Advanced Pd-Catalyzed Synthesis of Chiral 3,4-Dihydro-2(1H)-Quinolinones for Commercial Scale-Up
Introduction to Next-Generation Quinolinone Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for privileged scaffolds that exhibit broad biological activity. Among these, chiral 3,4-dihydro-2(1H)-quinolinones stand out as critical intermediates due to their presence in numerous bioactive molecules possessing anti-tumor, anti-hypertensive, and analgesic properties. A significant breakthrough in this domain is documented in patent CN109354583B, which discloses a novel, efficient diastereoselective synthesis method. This technology leverages a palladium-catalyzed [4+2] cycloaddition between vinyl benzoxazinones and oxazolones, offering a streamlined pathway to complex heterocyclic systems. Unlike traditional methods that often require harsh conditions or multiple steps, this approach operates under mild parameters, utilizing accessible building blocks to construct molecules with two contiguous chiral centers. For R&D teams and procurement specialists, understanding this methodology is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity compounds with improved cost-efficiency.
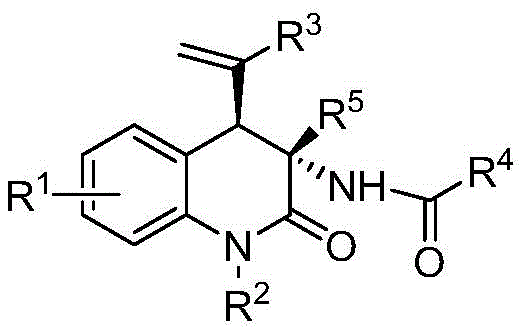
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the stereoselective synthesis of 3,4-dihydro-2(1H)-quinolinones has been fraught with challenges that impede efficient commercial production. Conventional routes often rely on multi-step sequences involving expensive chiral auxiliaries or resolution processes that inherently limit overall yield and increase waste generation. Many existing protocols necessitate elevated temperatures, strong acids or bases, and inert atmospheres that complicate reactor operations and safety profiles. Furthermore, controlling stereochemistry at multiple centers simultaneously remains a persistent hurdle, frequently resulting in mixtures of diastereomers that require difficult and costly separation techniques. These limitations not only extend lead times for high-purity pharmaceutical intermediates but also inflate the cost of goods sold (COGS), making it difficult for manufacturers to remain competitive in a price-sensitive market. The environmental footprint of these older methods, characterized by poor atom economy and hazardous waste streams, further complicates regulatory compliance and sustainability goals.
The Novel Approach
In stark contrast, the methodology described in patent CN109354583B introduces a paradigm shift by employing a direct, catalytic assembly strategy. This novel approach utilizes vinyl benzoxazinones and oxazolones as versatile synthetic building blocks, which undergo a synergistic transformation mediated by a palladium catalyst and a phosphorus-containing ligand. The reaction proceeds smoothly at room temperature in common organic solvents such as dichloromethane, eliminating the need for energy-intensive heating or cooling cycles. By generating a highly reactive allylpalladium zwitterion intermediate in situ, the system facilitates a rapid [4+2] cycloaddition that constructs the quinolinone core with excellent diastereoselectivity. This efficiency translates directly into simplified post-treatment procedures, where the crude product can often be purified via standard column chromatography. For supply chain managers, this represents a substantial opportunity for cost reduction in API manufacturing, as the process minimizes unit operations and maximizes throughput without compromising on the structural integrity or purity of the final active pharmaceutical ingredient.
Mechanistic Insights into Pd-Catalyzed [4+2] Cycloaddition
To fully appreciate the technical superiority of this synthesis, one must examine the underlying catalytic cycle. The reaction initiates with the coordination of the palladium catalyst to the vinyl benzoxazinone substrate. Under the influence of the phosphorus ligand, the vinyl benzoxazinone undergoes an intramolecular decarboxylation, a critical step that generates a highly reactive allylpalladium zwitterionic species. This transient intermediate acts as a four-atom synthon, possessing significant electrophilic character that drives the subsequent bond-forming events. The oxazolone, acting as a nucleophilic partner, attacks this activated complex, leading to the formation of new carbon-carbon and carbon-nitrogen bonds in a concerted manner. The presence of an acid-base additive, such as benzoic acid, plays a pivotal role in stabilizing transition states and facilitating proton transfer steps, thereby ensuring high conversion rates and selectivity. This mechanistic elegance allows for the construction of complex molecular architectures containing multiple functional groups and contiguous stereocenters in a single operation.
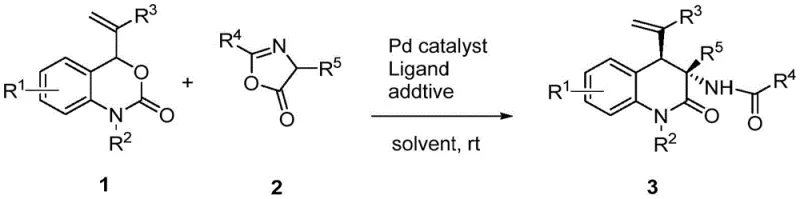
From an impurity control perspective, the specificity of this catalytic cycle is paramount. The well-defined coordination sphere around the palladium center dictates the spatial orientation of the approaching reactants, effectively suppressing the formation of unwanted regioisomers or diastereomers. This intrinsic selectivity reduces the burden on downstream purification processes, which is a key consideration for maintaining stringent purity specifications required by global regulatory bodies. The robustness of the catalyst system also means that side reactions, such as polymerization of the vinyl group or hydrolysis of the oxazolone, are minimized. For quality assurance teams, this predictability ensures batch-to-batch consistency, a critical factor when scaling from laboratory grams to commercial metric tons. The ability to tolerate a wide range of substituents on both the benzoxazinone and oxazolone rings further underscores the versatility of this mechanism, enabling the synthesis of diverse libraries for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new analog.
