Scalable Nickel-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Amines
The pharmaceutical and agrochemical industries are constantly seeking robust, cost-effective methodologies for the synthesis of chiral amines, which serve as critical building blocks for bioactive molecules. Patent CN112824374B introduces a groundbreaking approach utilizing nickel-catalyzed asymmetric hydrogenation of imines, marking a significant departure from traditional noble metal-dependent processes. This technology leverages a novel class of chiral ferrocene-imidazole bisphosphine ligands to generate highly active nickel catalysts in situ. By shifting the paradigm from scarce precious metals like rhodium and iridium to earth-abundant nickel, this invention addresses critical supply chain vulnerabilities while maintaining exceptional stereocontrol. The method operates under mild conditions in alcoholic solvents, offering a green and scalable pathway for producing high-purity chiral amine compounds essential for modern drug discovery and agricultural chemical manufacturing.
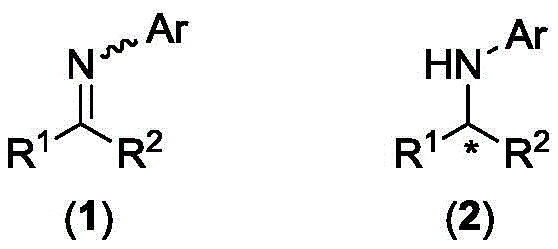
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of imines has been dominated by catalysts based on precious transition metals such as rhodium (Rh), ruthenium (Ru), iridium (Ir), and palladium (Pd). While these systems often deliver high enantioselectivity, they suffer from severe economic and environmental drawbacks that hinder their widespread industrial adoption. The global reserves of these heavy metals are rapidly declining, leading to volatile pricing and supply insecurity for large-scale manufacturers. Furthermore, the toxicity of residual heavy metal ions poses a significant regulatory hurdle in pharmaceutical production, necessitating complex and costly downstream purification steps to meet stringent purity specifications. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, making conventional noble metal catalysis less attractive for commodity-level chemical production where margin compression is a constant threat.
The Novel Approach
The methodology disclosed in CN112824374B overcomes these barriers by employing nickel, a base metal that is abundant, inexpensive, and environmentally benign. The core innovation lies in the specific design of the chiral ligand system, which compensates for the historically lower activity and selectivity of nickel compared to its noble counterparts. By utilizing a chiral ferrocene-imidazole bisphosphine ligand, the system achieves a precise coordination environment around the nickel center, enabling efficient activation of molecular hydrogen and effective facial differentiation of the imine substrate. This approach not only drastically reduces the raw material cost associated with the catalyst but also simplifies the operational workflow. The ability to generate the active catalyst species in situ within alcohol proton solvents eliminates the need for pre-isolating sensitive metal complexes, thereby enhancing process safety and operational simplicity for plant operators.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Hydrogenation
The success of this transformation hinges on the unique electronic and steric properties of the chiral ferrocene-imidazole bisphosphine ligand. The ferrocene backbone provides a rigid, chiral scaffold that dictates the spatial arrangement of the phosphine donors, while the imidazole moiety offers additional coordination potential that stabilizes the nickel center during the catalytic cycle. As illustrated in the ligand structure below, the specific substitution patterns on the phosphine atoms (R4, R5) and the chiral center (R3) can be finely tuned to optimize performance for different substrate classes. This modularity allows chemists to tailor the catalyst for specific steric demands, ensuring high enantiomeric excess (ee) across a broad range of imine derivatives.
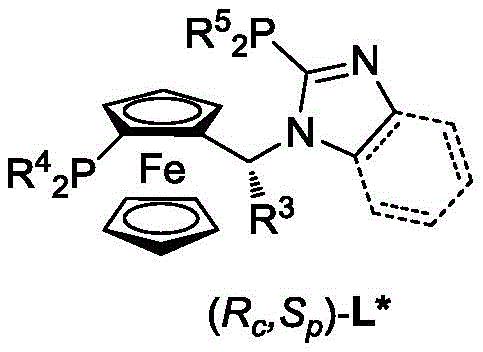
From a mechanistic standpoint, the reaction proceeds through a concerted hydrogenation pathway where the nickel-hydride species transfers hydrogen to the C=N bond of the imine. The choice of solvent plays a pivotal role in this mechanism; protic solvents like trifluoroethanol are preferred as they facilitate proton transfer steps and stabilize charged intermediates. The patent data indicates that the system tolerates a wide variety of functional groups on the imine substrate, including electron-donating and electron-withdrawing substituents on the aromatic rings. This robustness suggests that the catalytic cycle is resilient against potential poisoning by heteroatoms, a common failure mode for base metal catalysts. Consequently, the impurity profile of the resulting chiral amine is clean, with minimal formation of over-reduced byproducts or racemic material, simplifying the subsequent crystallization or chromatographic purification steps required for API intermediate production.
