extensive purification burdens, this novel approach streamlines the entire workflow into a concise five-step sequence. This reduction in synthetic complexity not only enhances the overall reaction yield but also drastically simplifies the operational requirements for industrial production. By leveraging commercially readily available intermediate compounds and employing strategic protection and deprotection strategies, the process ensures a robust supply of high-purity active pharmaceutical ingredients. The technical breakthrough lies in the efficient construction of the core molecular architecture, minimizing waste generation and maximizing atom economy throughout the synthetic pathway.
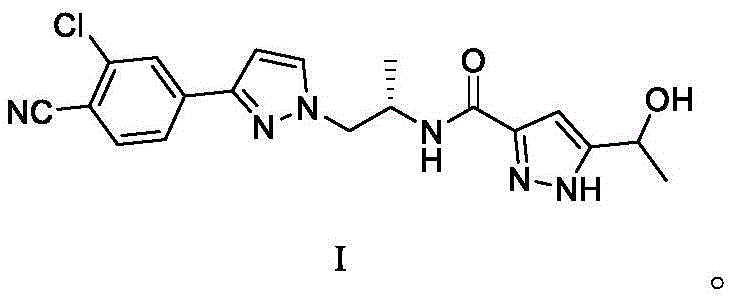
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of complex oncology intermediates like Darolutamide has been plagued by lengthy synthetic routes that hinder cost-effective production. Conventional methodologies typically involve more than ten reaction steps, each introducing potential points of failure, yield degradation, and impurity accumulation. The reliance on multiple protection and deprotection cycles in older routes often requires harsh reagents and stringent conditions that are difficult to maintain consistently on a large scale. Furthermore, the cumulative effect of low yields in each individual step results in a dismal overall throughput, driving up the cost of goods sold and limiting supply availability. The extensive purification required between numerous steps also consumes significant volumes of solvents and stationary phases, creating environmental burdens and increasing the operational footprint. These inefficiencies pose substantial risks to supply chain continuity, making it challenging for procurement teams to secure reliable volumes of high-purity pharmaceutical intermediates without incurring excessive lead times.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the synthesis method disclosed in patent CN111116477A offers a streamlined pathway that fundamentally restructures the production logic. By condensing the synthesis into just five high-yielding steps, the new route eliminates unnecessary chemical transformations that do not contribute directly to the final molecular assembly. This approach utilizes readily accessible starting materials, such as specific pyrazole derivatives and protected amine precursors, which reduces dependency on custom-synthesized building blocks. The operational simplicity is further enhanced by the use of standard reaction conditions, including ambient temperature stirrings and common solvent systems like dichloromethane and ethanol. The strategic design of the route ensures that each intermediate is stable and easily isolable, facilitating rigorous quality control at every stage. This modernization of the synthetic strategy effectively lowers the barrier to entry for commercial manufacturing, enabling a more agile and responsive supply chain for this critical antitumor drug.
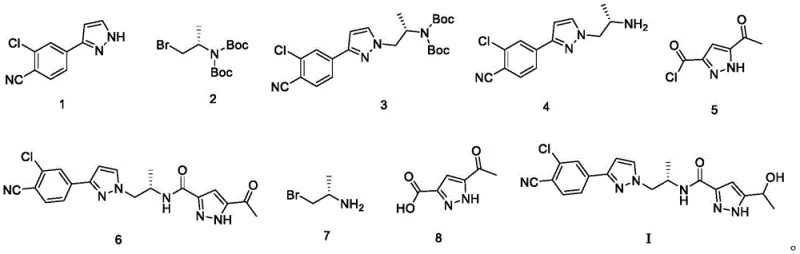
Mechanistic Insights into Boc-Protection and Amidation Strategy
The core of this synthetic innovation relies on a sophisticated sequence of protection, coupling, and reduction reactions that ensure high fidelity in molecular construction. The process initiates with the protection of a primary amine using di-tert-butyl dicarbonate ((Boc)2O) in the presence of triethylamine, forming a stable carbamate intermediate that prevents unwanted side reactions during subsequent alkylation. This protection step is critical for maintaining the integrity of the nitrogen center, which is later revealed through controlled acidic deprotection using hydrochloric acid in dioxane. The subsequent amidation step involves the activation of a carboxylic acid precursor into a reactive acid chloride using oxalyl chloride, which then couples efficiently with the deprotected amine to form the key amide bond. The final transformation utilizes sodium borohydride to selectively reduce a ketone moiety to the corresponding secondary alcohol, completing the pharmacophore without affecting other sensitive functional groups. This mechanistic precision ensures that the final product retains the necessary stereochemistry and functional group arrangement required for biological activity.