How to Synthesize Chiral 3,4-Dihydro-2(1H)-Quinolinones Efficiently
Implementing this synthesis in a practical setting requires adherence to specific operational parameters to maximize yield and reproducibility. The process begins by charging a reaction vessel with the vinyl benzoxazinone and oxazolone substrates in a molar ratio typically favoring the oxazolone slightly, often around 1:1.2, to drive the equilibrium towards product formation. A palladium source, such as tris(dibenzylideneacetone)dipalladium-chloroform adduct, is introduced alongside a phosphine ligand like triphenylphosphine. The choice of solvent is flexible but dichloromethane is often preferred for its ability to dissolve both organic substrates and the catalyst system effectively. An acid additive, commonly benzoic acid, is added to modulate the reaction environment. The mixture is then stirred at ambient temperature, with reaction progress monitored by thin-layer chromatography (TLC). Upon completion, usually within 2 to 6 hours, the workup involves simple filtration and concentration, followed by purification via silica gel chromatography.
- Combine vinyl benzoxazinone and oxazolone substrates with a palladium catalyst (e.g., Pd2(dba)3·CHCl3) and a phosphorus ligand (e.g., PPh3) in an organic solvent like dichloromethane.
- Add an acid-base additive such as benzoic acid to facilitate the reaction progress and stir the mixture at room temperature for 2 to 6 hours.
- Monitor reaction completion via TLC, then purify the crude product through simple column chromatography using an ethyl acetate and petroleum ether mixture to isolate the target quinolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing workflow. By consolidating what was previously a multi-step sequence into a single catalytic event, the process significantly reduces the number of unit operations, equipment occupancy time, and labor costs associated with production. This streamlining directly contributes to cost reduction in pharmaceutical intermediates manufacturing, allowing for more competitive pricing structures without sacrificing margin. Furthermore, the use of commercially available starting materials mitigates supply risk; vinyl benzoxazinones and oxazolones are established building blocks with stable supply chains, ensuring continuity of supply even during market fluctuations. The mild reaction conditions also imply lower energy consumption, aligning with corporate sustainability targets and reducing the overall carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the reduction in solvent usage due to fewer processing steps lead to substantial cost savings. The low catalyst loading required (often in the range of 5 mol%) further decreases the raw material cost per kilogram of product. Additionally, the simplified purification protocol reduces the consumption of silica gel and eluents, which are significant cost drivers in fine chemical production. These cumulative efficiencies allow for a more lean manufacturing model, optimizing capital expenditure and operational expenditure simultaneously.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to variations in utility supply, such as steam or chilled water, which can be bottlenecks in multi-purpose plants. The wide substrate scope ensures that a single platform technology can be used to produce a variety of derivatives, increasing asset utilization rates. This flexibility allows suppliers to respond rapidly to changing customer demands for different analogs, reducing lead times for high-purity pharmaceutical intermediates. The stability of the reagents also simplifies logistics and storage requirements, lowering inventory carrying costs and minimizing the risk of raw material degradation.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is straightforward due to the absence of exothermic hazards associated with strong reagents or high temperatures. The benign nature of the byproducts and the potential for solvent recovery enhance the environmental profile of the process. This ease of scale-up ensures that commercial quantities can be produced reliably, supporting the transition from clinical trials to commercial launch without the need for extensive process re-engineering. Compliance with environmental regulations is facilitated by the reduced waste generation, making it easier to obtain necessary permits and maintain good standing with regulatory agencies.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is vital for stakeholders evaluating its potential for their specific projects. The following questions address common inquiries regarding the technical feasibility and commercial viability of this synthesis route. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for decision-making. Whether you are concerned about impurity profiles, catalyst removal, or substrate compatibility, the following insights aim to clarify the operational realities of implementing this chemistry in a GMP environment.
Q: What are the key advantages of this Pd-catalyzed method over traditional quinolinone synthesis?
A: This method operates at room temperature with mild conditions, avoiding harsh reagents. It offers high diastereoselectivity, simple post-treatment via column chromatography, and utilizes readily available starting materials, significantly reducing operational complexity.
Q: What is the substrate scope for this reaction?
A: The process demonstrates a wide substrate range, accommodating various substituents on the benzene ring (fluoro, chloro, bromo, methyl) and different groups on the oxazolone moiety, allowing for the synthesis of diverse structural analogs.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of low catalyst loading, ambient temperature conditions, and simple purification steps makes this route highly amenable to commercial scale-up, ensuring consistent quality and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydro-2(1H)-Quinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development pipelines. Our team of expert chemists has thoroughly analyzed the methodology described in patent CN109354583B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex catalytic transformations ensures that we can translate this innovative laboratory protocol into a robust, industrial-grade process that guarantees supply security for your long-term projects.
We invite you to engage with our technical procurement team to discuss how this specific synthesis route can be tailored to your unique requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing method is seamless and scientifically sound. Let us be your trusted partner in bringing next-generation therapeutics to market efficiently.
