Advanced Enantioselective Hydrogenation for Scalable Chiral Amine Manufacturing
Advanced Enantioselective Hydrogenation for Scalable Chiral Amine Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access chiral building blocks, which are critical for the synthesis of active pharmaceutical ingredients (APIs). Patent CN102076634A introduces a significant technological breakthrough in the field of asymmetric synthesis by detailing a robust method for the enantioselective hydrogenation of isomeric N-H imines. Unlike traditional approaches that necessitate cumbersome protection and deprotection sequences for the amine functionality, this invention utilizes transition metal-based catalysts modified with chiral phosphine derivatives to directly reduce N-unsubstituted imines. This capability represents a paradigm shift for reliable pharmaceutical intermediate suppliers, as it streamlines the production of enantiomerically enriched chiral amines, reducing both material costs and processing time while maintaining high stereochemical integrity.
The core innovation lies in the stability and reactivity of the N-H imine substrates. Historically, the presence of an N-H bond in imines has posed significant challenges due to potential tautomerization or catalyst poisoning. However, the patent elucidates that these N-H ketoimines can be isolated and handled as stable hydrochloride salts. This stability allows for a direct reduction pathway that bypasses the need for N-protecting groups, thereby simplifying the synthetic route significantly. For R&D directors focusing on process chemistry, this means a reduction in the number of unit operations and a cleaner impurity profile, which is essential for meeting the stringent regulatory requirements of modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing chiral amines often rely on the reductive amination of ketones or the hydrogenation of protected imines. A major drawback of these conventional methods is the requirement for additional synthetic steps to install and subsequently remove protecting groups on the nitrogen atom. These extra steps not only increase the overall cost of goods sold (COGS) but also generate significant amounts of chemical waste, negatively impacting the environmental footprint of the manufacturing process. Furthermore, the use of protecting groups can introduce new chiral centers or complicate the purification process, leading to lower overall yields and reduced throughput. In a competitive market where cost reduction in API manufacturing is paramount, these inefficiencies create substantial bottlenecks that hinder rapid scale-up and commercial viability.
The Novel Approach
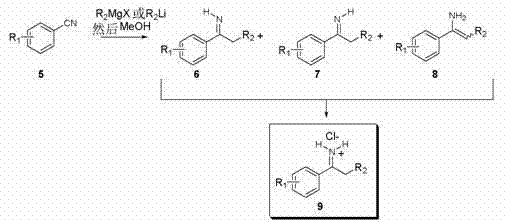
The novel approach described in the patent overcomes these limitations by leveraging the unique reactivity of N-H imine hydrochloride salts. By preparing the NH-imine through the addition of suitable organometallic reagents to nitriles, followed by quenching and salt formation, the process generates a stable substrate ready for direct hydrogenation. This strategy eliminates the protection/deprotection cycle entirely, resulting in a more atom-economical process. The ability to perform enantioselective hydrogenation directly on the N-H imine salt using transition metal catalysts allows for a telescoped process that is both faster and cleaner. This methodology aligns perfectly with the goals of green chemistry and offers a compelling value proposition for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Transition Metal-Catalyzed Asymmetric Hydrogenation
The success of this synthetic route hinges on the precise selection of the transition metal catalyst and the chiral ligand environment. The patent highlights the efficacy of Iridium, Rhodium, and Ruthenium catalysts modified with chiral phosphine derivatives. Specifically, the use of Iridium precursors such as [Ir(cod)2Cl]2 in combination with ligands like (R,S)-PFP-P(tBu)2 has demonstrated superior enantioselectivity. The mechanism likely involves the coordination of the imine substrate to the metal center, followed by the migratory insertion of hydrogen and subsequent reductive elimination to release the chiral amine. The chiral ligand creates a sterically defined pocket that discriminates between the prochiral faces of the imine, ensuring the formation of the desired enantiomer with high fidelity.
Impurity control is another critical aspect addressed by this mechanistic understanding. The choice of solvent plays a pivotal role in stabilizing the catalytic species and the substrate. Solvents such as 1,2-dichloroethane (DCE) and 2,2,2-trifluoroethanol (TFE) are identified as particularly effective media. The patent data indicates that reaction conditions, including hydrogen pressure (150-500 psi) and temperature (25-40°C), can be tuned to optimize both conversion and enantiomeric excess (ee). For instance, Example 1 achieved a 76.9% ee with 71% conversion using an Iridium system in DCE or TFE. Understanding these parameters allows process chemists to minimize side reactions such as over-reduction or racemization, ensuring the production of high-purity pharmaceutical intermediates that meet strict quality specifications.