Impurity control is inherently built into the design of this reaction sequence through the selection of specific reagents and purification protocols. The use of column chromatography with optimized solvent systems, such as mixtures of petroleum ether and ethyl acetate, allows for the effective separation of closely related by-products and unreacted starting materials. The high yields observed in each step, ranging from approximately 85% to over 93%, indicate a clean reaction profile with minimal formation of difficult-to-remove impurities. Furthermore, the final reduction step is carefully quenched and worked up to ensure that boron residues are completely removed, contributing to the high HPLC purity of the final Darolutamide compound. The robustness of the mechanism allows for consistent reproduction of results, which is essential for meeting the stringent regulatory requirements of the pharmaceutical industry. By minimizing the generation of complex impurity profiles, the process reduces the analytical burden and accelerates the release of batches for clinical or commercial use.
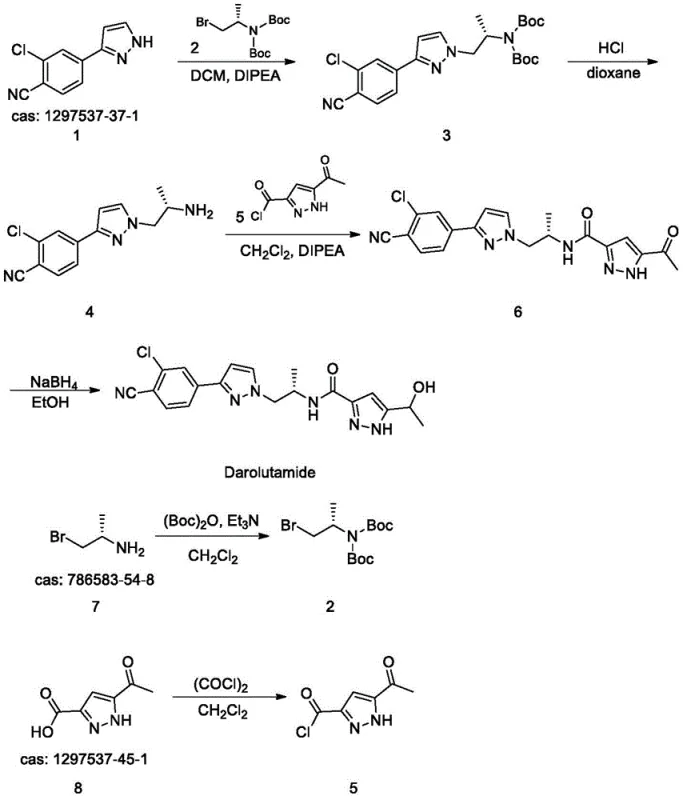
How to Synthesize Darolutamide Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters and stoichiometry to maximize the efficiency of each transformation. The process begins with the preparation of the protected amine intermediate, followed by coupling with the pyrazole core, and concludes with the final reduction to the target alcohol. Detailed standard operating procedures for each step, including specific molar ratios, temperature controls, and work-up methods, are essential for replicating the high yields reported in the patent data. Operators must ensure strict anhydrous conditions during the acid chloride formation and maintain precise temperature profiles during the reduction phase to prevent over-reduction or side reactions. The following guide outlines the critical operational milestones required to execute this pathway successfully in a pilot or production environment.
- Protect the amine group of the starting material using (Boc)2O and triethylamine in dichloromethane to form the protected intermediate.
- Perform alkylation with the pyrazole derivative followed by deprotection using hydrochloric acid to reveal the reactive amine.
