Advanced Manufacturing Strategy for High-Purity Aripiprazole Intermediates via Optimized Condensation
Advanced Manufacturing Strategy for High-Purity Aripiprazole Intermediates via Optimized Condensation
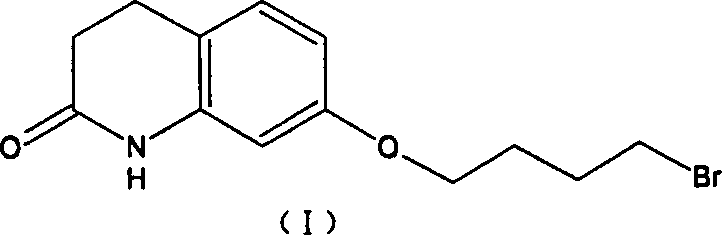
The pharmaceutical industry continuously seeks robust synthetic pathways that balance chemical efficiency with commercial viability, particularly for high-volume antipsychotic agents like Aripiprazole. Patent CN1289484C introduces a transformative preparation method that fundamentally restructures the synthesis of 7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl]butoxy]-3,4-dihydro-2(1H)-quinolinone. Unlike traditional approaches that rely on unstable di-haloalkanes prone to hydrolysis, this innovation leverages a sequential alkylation and sulfonylation strategy to construct the critical ether linkage. By replacing the problematic 1,4-dibromobutane with 4-halo-1-butanol derivatives and utilizing aryl sulfonyl halides as activating agents, the process achieves mild reaction conditions and exceptional impurity control. This technical breakthrough not only streamlines the operational workflow but also ensures that the final active pharmaceutical ingredient meets stringent quality standards without the need for resource-intensive chromatographic purification, representing a significant leap forward for reliable API intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Aripiprazole, as described in earlier patents like EP367141, relied heavily on the condensation of 7-hydroxy-3,4-dihydroquinolinone with 1,4-dibromobutane to form a bromo-butoxy intermediate. This legacy approach presents severe chemical engineering challenges, primarily due to the susceptibility of 1,4-dibromobutane to hydrolysis under the requisite alkaline reflux conditions. When water is used as a solvent or co-solvent in these basic environments, competitive hydrolysis reactions occur rapidly, generating hydroxy-butoxy byproducts that are structurally similar to the desired intermediate and notoriously difficult to separate. Consequently, the purification of this key intermediate becomes a bottleneck, often requiring complex extraction protocols or repeated recrystallizations that drastically erode overall yield. Furthermore, the impurities introduced at this early stage propagate through subsequent coupling steps, complicating the final isolation of the drug substance and necessitating aggressive purification techniques such as column chromatography, which are economically unfeasible for large-scale commercial production.
The Novel Approach
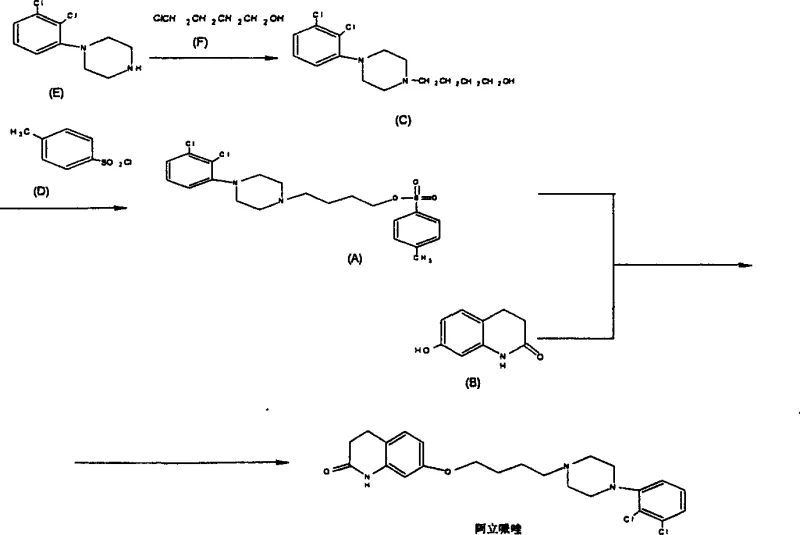
In stark contrast, the methodology disclosed in CN1289484C circumvents these hydrolytic pitfalls by employing a modular three-step sequence centered around a stable sulfonate ester intermediate. The process initiates with the nucleophilic substitution of 1-(2,3-dichlorophenyl)piperazine with 4-chloro-1-butanol, a reagent chosen for its superior specificity and reduced tendency toward side reactions compared to its dibromo counterpart. The resulting hydroxy-butyl piperazine is then activated via sulfonylation using reagents such as p-toluenesulfonyl chloride, creating a highly reactive sulfonate ester that serves as an excellent electrophile for the final coupling. This strategic activation allows the final condensation with 7-hydroxy-3,4-dihydroquinolinone to proceed under controlled basic conditions with minimal byproduct formation. The result is a synthetic route that delivers crude product purity exceeding 97%, enabling the attainment of pharmaceutical grade material through simple ethanol recrystallization rather than costly chromatographic separation.
Mechanistic Insights into Nucleophilic Substitution and Sulfonylation
The core chemical innovation lies in the manipulation of leaving group ability and nucleophilicity to favor the desired ether bond formation while suppressing hydrolysis. In the initial alkylation step, the secondary amine of the dichlorophenyl piperazine acts as a potent nucleophile attacking the terminal carbon of the chloro-butanol. The use of polar aprotic solvents like acetonitrile or DMF enhances the nucleophilicity of the amine while stabilizing the transition state, facilitating the displacement of the chloride ion. Crucially, the reaction temperature is maintained within a moderate range (typically 80-140°C), which is sufficient to drive the kinetics without inducing thermal degradation or elimination reactions that could compromise the integrity of the butyl chain. The subsequent sulfonylation step converts the primary alcohol of the intermediate into a sulfonate ester, effectively transforming a poor leaving group (hydroxyl) into an exceptional one (sulfonate). This activation is pivotal because it lowers the activation energy for the final nucleophilic attack by the phenolic oxygen of the quinolinone moiety, allowing the reaction to proceed efficiently even with sterically hindered substrates.