How to Synthesize 1-(3-bromophenyl)-1-propanamine Efficiently
The synthesis of specific chiral amines, such as 1-(3-bromophenyl)-1-propanamine, serves as a prime example of the utility of this patented technology. The process begins with the preparation of the corresponding NH-imine hydrochloride salt, which is then subjected to asymmetric hydrogenation. The operational simplicity of mixing the catalyst, ligand, and substrate in a suitable solvent under hydrogen pressure makes this route highly attractive for both laboratory and pilot plant settings. The detailed standardized synthesis steps provided in the patent offer a clear roadmap for replicating these results, ensuring consistency and reliability in production. For further technical details on the specific stoichiometry and workup procedures, please refer to the comprehensive guide below.
- Prepare the N-H imine substrate by reacting a nitrile with an organometallic reagent, followed by quenching and salt formation with anhydrous HCl to ensure stability.
- Mix the NH-imine hydrochloride salt with a chiral transition metal catalyst system (e.g., Iridium or Rhodium based) in a suitable solvent like 1,2-DCE or TFE.
- Conduct the enantioselective hydrogenation under H2 pressure (150-500 psi) at mild temperatures (25-40°C) to yield the target chiral amine with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this direct hydrogenation technology offers transformative benefits. The elimination of protection and deprotection steps translates directly into a reduction in raw material consumption and waste disposal costs. By shortening the synthetic sequence, manufacturers can achieve faster turnaround times and improve overall equipment effectiveness (OEE). This efficiency gain is crucial for maintaining a resilient supply chain capable of responding to fluctuating market demands. Furthermore, the use of stable intermediate salts enhances storage and handling safety, reducing the risks associated with hazardous reagents typically used in protection chemistry.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the removal of auxiliary chemical steps. Without the need for protecting group reagents and the solvents required for their removal, the variable cost per kilogram of the final product is substantially lowered. Additionally, the high atom economy of the direct hydrogenation process minimizes waste generation, leading to reduced environmental compliance costs. The ability to operate at mild temperatures (25-40°C) also reduces energy consumption compared to processes requiring cryogenic conditions or high-temperature reflux, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the N-H imine salt intermediates ensures a stable supply of key starting materials. Unlike sensitive free-base imines that may degrade upon storage, the hydrochloride salts are free-flowing solids that can be stockpiled safely. This stability mitigates the risk of supply disruptions caused by material degradation. Moreover, the versatility of the catalyst system, which accommodates various substrates with different electronic and steric properties, allows for a flexible production schedule. This adaptability is essential for reducing lead time for high-purity pharmaceutical intermediates in a multi-product facility.
- Scalability and Environmental Compliance: The process conditions described, specifically the hydrogen pressure range of 150-500 psi, are well within the capabilities of standard industrial hydrogenation reactors. This compatibility facilitates seamless technology transfer from the laboratory to commercial-scale production without the need for specialized high-pressure equipment. The reduction in chemical waste streams, particularly those associated with protecting group removal, simplifies effluent treatment and aligns with increasingly stringent global environmental regulations. This sustainability profile enhances the long-term viability of the supply chain and supports corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in patent CN102076634A, providing a factual basis for decision-making. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of using N-H imines over protected imines in this synthesis?
A: The primary advantage is the elimination of protection and deprotection steps. Traditional methods require protecting the amine group to prevent side reactions, which adds cost and waste. This patent demonstrates that N-H imines can be stabilized as hydrochloride salts, allowing for direct asymmetric hydrogenation.
Q: Which catalyst systems provide the best enantioselectivity for this reaction?
A: According to the patent data, Iridium-based catalysts modified with chiral phosphine ligands like (R,S)-PFP-P(tBu)2 show superior performance. For instance, Example 1 achieved 76.9% ee using an Iridium precursor, whereas Rhodium and Ruthenium systems showed lower enantioselectivity (43.1% and 38.6% ee respectively) under similar conditions.
Q: What are the typical reaction conditions required for scale-up?
A: The process operates under relatively mild conditions suitable for industrial scale-up. The hydrogenation is performed at pressures between 150-500 psi and temperatures ranging from 25°C to 40°C. Solvents such as 1,2-dichloroethane (DCE) or 2,2,2-trifluoroethanol (TFE) are effective media for these transformations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
The technological advancements detailed in this patent underscore the immense potential of direct asymmetric hydrogenation for the efficient production of chiral amines. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative methodologies for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory routes are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your strategic goals in developing next-generation therapeutics.
