Revolutionizing Alkenyl Boronate Production with Earth-Abundant Cobalt Catalysts for Global Supply Chains
The chemical synthesis landscape is undergoing a paradigm shift towards sustainability and cost-efficiency, driven by the urgent need for scalable manufacturing of complex intermediates. Patent CN111943967B introduces a groundbreaking methodology for the synthesis of alkenyl boronate compounds, utilizing an earth-abundant cobalt metal catalyst to facilitate efficient olefin dehydroboration. This technology represents a significant departure from traditional noble-metal-dependent processes, offering a robust pathway to generate high-value vinyl boronates from stable and inexpensive olefin feedstocks. The core innovation lies in the ability to achieve exclusive E-stereoselectivity while producing hydrogen gas as the sole by-product, ensuring exceptional atom economy. For R&D directors and process chemists, this patent provides a critical blueprint for accessing versatile synthetic building blocks that are essential for Suzuki-Miyaura cross-coupling reactions and other downstream transformations widely used in API production.
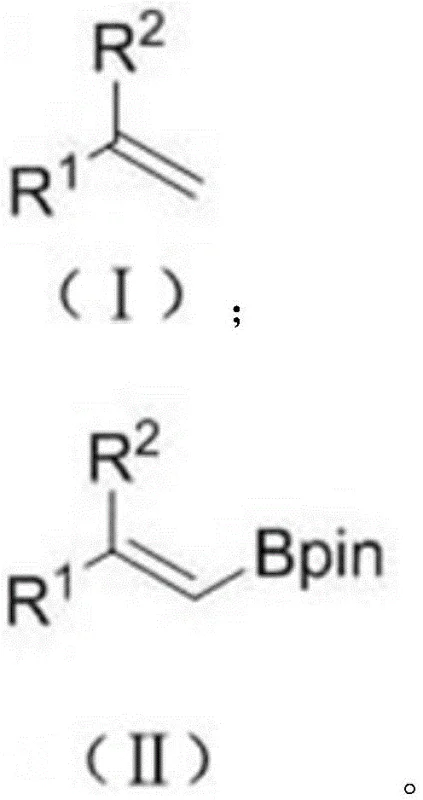
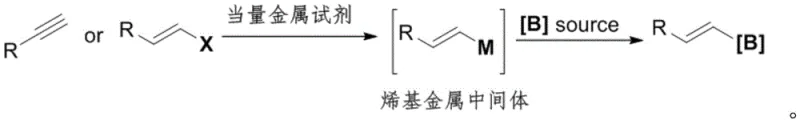
The limitations of conventional methods for synthesizing alkenyl boron compounds have long posed challenges for industrial scalability and cost management. Historically, the standard approach involved the use of alkynes or alkenyl halides as starting materials, requiring stoichiometric amounts of metal reagents to form alkenyl metal intermediates prior to borylation. As illustrated in the traditional reaction pathways, these methods often suffer from poor atom economy, the generation of significant metal waste, and the high cost associated with alkyne precursors. Furthermore, alternative transition-metal catalyzed hydroboration of alkynes, while efficient, relies on alkynes that are notoriously difficult to prepare and expensive to source commercially. These economic and logistical bottlenecks necessitate a more economical synthesis method that leverages readily available feedstocks.
The novel approach detailed in this patent overcomes these hurdles by employing a cobalt-catalyzed dehydrogenative borylation of simple olefins. Instead of relying on costly alkynes, this method utilizes stable and cheap olefins, such as styrenes, which are abundantly available in the global chemical supply chain. The reaction proceeds under mild thermal conditions (60-70°C) in common organic solvents like THF, utilizing a specialized ligand system to activate the cobalt center. This shift from alkyne-based to olefin-based feedstocks drastically reduces raw material costs and simplifies the supply chain logistics. Moreover, the reaction produces hydrogen gas as the only by-product, which aligns perfectly with green chemistry principles by minimizing waste disposal burdens and maximizing atom utilization compared to halide-based displacement reactions.
Mechanistic Insights into Cobalt-Catalyzed Dehydrogenative Borylation
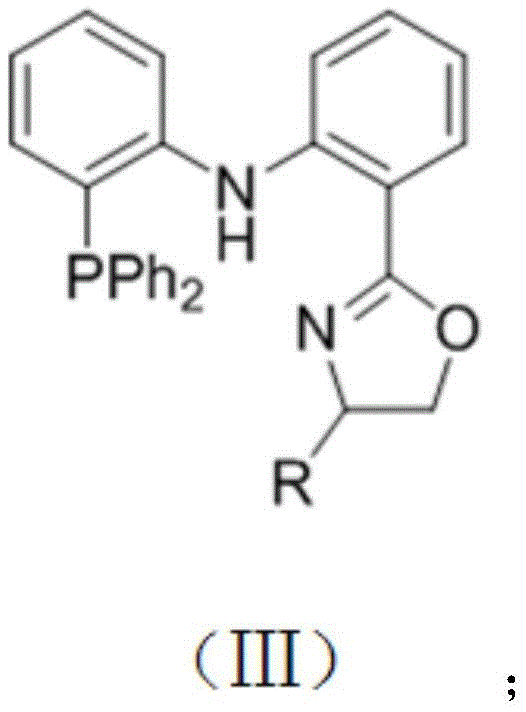
The success of this transformation hinges on the precise design of the ligand environment surrounding the cobalt center, which dictates both activity and stereoselectivity. The patent specifies the use of tridentate PNN or PNP type ligands, particularly those featuring an oxazoline or similar heterocyclic backbone, which stabilize the active cobalt species and facilitate the crucial C-H activation step. The mechanistic cycle likely involves the oxidative addition of the diboron reagent to the cobalt center, followed by coordination of the olefin and subsequent migratory insertion. The unique electronic properties of the ligand, such as the PNBnN variant, promote the formation of the thermodynamically more stable E-alkenyl cobalt intermediate, thereby ensuring exclusive E-selectivity in the final product. This level of control is critical for pharmaceutical applications where isomeric purity directly impacts biological activity and regulatory approval.
