Revolutionizing Alkenyl Boronate Production: A Deep Dive into Zirconium-Catalyzed Transfer Boronation
The landscape of organic synthesis is continually evolving, driven by the urgent need for safer, more efficient, and cost-effective methodologies, particularly in the realm of complex intermediate production. A significant breakthrough in this domain is documented in Chinese Patent CN110407863B, which discloses a novel method for synthesizing alkenyl boronate compounds through a transfer boronation reaction. This technology represents a paradigm shift from traditional hydroboration techniques, offering a robust pathway that utilizes substituted olefins and styrene boronic acid pinacol esters as key starting materials. By employing a zirconium-based catalyst system, specifically bis(cyclopentadienyl)zirconium dihydride, the process achieves high conversion rates under relatively mild thermal conditions ranging from 60°C to 150°C. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic nuances and commercial implications of this patent is critical for optimizing supply chains and reducing overall manufacturing expenditures in fine chemical production.
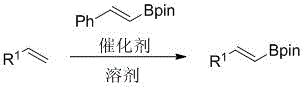
The limitations of conventional methods for constructing alkenyl boronate motifs have long plagued the fine chemical industry, primarily due to the reliance on hazardous reagents and苛刻 reaction conditions that complicate scale-up. Traditional hydroboration reactions typically require the use of diborane or alkyl boranes, which are extremely sensitive to moisture and oxygen, necessitating rigorous anhydrous conditions and specialized equipment that drive up capital and operational expenses. Furthermore, these classical approaches often suffer from poor chemical selectivity and functional group compatibility, leading to complex impurity profiles that require extensive and costly downstream purification processes to meet the stringent purity specifications demanded by the pharmaceutical sector. In contrast, the novel approach detailed in the patent leverages a transfer boronation strategy that bypasses the direct use of unstable borane species, instead utilizing stable styrene boronic acid pinacol esters as the boron source. This strategic substitution not only enhances the safety profile of the reaction by eliminating pyrophoric hazards but also simplifies the operational workflow, allowing for broader substrate scope and improved tolerance towards various functional groups present on the aromatic rings of the olefin substrates.
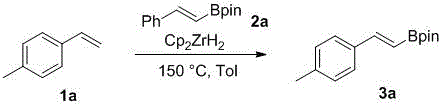
Mechanistic insights into the zirconium-catalyzed cyclization and transfer process reveal a sophisticated interplay between the metal center and the olefinic substrates that dictates both efficiency and selectivity. The catalytic cycle likely initiates with the activation of the zirconium hydride species, which facilitates the insertion of the substituted olefin into the metal-hydrogen bond, forming a transient organozirconium intermediate. This intermediate subsequently undergoes a transmetallation event with the styrene boronic acid pinacol ester, effectively transferring the boronate moiety to the alkyl chain while regenerating the active catalytic species. This mechanism is crucial for R&D teams focusing on impurity control, as the specific coordination geometry of the zirconium catalyst helps suppress side reactions such as polymerization or over-boronation, which are common pitfalls in less selective catalytic systems. The ability to fine-tune the electronic properties of the catalyst ligands further allows for the optimization of reaction kinetics, ensuring that the desired alkenyl boronate product is formed with high regioselectivity and minimal formation of structural isomers that could compromise the quality of the final API intermediate.
How to Synthesize Alkenyl Boronate Compounds Efficiently
To implement this synthesis route effectively, operators must adhere to precise stoichiometric ratios and environmental controls to maximize yield and reproducibility. The process begins with the dissolution of the substituted olefin and the styrene boronic acid pinacol ester in an appropriate solvent such as toluene, xylene, or tetrahydrofuran, ensuring a homogeneous reaction mixture before the introduction of the catalyst. It is imperative to maintain a protective atmosphere of nitrogen or argon throughout the procedure to prevent oxidation of the sensitive zirconium species, which could lead to catalyst deactivation and reduced conversion rates. While the specific step-by-step laboratory protocol involves careful monitoring of temperature and reaction time, the detailed standardized synthesis steps are provided in the guide below for technical reference.
- Dissolve substituted olefin and styrene boronic acid pinacol ester in a suitable solvent such as toluene or xylene under inert atmosphere.
- Add the zirconium catalyst (Cp2ZrH2) to the reaction mixture with a molar ratio of substrate to catalyst between 1: 0.05 and 1:0.10.
- Heat the reaction mixture to temperatures between 60°C and 150°C for 8 to 36 hours, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this zirconium-catalyzed transfer boronation technology offers substantial benefits for procurement managers and supply chain heads focused on cost reduction in fine chemical manufacturing. The elimination of expensive noble metal catalysts, such as palladium or rhodium, in favor of abundant and low-cost zirconium complexes directly translates to significant raw material cost savings, thereby improving the overall margin structure of the production process. Additionally, the avoidance of hazardous diborane reagents reduces the regulatory burden and safety compliance costs associated with handling toxic substances, while the use of common industrial solvents like toluene ensures that the process can be easily integrated into existing manufacturing infrastructure without requiring specialized retrofits. These factors collectively enhance supply chain reliability by minimizing the risk of production delays caused by reagent shortages or safety incidents, ensuring a steady flow of high-quality intermediates to downstream customers.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with economical zirconium derivatives fundamentally alters the cost equation, removing the volatility associated with noble metal pricing and reducing the expense of catalyst recovery or disposal. Furthermore, the simplified workup procedure, which avoids complex quenching steps required for reactive boranes, lowers labor and utility costs, contributing to a more lean and efficient manufacturing operation that delivers substantial cost savings without compromising product quality.
- Enhanced Supply Chain Reliability: By utilizing stable and commercially available starting materials like substituted styrenes and pinacol boronic esters, the process mitigates the risk of supply disruptions often associated with specialized or hazardous reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, provides flexibility in sourcing and logistics, allowing for diversified supplier bases and reducing lead time for high-purity pharmaceutical intermediates even in fluctuating market conditions.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic byproducts make this method highly amenable to commercial scale-up of complex polymer additives and pharmaceutical precursors. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations and sustainability goals, which not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for global enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent's background and beneficial effect sections. These insights are intended to clarify the operational advantages and potential applications for stakeholders evaluating this methodology for their own production lines or procurement strategies. Understanding these details is essential for making informed decisions about adopting new synthetic routes that balance technical performance with economic viability.
Q: What are the primary safety advantages of this transfer boronation method over traditional hydroboration?
A: This method eliminates the need for diborane, which is highly toxic and pyrophoric, and avoids water-oxygen sensitive borane reagents, significantly enhancing operational safety and reducing hazardous waste handling costs.
Q: How does the zirconium catalyst impact the cost structure of alkenyl boronate manufacturing?
A: By utilizing early transition metal zirconium complexes which are abundant and low-cost compared to precious metals like palladium or rhodium, the process drastically reduces catalyst expenditure while maintaining high catalytic efficiency.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses stable raw materials and simple solvents like toluene, with robust temperature tolerance up to 150°C, making it highly suitable for commercial scale-up and continuous manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Boronate Supplier
As the demand for specialized intermediates grows, partnering with an experienced CDMO becomes essential for navigating the complexities of modern chemical synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the zirconium-catalyzed transfer boronation can be successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of alkenyl boronate compound meets the exacting standards required for pharmaceutical and optoelectronic applications, thereby securing your supply chain against quality fluctuations.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of technical excellence and commercial reliability.