From an impurity control perspective, this mechanism offers distinct advantages over direct alkylation strategies. By isolating the sulfonate intermediate, manufacturers can implement a 'purge point' in the process where non-polar impurities and unreacted starting materials are removed before the final coupling. The sulfonate ester is generally more stable and easier to crystallize than the corresponding alkyl halides, providing a physical means of purification that relies on solubility differences rather than complex chromatography. Furthermore, the final condensation step utilizes the sulfonate's high reactivity to ensure complete consumption of the valuable quinolinone precursor, minimizing the presence of unreacted phenolic impurities in the final API. This mechanistic precision ensures that the impurity profile remains well-defined and manageable, a critical factor for regulatory compliance and consistent batch-to-batch quality in pharmaceutical manufacturing.
How to Synthesize Aripiprazole Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and solvent selection to maximize the benefits of the sulfonate strategy. The process is designed to be operationally simple, utilizing common laboratory and plant equipment without the need for specialized high-pressure reactors or cryogenic conditions. Operators should focus on maintaining anhydrous conditions during the sulfonylation step to prevent premature hydrolysis of the activating agent, while the final coupling can tolerate a broader range of solvent systems including acetonitrile and DMF. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational protocol for process development teams looking to adopt this technology.
- Condense 1-(2,3-dichlorophenyl)piperazine with 4-halo-1-butanol in the presence of an alkali to form the butanol intermediate.
- React the resulting butanol intermediate with an aryl sulfonyl halide to generate the stable sulfonate ester.
- Perform a final condensation between the sulfonate ester and 7-hydroxy-3,4-dihydroquinolinone under basic conditions to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into enhanced operational resilience and cost predictability. By eliminating the reliance on 1,4-dibromobutane, a reagent known for its handling difficulties and side-reaction profile, the process reduces the risk of batch failures and off-spec material that can disrupt production schedules. The ability to achieve high purity through simple recrystallization rather than column chromatography represents a massive reduction in processing time and solvent consumption, which are major cost drivers in fine chemical manufacturing. This efficiency gain allows for a more streamlined workflow where throughput is limited by reaction kinetics rather than purification bottlenecks, ultimately supporting a more reliable supply of high-purity pharmaceutical intermediates to downstream formulation partners.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial simplification of the purification train. Traditional methods often require multiple recrystallization cycles or preparative chromatography to remove hydrolysis byproducts, which consumes vast quantities of solvents and silica gel while sacrificing yield. By contrast, the sulfonate-mediated route produces a crude product of such high quality that a single recrystallization step is sufficient to meet pharmacopeial standards. This drastic reduction in downstream processing steps lowers utility costs, waste disposal fees, and labor hours, resulting in significant cost reduction in pharmaceutical manufacturing without compromising on the stringent quality requirements of the final drug substance.
- Enhanced Supply Chain Reliability: The raw materials utilized in this novel pathway, specifically 4-chloro-1-butanol and aryl sulfonyl chlorides, are commodity chemicals with robust global supply chains, unlike specialized reagents that may face sourcing volatility. The mild reaction conditions (often below 150°C) reduce the energy intensity of the process and minimize wear on reactor vessels, contributing to higher equipment availability and lower maintenance downtime. Furthermore, the improved yield stability and reduced sensitivity to moisture variations mean that production batches are more consistent, allowing supply chain planners to forecast output with greater accuracy and maintain safer inventory levels of critical antipsychotic intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the absence of extreme temperatures or pressures, which simplifies heat transfer management in large reactors. The reduction in solvent usage and the elimination of silica-based chromatography significantly decrease the volume of hazardous waste generated per kilogram of product, aligning with modern green chemistry principles and environmental regulations. This environmental efficiency not only reduces the carbon footprint of the manufacturing site but also mitigates regulatory risks associated with waste disposal, making the commercial scale-up of complex pharmaceutical intermediates more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Aripiprazole synthesis technology. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of purity, yield, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this new process improve impurity profiles compared to the EP367141 method?
A: The conventional method utilizes 1,4-dibromobutane which is prone to hydrolysis under alkaline reflux conditions, generating difficult-to-remove impurities. The novel route employs a stable sulfonate intermediate formed under milder conditions, significantly reducing side reactions and eliminating the need for complex column chromatography.
Q: What are the scalability advantages of using 4-chloro-1-butanol over 1,4-dibromobutane?
A: 4-chloro-1-butanol offers higher reaction specificity and reduced hydrolysis risks compared to the dibromo analog. This stability allows for easier quality control of intermediates and simplifies the workup procedure, making the process more robust for multi-kilogram commercial production.
Q: Can the final product meet pharmaceutical grade standards without extensive purification?
A: Yes, the crude product obtained via this synthetic route typically exhibits purity exceeding 97% as detected by HPLC. A single recrystallization step using 95% ethanol is sufficient to remove organic impurities and achieve pharmaceutical grade specifications, avoiding the yield losses associated with multiple purification cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Aripiprazole intermediate meets the highest global standards, providing our clients with the confidence needed to navigate complex regulatory filings and market demands efficiently.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this sulfonate-based route for your operations. We encourage you to contact us today to discuss your specific requirements, obtain specific COA data from our recent pilot runs, and review detailed route feasibility assessments tailored to your production capacity and quality goals.