Impurity control in this catalytic system is managed through the careful selection of the base and the optimization of reaction stoichiometry. The presence of a base, such as potassium carbonate (K2CO3) or alkoxides, is essential for regenerating the active catalyst and driving the equilibrium towards product formation by neutralizing acidic by-products. Experimental data indicates that increasing the base loading can significantly improve yields, pushing conversions up to 99% in optimized scenarios. Furthermore, the choice of base influences the reaction kinetics; while strong alkoxides like KOtBu are effective, milder carbonates offer a balance of reactivity and operational safety that is preferable for large-scale manufacturing. The robustness of the catalyst system against various functional groups ensures that side reactions, such as homocoupling or over-reduction, are minimized, resulting in a clean crude profile that simplifies downstream purification.
How to Synthesize Alkenyl Boronate Efficiently
To implement this synthesis effectively, operators must adhere to strict inert atmosphere protocols to prevent catalyst deactivation by oxygen or moisture. The process begins with the in-situ generation of the active cobalt-ligand complex, followed by the sequential addition of substrates and base. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the catalytic system by mixing Cobalt(II) chloride (CoCl2) with a specialized PNN or PNP ligand additive in anhydrous THF under inert atmosphere.
- Add the olefin substrate, bis(pinacolato)diboron (B2pin2), and a carbonate or alkoxide base to the mixture, then heat to 60-70°C for 3-12 hours.
- Upon completion, remove the solvent under reduced pressure and purify the crude product via flash column chromatography to isolate the pure E-alkenyl boronate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology offers transformative benefits in terms of cost structure and supply reliability. By shifting away from precious metal catalysts like palladium or rhodium, manufacturers can achieve substantial cost savings on catalyst procurement and eliminate the complex, expensive processes required for removing trace heavy metals from the final API. The reliance on commodity chemicals like styrenes and bis(pinacolato)diboron ensures a stable and continuous supply of raw materials, reducing the risk of production delays caused by niche precursor shortages. Additionally, the high atom economy and minimal waste generation translate to lower environmental compliance costs and simplified waste treatment protocols, further enhancing the overall economic viability of the process.
- Cost Reduction in Manufacturing: The replacement of expensive noble metals with inexpensive cobalt salts drastically lowers the direct material cost of the catalytic system. Since the catalyst loading can be kept low (as low as 0.1 mol% in scaled examples) while maintaining high efficiency, the overall cost per kilogram of product is significantly reduced. This economic advantage is compounded by the use of cheap olefin starting materials instead of premium-priced alkynes, creating a dual-layer cost optimization strategy that improves profit margins without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing stable olefins and common boron reagents is far less risky than procuring specialized alkynes or sensitive organometallic reagents. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the reagents have long shelf lives and do not require cryogenic storage. This stability ensures that production lines can run continuously with minimal interruption, securing the supply of critical intermediates for downstream drug synthesis and mitigating the risks associated with volatile global chemical markets.
- Scalability and Environmental Compliance: The generation of hydrogen gas as the sole by-product simplifies the workup procedure, as there are no stoichiometric salt wastes or halogenated by-products to dispose of. This clean reaction profile facilitates easier scale-up from gram to tonnage quantities, as heat management and waste treatment become less burdensome. The process aligns with increasingly stringent environmental regulations regarding heavy metal discharge and solvent waste, positioning manufacturers as leaders in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed borylation technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential adopters.
Q: Why is cobalt preferred over palladium or rhodium for this synthesis?
A: Cobalt is an earth-abundant base metal that offers a significant cost advantage over precious metals like palladium or rhodium. Furthermore, the specific ligand system described in CN111943967B enables high activity and exclusive E-selectivity without requiring expensive noble metal catalysts.
Q: What is the stereochemical outcome of this dehydrogenative borylation?
A: The process exclusively yields the E-configuration (trans) alkenyl boronate products. This high stereoselectivity eliminates the need for difficult separation of Z/E isomers, thereby simplifying downstream purification and improving overall process efficiency.
Q: Can this method tolerate diverse functional groups on the styrene substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance. It successfully processes substrates containing electron-donating groups (methoxy, alkyl), electron-withdrawing groups (halogens, trifluoromethyl, esters), and heterocycles (pyridine), making it highly versatile for complex intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Boronate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this cobalt-catalyzed technology in modernizing the production of pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkenyl boronate meets the exacting standards required for GMP manufacturing. Our commitment to technological excellence allows us to offer high-purity intermediates that empower our clients to accelerate their drug development pipelines with confidence.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this technology can optimize your supply chain and reduce your overall manufacturing costs.
