Revolutionizing Alkenyl Boronate Production with Efficient Cobalt-Catalyzed Dehydroboration
Revolutionizing Alkenyl Boronate Production with Efficient Cobalt-Catalyzed Dehydroboration
In the rapidly evolving landscape of fine chemical synthesis, the demand for efficient, cost-effective routes to versatile building blocks like alkenyl boronates has never been higher. Patent CN111943967A introduces a groundbreaking methodology that addresses long-standing economic and technical bottlenecks in the production of these critical intermediates. By shifting the paradigm from expensive alkyne precursors to readily available olefins, this technology leverages a robust cobalt-catalyzed dehydroboration system. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize supply chains for pharmaceutical intermediates and agrochemical precursors. The process not only simplifies the synthetic route but also ensures high atom economy, generating hydrogen gas as the sole by-product, thereby aligning with modern green chemistry principles while delivering high-purity outputs essential for complex drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkenyl boron compounds has relied heavily on two primary pathways, both of which suffer from significant economic and operational drawbacks. The first conventional approach involves the use of alkynes or alkenyl halides as starting materials, which are transformed into alkenyl metal intermediates via carbon-metallation or hydrogen-metallation using stoichiometric amounts of metal reagents before reacting with boron sources. As illustrated in the reaction scheme below, this multi-step process often requires harsh conditions and generates substantial metal waste.
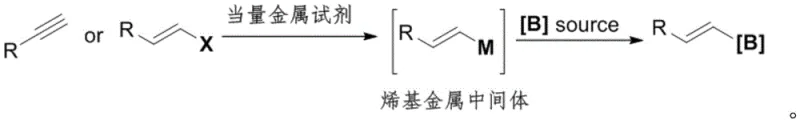
Furthermore, a more recent development involving transition metal-catalyzed hydroboration of alkynes, while efficient in terms of steps, is severely limited by the raw material itself. Alkynes are notoriously difficult to prepare, unstable, and significantly more expensive than their olefin counterparts. The reliance on these costly precursors, combined with the frequent need for precious metal catalysts like Palladium or Rhodium, creates a high barrier to entry for large-scale manufacturing. This dependency inflates the cost of goods sold (COGS) and introduces supply chain vulnerabilities, making it an less ideal choice for the mass production of high-purity OLED materials or bulk pharmaceutical ingredients.
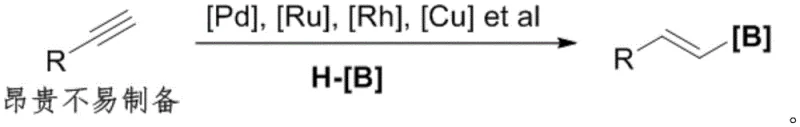
The Novel Approach
The technology disclosed in patent CN111943967A fundamentally disrupts this status quo by utilizing stable, cheap, and easily obtained olefins as the starting feedstock. Instead of adding across a triple bond, this novel method performs a dehydroboration reaction on a double bond. The core transformation, depicted below, shows the direct conversion of a styrene derivative into the corresponding alkenyl boronate using a diboron reagent. This shift eliminates the need for expensive alkyne synthesis entirely.
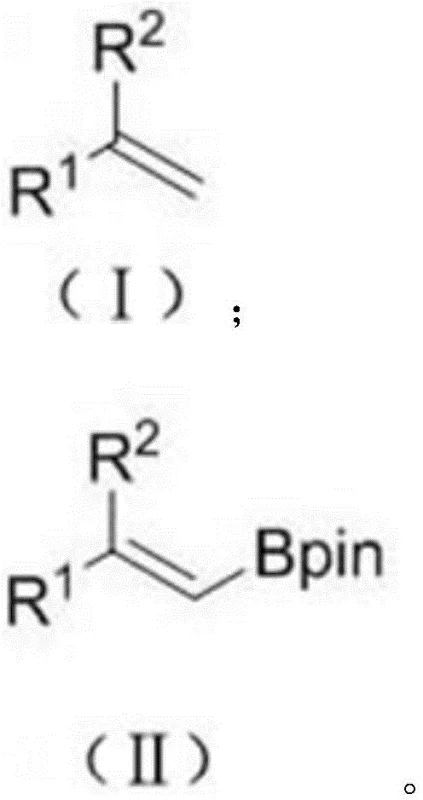
By employing a cheap cobalt metal catalyst, this approach achieves high efficiency without the financial burden of noble metals. The reaction is characterized by its exceptional atom economy, as the only by-product generated is hydrogen gas, which easily vents from the system, driving the equilibrium forward and simplifying purification. This results in a single E-configuration product with high selectivity, drastically reducing the need for difficult isomer separations that typically plague conventional methods. For a reliable agrochemical intermediate supplier, this translates to a streamlined process that is both economically superior and environmentally friendlier.
Mechanistic Insights into Cobalt-Catalyzed Dehydroboration
The success of this synthesis lies in the precise tuning of the catalytic system, specifically the interplay between the cobalt center and the specialized ligand environment. The patent details the use of specific ligand additives, such as PNN or PNP type ligands, which are crucial for stabilizing the active cobalt species and directing the stereoselectivity of the reaction. As shown in the structural diagrams, these ligands feature rigid backbones and specific donor atoms that facilitate the oxidative addition and reductive elimination steps necessary for the catalytic cycle.