- Convert the carboxylic acid precursor to an acid chloride using oxalyl chloride, then couple with the amine and reduce the ketone using NaBH4.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this streamlined synthesis route offers profound advantages for procurement managers and supply chain directors seeking to optimize their API sourcing strategies. The reduction in reaction steps directly correlates to a significant decrease in manufacturing costs, as fewer unit operations translate to lower labor, energy, and solvent consumption. By eliminating the need for lengthy multi-step sequences, the process inherently reduces the risk of batch failures and production delays, thereby enhancing the reliability of supply. The use of commercially available starting materials mitigates the risk of raw material shortages, ensuring a more stable and predictable procurement pipeline. Additionally, the simplified workflow facilitates faster technology transfer and scale-up, allowing manufacturers to respond more quickly to market demand fluctuations. These operational efficiencies collectively contribute to a more resilient supply chain capable of sustaining long-term production of this vital oncology medication.
- Cost Reduction in Manufacturing: The consolidation of the synthesis into five steps fundamentally alters the cost structure of Darolutamide production by removing the overhead associated with intermediate isolation and purification in longer routes. Eliminating transition metal catalysts or exotic reagents in favor of standard organic synthesis tools further drives down the cost of raw materials and waste disposal. The high yield at each stage ensures that less starting material is required to produce the same amount of final product, effectively lowering the material cost per kilogram. This efficiency allows for substantial cost savings that can be passed down the supply chain, making the treatment more accessible while maintaining healthy margins for manufacturers. The reduction in solvent usage and processing time also contributes to a lower overall environmental footprint, aligning with modern sustainability goals in chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commercially readily available intermediates significantly de-risks the supply chain by reducing dependency on custom synthesis providers with long lead times. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent output. This stability is crucial for maintaining continuous supply to downstream formulators and preventing drug shortages that could impact patient care. The simplified process also allows for greater flexibility in sourcing, as multiple suppliers can potentially manufacture the key intermediates without requiring specialized proprietary technology. This diversification of the supply base strengthens the overall resilience of the procurement strategy against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The operational simplicity of the route, involving standard stirring and washing techniques, makes it highly amenable to scale-up from laboratory to industrial production volumes. The avoidance of hazardous reagents and the use of common solvents simplify the waste management process, ensuring compliance with increasingly strict environmental regulations. The high atom economy of the synthesis minimizes the generation of chemical waste, reducing the burden on effluent treatment facilities and lowering disposal costs. This scalability ensures that the process can meet the growing global demand for Darolutamide without requiring massive capital investment in new specialized equipment. The alignment with green chemistry principles further enhances the marketability of the product to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Darolutamide intermediates. These answers are derived from the specific technical disclosures and beneficial effects outlined in the patent literature, providing clarity on the process capabilities. Understanding these details is essential for R&D and procurement teams evaluating the feasibility of this route for their specific manufacturing needs. The information provided here serves as a foundational guide for further technical discussions and feasibility assessments.
Q: How does this new synthesis route improve upon conventional methods for Darolutamide?
A: The conventional synthesis routes often exceed 10 reaction steps, leading to cumulative yield losses and complex purification requirements. This novel patent CN111116477A introduces a concise 5-step pathway that utilizes commercially available intermediates, significantly reducing operational complexity and improving total yield.
Q: What are the critical purity specifications achievable with this process?
A: The described process demonstrates robust impurity control, achieving HPLC purity levels up to 99.6% in the final reduction step. The use of specific solvent systems like dichloromethane and methanol during chromatography ensures the removal of side products effectively.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed with industrial operability in mind. It avoids exotic reagents and utilizes standard unit operations such as stirring, washing, and column chromatography, making it highly adaptable for commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Darolutamide Supplier
The technical potential of this 5-step synthesis route represents a significant opportunity for optimizing the production of Darolutamide and its key intermediates. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to fruition. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the highest standards of quality and consistency. We understand the critical nature of oncology supply chains and are committed to delivering reliable volumes of high-purity pharmaceutical intermediates to support global health initiatives. Our team is ready to leverage this patent technology to provide a competitive edge in the manufacturing of this vital antitumor agent.
We invite you to initiate a dialogue regarding your specific supply chain requirements and explore how this optimized synthesis can benefit your production goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to execute this chemistry at scale. Partnering with us ensures access to a robust, compliant, and cost-effective supply of Darolutamide intermediates, securing your position in the competitive pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