How to Synthesize Chiral Amine Compounds Efficiently
The synthesis protocol outlined in the patent is designed for practical implementation in both laboratory and pilot plant settings. The process begins with the preparation of the chiral ligand, followed by the in situ formation of the nickel catalyst and the subsequent hydrogenation reaction. The detailed procedure emphasizes the importance of inert atmosphere techniques during catalyst preparation to prevent oxidation of the low-valent nickel species. For a comprehensive understanding of the operational parameters, including specific molar ratios, temperature gradients, and workup procedures, please refer to the standardized synthesis guide provided below.
- Prepare the chiral nickel catalyst by stirring a nickel salt (e.g., Ni(OAc)2·4H2O) with a chiral ferrocene-imidazole bisphosphine ligand in an alcoholic solvent under nitrogen protection for 2 hours.
- Dissolve the imine substrate in the reaction medium and add it to the prepared catalyst solution in a high-pressure reactor.
- Purge the reactor with hydrogen three times, pressurize to 20-100 bar, and react at 20-100°C for 1-24 hours, followed by workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed technology represents a strategic opportunity to de-risk the supply of critical chiral intermediates. The shift from precious metals to base metals fundamentally alters the cost structure of the manufacturing process, removing exposure to the volatile commodities market associated with rhodium and iridium. Furthermore, the simplified ligand synthesis and the use of common alcohol solvents reduce the dependency on specialized reagents, enhancing the overall resilience of the supply chain against geopolitical disruptions or raw material shortages.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of expensive noble metal precursors with inexpensive nickel salts. Since the catalyst loading can be kept low while maintaining high turnover numbers, the direct material cost for the catalytic system is significantly reduced. Additionally, the elimination of complex ligand synthesis steps and the use of readily available solvents contribute to substantial cost savings in the overall process. This economic efficiency allows for more competitive pricing of the final chiral amine products, improving margins for downstream pharmaceutical formulations.
- Enhanced Supply Chain Reliability: Nickel is one of the most abundant transition metals in the earth's crust, ensuring a stable and long-term supply availability compared to the geographically concentrated reserves of platinum group metals. This abundance translates to reliable lead times and consistent pricing, allowing procurement teams to forecast budgets with greater accuracy. Moreover, the robustness of the catalyst system means that production schedules are less likely to be disrupted by catalyst deactivation or sensitivity to feedstock variations, ensuring continuous operation capabilities.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by its successful application in the synthesis of the herbicide (S)-metolachlor key intermediate. The use of alcohol solvents and the absence of toxic heavy metals simplify waste stream management and reduce the environmental footprint of the manufacturing facility. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with hazardous waste disposal, making it an ideal candidate for sustainable large-scale production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed asymmetric hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: How does this nickel-catalyzed method compare to traditional rhodium or iridium catalysis regarding metal residues?
A: Unlike traditional methods using expensive noble metals like Rh or Ir which leave toxic residues difficult to remove, this nickel-based system utilizes earth-abundant metals. While nickel removal is still required, the significantly lower cost of the metal precursor allows for more aggressive purification strategies without impacting overall process economics, and the ligand design promotes high turnover, reducing the absolute metal load needed.
Q: Is the chiral ferrocene-imidazole ligand suitable for large-scale manufacturing?
A: Yes, the patent explicitly highlights the simplicity of the ligand preparation and its suitability for continuous operation. The synthesis involves standard organic transformations such as nucleophilic substitution and lithiation-phosphination, which are well-established in industrial settings, ensuring a reliable supply chain for the catalyst system.
Q: What is the substrate scope for this asymmetric hydrogenation process?
A: The method demonstrates broad applicability across various imine substrates, including those with diverse aryl groups (phenyl, substituted phenyls) and alkyl chains. It has been successfully applied to the synthesis of key intermediates for agrochemicals like (S)-metolachlor, achieving high yields and enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of base metal catalysis in modern organic synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112824374B are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral amine intermediate delivered meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to evaluate the feasibility of this nickel-catalyzed route for your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this base metal system. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-purity chiral amines for your pipeline.