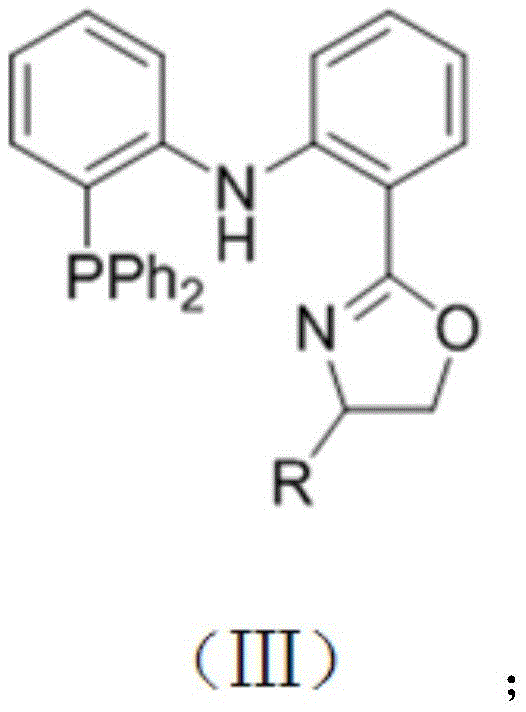
From a mechanistic standpoint, the cobalt catalyst activates the B-B bond of the diboron reagent, forming a reactive cobalt-boryl species. This species then coordinates with the olefin substrate, followed by migratory insertion and beta-hydride elimination to release the final alkenyl boronate product and regenerate the catalyst. The rigorous control over the ligand sphere ensures that the reaction proceeds exclusively through the pathway yielding the E-isomer. For R&D teams focused on cost reduction in electronic chemical manufacturing, understanding this mechanism is vital, as it allows for the rational design of substrates and the prediction of compatibility with various functional groups, ensuring robust process performance across diverse chemical spaces.
How to Synthesize Alkenyl Boronate Efficiently
The experimental protocols outlined in the patent provide a clear roadmap for implementing this technology in a laboratory or pilot plant setting. The procedure typically involves preparing the catalyst system in an inert atmosphere, followed by the sequential addition of substrates and base. The reaction is conducted at moderate temperatures, generally around 65°C, which balances reaction rate with energy efficiency. Detailed standard operating procedures for scaling this reaction from milligram to kilogram quantities are provided in the technical documentation below.
- Prepare the reaction vessel under inert atmosphere (Argon/Nitrogen) and add Cobalt catalyst (e.g., CoCl2), specific ligand additives, and organic solvent.
- Introduce the olefin substrate and diboron reagent (B2pin2) along with a base such as K2CO3 or KOtBu to the mixture.
- Heat the reaction mixture to 60-70°C, stir until completion, then purify the resulting E-configuration alkenyl boronate via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed olefin dehydroboration offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio. By switching from alkynes to olefins, manufacturers can leverage a global supply chain of commodity chemicals that are produced in massive volumes, ensuring consistent availability and price stability. This shift effectively decouples production costs from the volatile markets associated with specialty alkyne synthesis.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts (like Pd, Rh, Ru) with earth-abundant cobalt represents a direct reduction in catalyst costs. Additionally, the elimination of stoichiometric metal reagents reduces waste disposal costs and raw material consumption. The high atom economy means less material is wasted as by-products, further enhancing the overall yield per dollar spent. Qualitative analysis suggests that removing the need for cryogenic conditions or ultra-dry solvents often required for organometallic intermediates also lowers utility and infrastructure overheads significantly.
- Enhanced Supply Chain Reliability: Olefins and diboron reagents are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the cobalt catalyst system against moisture and air (relative to sensitive organolithium or Grignard reagents used in traditional methods) simplifies logistics and storage requirements. This resilience ensures that production schedules are less likely to be disrupted by minor environmental fluctuations or shipping delays, providing a more reliable flow of commercial scale-up of complex polymer additives and intermediates.
- Scalability and Environmental Compliance: The generation of hydrogen gas as the only by-product simplifies waste stream management, avoiding the accumulation of heavy metal salts or halogenated waste typical of halide-based routes. This cleaner profile facilitates easier regulatory approval and reduces the burden on wastewater treatment facilities. The moderate temperature range (60-70°C) and the use of common solvents like THF make the process highly amenable to scale-up in standard stainless steel reactors, enabling rapid transition from lab bench to 100 MT/annual commercial production without extensive re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional alkyne hydroboration?
A: This method utilizes stable and inexpensive olefins instead of costly and difficult-to-prepare alkynes. Furthermore, it employs a cheap cobalt catalyst rather than expensive noble metals, significantly reducing raw material costs while maintaining high atom economy with hydrogen as the only by-product.
Q: Does this synthesis method offer high stereoselectivity for pharmaceutical applications?
A: Yes, the process described in the patent yields products with exclusive E-configuration (E/Z > 100/1). This high stereoselectivity is critical for pharmaceutical intermediates where specific geometric isomers are required for biological activity and downstream coupling reactions.
Q: Is this process scalable for industrial production of fine chemicals?
A: The protocol uses common solvents like THF and operates at moderate temperatures (60-70°C). The use of earth-abundant cobalt and simple workup procedures (solvent removal and chromatography) suggests strong potential for commercial scale-up compared to methods requiring cryogenic conditions or sensitive organometallic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Boronate Supplier
The technological advancements detailed in patent CN111943967A underscore the immense potential of cobalt-catalyzed dehydroboration for modern chemical manufacturing. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate these academic and patent innovations into robust, industrial-grade processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this method are fully realized in practical application. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkenyl boronate meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with us to evaluate the feasibility of this route for your specific product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments to help you optimize your supply chain and achieve superior cost efficiency in your manufacturing operations.
